Abstract
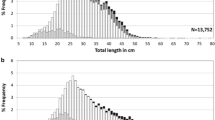
Notothenia coriiceps and Notothenia rossii are two widespread nototheniid fishes, that live sympatrically along the southern Scotia Arc from South Georgia to the South Shetland Islands. In this sector of the Southern Ocean, they experienced different exploitation rates in the past and exhibit different habitat and food preferences as adult. Aiming to evaluate the influence of these factors in shaping life history traits of these species, we compare the reproductive investment and the age structure between the species and in N. rossii, between populations inhabiting different areas. Based on histological analyses, the two species share the same pattern of gamete development in both sexes. The potential fecundity was similar and was positively related to fish size in both species, being relatively high with respect to other notothenioids and in terms of egg size at deposition. Based on sagittal otolith readings, the growth rate and maximum age recorded differed significantly between the two species. Notothenia rossii exhibited a higher growth rate and a comparatively lower maximum age than N. coriiceps. Similarly, N. rossii attained sexual maturity at the same age but at a larger size than N. coriiceps. At the intraspecific level, no differences in life history traits were observed between the populations of N. rossii collected from different areas. Consistent with the different levels of fishing pressure exerted on these species and their low resilience, a recent significant decrease over time in the maximum fish size and related reproductive potential has been observed only in the overexploited populations of N. rossii.







Similar content being viewed by others
References
Barrera-Oro E (2002) Review: the role of fish in the Antarctic marine food web: differences between inshore and offshore waters in the southern Scotia Arc and west Antarctic Peninsula. Antarct Sci 14:293–309
Barrera-Oro E, Marschoff E (2007) Information on the status of fjord Notothenia rossii, Gobionotothen gibberifrons and Notothenia coriiceps in the lower South Shetland Islands, derived from the 2000–2006 monitoring program at Potter Cove. CCAMLR Sci 14:83–87
Barrera-Oro E, Marschoff ER, Casaux RJ (2000) Trends in relative abundance of fjord Notothenia rossii, Gobionotothen gibberifrons and Notothenia coriiceps at Potter Cove, South Shetland Islands, after commercial fishing in the area. CCAMLR Sci 7:43–52
Barrera-Oro E, La Mesa M, Moreira E (2014) Early life history timings in marbled rockcod (Notothenia rossii) fingerlings from the South Shetland Islands as revealed by otolith microincrement. Polar Biol 37:1099–1109
Beamish RJ, Fournier DA (1981) A method of comparing the precision of a set of age determinations. Can J Fish Aquat Sci 38:982–983
Billard R (1986) Spermatogenesis and spermatology of some teleost fish species. Reprod Nutr Dev 26:877–920
Burchett MS (1983) The life cycle of Notothenia rossii from South Georgia. Br Antarct Surv Bull 61:71–73
Calvo J, Morriconi E, Rae GA (1999) Reproductive biology of the icefish Champsocephalus esox (Günther, 1861) (Channichthyidae). Antarct Sci 11:140–149
Campana SE (2001) Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. J Fish Biol 59:197–242
Camus S, Duhamel G (1985) Ponte et développement embryonnaire de Notothenia rossii rossii (Richardson 1844). Nototheniidae des Iles erKguelen. Cybium 9:283–293
Casaux R, Mazzotta A, Barrera-Oro E (1990) Seasonal aspects of the biology and diet of nearshore notothenioid fish at Potter Cove, South Shetland Islands, Antarctica. Polar Biol 11:63–72
Chambers RC (1997) Environmental influences on egg nd propagule sizes in marine fishes. In: Chambers RC, Trippel EA (eds) Early life history and recruitment in fish populations. Chapman and Hall, London, pp 63–102
Chang WYB (1982) A statistical method for evaluating the reproducibility of age determination. Can J Fish Aquat Sci 39:1208–1210
Cheng CC, Chen L, Near TJ, Jin Y (2003) Functional antifreeze glycoprotein genes in temperate-water New Zealand nototheniid fish infer an Antarctic evolutionary origin. Mol Biol Evol 20:1897–1908
DeWitt HH, Heemstra PC, Gon O (1990) Nototheniidae. In: Gon O, Heemstra PC (eds) Fishes of the Southern Ocean. JLB Smith Institute of Ichthyology, Grahamstown, pp 279–331
Duarte CM, Alcaraz M (1989) To produce many small or few large eggs: a size independent reproductive tactic of fish. Oecologia 80:401–404
Eastman JT (1993) Antarctic fish biology: evolution in a unique environment. Academic Press, San Diego
Eastman JT, Eakin RR (2000) An updated species list for notothenioid fish (Perciformes; Notothenioidei), with comments of Antarctic species. Arch Fish Mar Res 48:11–20
Eastman JT, Barrera-Oro E, Moreira E (2011) Adaptive radiation at a low taxonomic level: divergence in buoyancy of the ecologically similar Antarctic fish Notothenia coriiceps and N. rossii. Mar Ecol Prog Ser 438:195–206
Everson I (1970a) Reproduction in Notothenia neglecta Nybelin. Br Antarct Surv Bull 23:81–92
Everson I (1970b) The population dynamics and energy budget of Notothenia neglecta Nybelin at Signy Island, South Orkney Islands. Br Antarct Surv Bull 23:25–50
Everson I (1980) Antarctic fish age determination methods. BIOMASS Handb 8:1–24
Evseenko SA, Kock KH, Nevinsky MM (1995) Early life history of the Patagonian toothfish, Dissostichus eleginoides Smitt, 1898 in the Atlantic sector of the Southern Ocean. Antarct Sci 7:221–226
Freytag G (1980) Length, age and growth of Notothenia rossii marmorata Fischer 1885 in the West Antarctic waters. Arch Fisch Wiss 30:39–66
Gon O, Heemstra PC (1990) Fishes of the Southern Ocean. J. L. B. Smith Institute of Ichthyology, Grahamstown.
