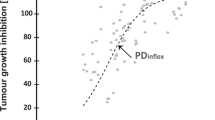
Abstract
Purpose
Pharmacokinetic–pharmacodynamic (PK–PD) models able to predict the action of anticancer compounds in tumor xenografts have an important impact on drug development. In case of anti-angiogenic compounds, many of the available models show difficulties in their applications, as they are based on a cell kill hypothesis, while these drugs act on the tumor vascularization, without a direct tumor cell kill effect. For this reason, a PK–PD model able to describe the tumor growth modulation following treatment with a cytostatic therapy, as opposed to a cytotoxic treatment, is proposed here.
Methods
Untreated tumor growth was described using an exponential growth phase followed by a linear one. The effect of anti-angiogenic compounds was implemented using an inhibitory effect on the growth function. The model was tested on a number of experiments in tumor-bearing mice given the anti-angiogenic drug bevacizumab either alone or in combination with another investigational compound. Nonlinear regression techniques were used for estimating the model parameters.
Results
The model successfully captured the tumor growth data following different bevacizumab dosing regimens, allowing to estimate experiment-independent parameters. A combination model was also developed under a ‘no-interaction’ assumption to assess the effect of the combination of bevacizumab with a target-oriented agent. The observation of a significant difference between model-predicted and observed tumor growth curves was suggestive of the presence of a pharmacological interaction that was further accommodated into the model.
Conclusions
This approach can be used for optimizing the design of preclinical experiments. With all the inherent limitations, the estimated experiment-independent model parameters can be used to provide useful indications for the single-agent and combination regimens to be explored in the subsequent development phases.







Similar content being viewed by others
References
Folkman J (2007) Angiogenesis: an organizing principle for drug discovery? Nat Rev Drug Disc 6:273–286
Cohen MH, Gootenberg J, Keegan P, Pazdur R (2007) FDA drug approval summary: bevacizumab plus FOLFOX4 as second-line treatment of colorectal cancer. Oncologist 12:356–361
Cohen MH, Johnson JR, Chen Y-F, Sridhara R, Pazdur R (2005) FDA drug approval summary: erlotinib (Tarceva®) tablets. Oncologist 10:461–466
Kane RC, Farrell AT, Saber H, Tang S, Williams G, Jee JM (2006) Sorafenib for the treatment of advanced renal cell carcinoma. Clin Cancer Res 12:7271–7278
Goodman VL, Rock EP, Dagher R, Ramchandani RP, Abraham S, Gobburu JVS et al (2007) Approval summary: sunitinib for the treatment of imatinib refractory or intolerant gastrointestinal stromal tumors and advanced renal cell carcinoma. Clin Cancer Res 13:1367–1373
Kerbel R, Folkman J (2002) Clinical translation of angiogenesis inhibitors. Nat Rev Cancer 2:727–739
Folkman J (2002) Role of angiogenesis in tumor growth and metastasis. Semin Oncol 29(suppl 16):15–18
Jain RK, Tong RT, Munn LL (2007) Effect of vascular normalization by anti-angiogenic therapy on interstitial hypertension, peritumor edema, and lymphatic metastasis: insights from a mathematical model. Cancer Res 67:2729–2735
Stephanou A, McDougall R, Anderson ARA, Chaplain MAJ (2005) Mathematical modelling of flow in 2D and 3D vascular networks: applications to anti-angiogenic and chemotherapeutic drug strategies. Math Comp Mod 41:1137–1156
D’Onofrio A, Gandolfi A (2004) Tumour eradication by anti-angiogenic therapy: analysis and extensions of the model by Hahnfeldt et al. (1999). Math Biosci 191:159–184
Ramanujan S, Koenig GC, Padera TP, Stoll BR, Jain RK (2000) Local imbalance of proangiogenic and anti-angiogenic factors: a potential mechanism of focal necrosis and dormancy in tumors. Cancer Res 60:1442–1448
Stoll BR, Migliorini C, Kadambi A, Munn LL, Jain RK (2003) A mathematical model of the contribution of endothelial progenitor cells to angiogenesis in tumors: implications for anti-angiogenic therapy. Blood 102:2555–2561
Ribba B, Colin T, Schnell S (2006) A multiscale mathematical model of cancer, and its use in analyzing irradiation therapies. Theor Biol Med Model 3:7
Hahnfeldt P, Dipak P, Folkman J, Hlatky L (1999) Tumor development under angiogenic signaling: a dynamic theory of tumor growth, treatment response, and postvascular dormancy. Cancer Res 59(4770):4775