Grier HJ, Linton JR, Leatherland JF, De Vlaming VL (1980) Structural evidence for two different testicular types in teleost fishes. Amer. J Anat 159:331–345
Hunter JR, Macewicz BJ, Lo NC, Kimbrell CA (1992) Fecundity, spawning and maturity of female Dover sole Microstomus pacificus, with an evaluation of assumption and precision. Fish Bull 90:101–128
Hutchings JA, Baum JK (2005) Measuring marine fish biodiversity: temporal changes in abundance, life history and demography. Phil Trans R Soc B (2005)360:315–338.
Hutchings JA, Fraser DJ (2008) The nature of fisheries- and farming-induced evolution. Mol Ecol 17:294–313
Knust R, Gerdes D, Mintenbeck K (2012) The expedition of the research vessel “Polarstern” to the Antarctic in 2011 (ANT-XXVII/3) (CAMBIO). Ber Polar Meeresforsch 644:1–202
Kock KH (1989) Reproduction in fish around Elephant Island. Arch FischWiss 39:171–210
Kock KH (1992) Antarctic fish and fisheries. Cambridge University Press, Cambridge
Kock KH, Kellermann A (1991) Reproduction in Antarctic notothenioid fish. Antarct Sci 3:125–150
Kock KH, Duhamel G, Hureau JC (1985) Biology and status of exploited antarctic fish stocks: a review. BIOMASS Sci Ser 6:1–143
Kock KH, Belchier M, Jones CD (2004) Is the attempt to estimate the biomass of Antarctic fish from a multi-species survey appropriate for all targeted species? Notothenia rossii in the Atlantic Ocean sector—revisited. CCAMLR Sci 11:141–153
La Mesa M, Caputo V, Rampa R, Vacchi M (2003) Macroscopic and histological analyses of gonads during the spawning season of Chionodraco hamatus (Pisces, Channichthyidae) off Terra Nova Bay, Ross Sea, Southern Ocean. Polar Biol 26:621–628
La Mesa M, Caputo V, Eastman JT (2006) Gametogenesis and reproductive strategies in some species of the Antarctic fish genus Trematomus (Nototheniidae) from Terra Nova Bay, Ross Sea. Polar Biol 29:963–970
La Mesa M, Caputo V, Eastman JT (2008) The reproductive biology of two epibenthic species of Antarctic nototheniid fish of the genus Trematomus. Antarct Sci 20(4):355–364
La Mesa M, Riginella E, Mazzoldi C, Ashford J (2014) Reproductive resilience of ice-dependent Antarctic silverfish in a rapidly changing system along the Western Antarctic Peninsula. Mar Ecol 36:235–245
Linkowski TB, Zukowski C (1980) Observation on the growth of Notothenia coriiceps neglecta Nybelin and Notothenia rossii marmorata Fischer in Admiralty Bay (King George Island, South Shetland Islands). Polish Polar Res 1:155–162
Lubzens E, Young G, Bobe J, Cerdà J (2010) Oogenesis in teleosts: how fish eggs are formed. Gen Comp Physiol 165:367–389
Lucassen M (2012) The expedition of the research vessel “Polarstern” to the Antarctic in 2012 (ANT-XXVIII/4). Ber Polar Meeresforsch 652:1–89
Marschoff E, Barrera-Oro E, Alescio N, Ainley D (2012) Slow recovery of previously depleted demersal fish at the South Shetland Islands, 1983–2010. Fish Res 125–126:206–213
Miller TJ, Crowder LB, Rice JA, Marschall EA (1988) Larval size and recruitment mechanisms in fishes: toward a conceptual framework. Can J Fish Aquat Sci 45:1657–1670
Murua H, Kraus G, Saborido-Rey F, Witthames PR, Thorsen A, Junquera S (2003) Procedures to estimate fecundity of marine fish species in relation to their reproductive strategy. J Northwest Atl Fish Sci 33:33–54
Pauly D, Watson R, Alder J (2005) Global trends in world fisheries: impacts on marine ecosystems and food security. Phil Trans R Soc B 360:5–12
Pearse AGE (1985) Histochemistry, theoretical and applied analytical technology. Churchill Livingstone, Edinburgh
Rae GA, Calvo J (1996) Histological analysis of gonadal development in Patagonotothen tessellata (Richardson 1845) (Nototheniidae: Pisces) from the Beagle Channel, Argentine. J Appl Ichthyol 12:31–38
Rice IA, Crowder LB, Marschall EA (1997) Predation on juvenile fishes: dynamic interactions between size-structured predator and prey. In: Chambers RC, Trippel EA (eds) Early life history and recruitment in fish populations. Chapman and Hall, London, pp 333–386
Rochet MJ (1998) Short-term effects of fishing on life history traits of fishes. ICES J Mar Sci 55:371–391
Rochet MJ (2000) May life history traits be used as indices of population viability? J Sea Res 44:145–157
Rowe S, Hutchings JA, Bekkevold D, Rakitin A (2004) Depensation, probability of fertilization, and the mating system of Atlantic cod (Gadus morhua L.). ICES J Mar Sci 61:1144–1150
Rutschmann S, Matschiner M, Damerau M, Muschik M, Lehmann MF, Hanel R, Salzburger W (2011) Parallel ecological diversification in Antarctic notothenioid fishes as evidence for adaptative radiation. Mol Ecol 20:4707–4721
Sapota MR (1999) Gonad development and embryogenesis of Notothenia coriiceps from South Shetlands—Antarctica. Polar Biol 22:164–168
Shandikov GA, Faleeva TI (1992) Features of gametogenesis and sexual cycles of six notothenioid fishes from East Antarctica. Polar Biol 11:615–621
Tankevich PB (1990) Growth, age and natural mortality of Notothenia rossii rossii in the Kerguelen islands area. Cybium 14:269–276
Thresher RE (1988) Latitudinal variation in egg sizes of tropical and sub-tropical North Atlantic shore fishes. Environ Biol Fishes 21:17–25
Van der Molen S, Matallanas J (2003) Oocyte development and maturity classification of Gerlachea australis from the Weddell Sea, Antarctica. Polar Biol 26:653–658
Van der Molen S, Matallanas J (2004) Reproductive biology of female Antarctic spiny plunderfish Harpagifer spinosus (Notothenioidei: Harpagiferidae), from Iles Crozet. Antarct Sci 16:99–105
Vanella FA, Calvo J, Morriconi ER, Aureliano DR (2005) Somatic energy content and histological analysis of the gonads in Antarctic fish from the Scotia Arc. Sci Mar 69:305–316
Wallace RA, Selman K (1981) Cellular and dynamic aspects of oocyte growth in teleosts. Am Zool 21:325–343
West G (1990) Methods of assessing ovarian development in fishes: a review. Aust J Mar Freshw Res 41:199–222
White MG (1991) Age determination in antarctic fish. In: di Prisco G, Maresca B, Tota B (eds) Biology of Antarctic fish. Springer, Berlin Heidelberg New York, pp 87–100
White MG, North AW, Twelves EL, Jones S (1982) Early development of Notothenia neglecta from the Scotia Sea, Antarctica. Cybium 6:43–51
White MG, Veit RR, North AW, Robinson K (1996) Egg-shell morphology of the Antarctic fish, Notothenia rossii Richardson, and the distribution and abundance of pelagic eggs at South Georgia. Antarct Sci 8:267–271
Young EF, Rock J, Meredith MP, Belchier M, Murphy EJ, Carvalho GR (2012) Physical and behavioural influences on larval fish retention: contrasting patterns in two Antarctic fishes. Mar Ecol Prog Ser 465:201–215
Young EF, Belchier M, Hauser L, Horsburgh GJ, Meredith MP, Murphy EJ, Pascoal S, Rock J, Tysklind N, Carvalho GR (2015) Oceanography and life history predict contrasting genetic population structure in two Antarctic fish species. Evol Appl 8:486–509
Acknowledgements
We are very grateful to Alfred-Wegener-Institut, Helmholtz-Zentrum für Polar- und Meeresforschung that provided us the opportunity to collect samples during two RV Polarstern expeditions, ANT-XXVII/3 and ANT-XXVIII/4. Thanks to all the scientific staff, crew members and personnel aboard the RV Polarstern for their essential support in sampling activities. A special thanks to F. Giomi and F. Bartolini for providing gonad and otolith samples of N. rossii from South Georgia (ANT XXVII/3). We are in debit with C. Papetti for her valuable help during samples collection in South Shetland Islands, with C. Sands, P. Brickle and two anonymous referees for the helpful revision of the manuscript. This research was supported by the Italian National Program for Antarctic Research (PNRA) to MLM and MIUR (Ministero dell’Istruzione, dell’Universita` e della Ricerca) to CM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Calì, F., Riginella, E., La Mesa, M. et al. Life history traits of Notothenia rossii and N. coriiceps along the southern Scotia Arc. Polar Biol 40, 1409–1423 (2017). https://doi.org/10.1007/s00300-016-2066-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-016-2066-z