Ribba B, Watkin E, Tod M, Girard P, Grenier E, You B, Giraudo E, Freyer G (2011) A model of vascular tumour growth in mice combining longitudinal tumour size data with histological biomarkers. Eur J Cancer 47:479–490
Simeoni M, Magni P, Cammia C, De Nicolao G, Croci V, Pesenti E et al (2004) Predictive pharmacokinetic-pharmacodynamic modeling of tumour growth kinetics in xenograft models after administrations of anticancer agents. Cancer Res 64:1094–1101
Rocchetti M, Poggesi I, Germani M, Fiorentini F, Pellizzoni C, Zugnoni P et al (2005) A PK-PD model for predicting tumour growth inhibition in mice: a useful tool in oncology drug development. Basic Clin Pharm Tox 96:265–268
Magni P, Simeoni M, Poggesi I, Rocchetti M, De Nicolao G (2006) A mathematical model to study the effects of drugs administration on tumour growth dynamics. Math Biosci 200:127–151
Rocchetti M, Simeoni M, Pesenti E, De Nicolao G, Poggesi I (2007) Predicting the active doses in humans from animal studies: a novel approach in oncology. Eur J Cancer 43:1862–1868
Liang WC, Wu X, Peale FV, Lee CV, Meng YG, Gutierrez J, Fu L, Malik AK, Greber HP, Ferrara N, Fuh G (2006) Cross-species VEGF-blocking antibodies completely inhibit the growth of human tumor xenografts and measure the contribution of stromal VEGF. J Biol Chem 281:951–961
Rocchetti M, Del Bene F, Germani M, Fiorentini F, Poggesi I, Pesenti E et al (2009) Testing additivity of anticancer agents in pre-clinical studies: a PK/PD modelling approach. Eur J Cancer 45:3336–3346
Valsasina B, Beria I, Alli C, Alzani R, Avanzi N, Ballinari D et al (2012) NMS-P937, an orally available, specific small molecule Polo-Like Kinase 1 inhibitor with antitumor activity in solid and haematological malignancies. Mol Can Ther. doi:10.1158/1535-7163.MCT-11-0765
Lin YS, Nguyen C, Mendoza J-L, Escandon E, Fei D, Meng YG, Modi NB (1999) Preclinical pharmacokinetics, interspecies scaling, and tissue distribution of a humanized monoclonal antibody against vascular endothelial growth factor. J Pharmacol Exp Ther 288:371–378
AVASTIN: EPAR Scientific discussion. European Medicin Agency. http://www.emea.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/human/000582/WC500029262.pdf, accessed November 16, 2012
James CA, Breda M, Frigerio E, Long J, Munesada K (2002) Fast turnaround bioanalysis in discovery and early clinical development. Chromatogr Suppl 55:S41–S43
Mager DE, Wyska E, Jusko WJ (2003) Diversity of mechanism-based pharmacodynamic models. Drug Met Dispos 31:510–519
Danhof M, de Lange ECM, Della Pasqua OE, Ploeger BA, Voskuyl RA (2008) Mechanism-based pharmacokinetic-pharmacodynamic (PK-PD) modeling in translational drug research. Trends Pharmacol Sci 29:186–187
Graff JR, McNulty AM, Ross Hanna K, Konicek BW, Lynch RL, Bailey SN et al (2005) The protein kinase CB—selective inhibitor, Enzastaurin (LY317615.HCl), suppresses signaling through the AKT pathway, induces apoptosis, and suppresses growth of human colon cancer and glioblastoma xenografts. Cancer Res 65:7462–7469
Yang J, Mager DE, Straubinger RM (2010) Comparison of two pharmacodynamic transduction models for the analysis of tumor therapeutic responses in model systems. AAPS J 12:1–10
Ma J, Chen C-S, Blute T, Waxman DJ (2011) Antiangiogenesis enhances intratumoral drug retention. Cancer Res. doi:10.1158/0008-5472.CAN-10-3242
Acknowledgments
The authors thank Massimo Breda and Enrico Frigerio, from Accelera S.r.l., for the bioanalytical support to the pharmacokinetic evaluations and the Experimental Therapy group in NMS for performing the pharmacological studies. The research leading to these results has received support (PM and GDN) from the Innovative Medicines Initiative Joint Undertaking under grant agreement no 115156, resources of which are composed of financial contributions from the European Union’s Seventh Framework Programme (FP7/2007-2013) and EFPIA companies’ in kind contribution. The DDMoRe project is also supported by financial contribution from Academic and SME partners. This work does not necessarily represent the view of all DDMoRe partners.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rocchetti, M., Germani, M., Del Bene, F. et al. Predictive pharmacokinetic–pharmacodynamic modeling of tumor growth after administration of an anti-angiogenic agent, bevacizumab, as single-agent and combination therapy in tumor xenografts. Cancer Chemother Pharmacol 71, 1147–1157 (2013). https://doi.org/10.1007/s00280-013-2107-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-013-2107-z