Abstract
Purpose
To evaluate the usefulness and pharmacokinetics of docetaxel in the treatment of elderly patients with advanced non-small-cell lung cancer.
Patients and methods
Chemotherapy-naive elderly patients (aged at least 76 years) with locally advanced or metastatic non-small-cell lung cancer were accrued. Eligible patients received at least two cycles of docetaxel at a dose of 60 mg/m2 on day 1 over 1 h every 3 weeks. Patients who were considered ineligible for this study were also registered. Symptom control was assessed using a questionnaire during the treatment period. The pharmacokinetics of docetaxel were evaluated in the first cycle of chemotherapy.
Results
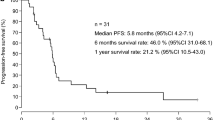
Of 35 elderly patients, 15 (43%) met the study eligibility criteria. The reasons for ineligibility consisted mainly of poor performance status, poor bone marrow function, and hypoxemia (six patients each). A total of 49 cycles of chemotherapy (median 2 cycles, range 1–12 cycles) were administered to the eligible patients, six of whom achieved a partial response (overall response rate 40%, 95% confidence interval 15–65%). The major toxicity was hematologic, with grade 3 or greater neutropenia and grade 3 neutropenic fever developing in 13 patients (87%) and five patients (33%), respectively. Symptoms, as assessed in terms of the symptom control score, did not clearly decline during the treatment period. The values (mean±SD) of Cmax, AUC0→inf, and t1/2 were 1.35±0.32 μg/ml, 1.79±0.52 μg h/ml, and 4.1±2.3 h, respectively.
Conclusions
Although the validity of the results of this study is limited due to the small sample size, docetaxel appears effective in selected elderly patients with advanced non-small-cell lung cancer.

Similar content being viewed by others
References
Balducci L, Extermann M (1997) Cancer chemotherapy in the older patient: what the medical oncologist needs to know. Cancer 80:1317–1322
Bunn PA, Kelly K (1998) New chemotherapeutic agents prolong survival and improve quality of life in non-small cell lung cancer: a review of the literature and future directions. Clin Cancer Res 4:1087–1100
Hutchins LF, Unger JM, Crowley JJ, Coltman CA Jr, Albain KS (1999) Under representation of patients 65 years of age or older in cancer-treatment trials. N Engl J Med 30:2061–2067
Kunitoh H, Watanabe K, Onoshi T, Furuse K, Niitani H, Taguchi T (1996) Phase II trial of docetaxel in previously untreated advanced non-small-cell lung cancer: a Japanese cooperative study. J Clin Oncol 14:1649–1655
Kurihara M, Shimizu H, Tsuboi K, Kobayashi K, Murakami M, Eguchi K, Shimozuma K (1999) Development of quality of life questionnaire in Japan: quality of life assessment of cancer patients receiving chemotherapy. Psychooncology 8:355–363
Lichtman SM, Villani G (2000) Chemotherapy in the elderly: pharmacologic considerations. Cancer Control 7:548–556
Marre F, Sanderink G, de Sousa G, Gaillard C, Martinet M, Rahmani R (1996) Hepatic biotransformation of docetaxel (Taxotere) in vitro: involvement of the CYP3A subfamily in humans. Cancer Res 56:1296–1302
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47:207–214
Mukohara T, Takeda K, Miyazaki M, Takifuji N, Terakawa K, Negoro S (2001) Japanese experience with second-line chemotherapy with low-dose (60 mg/m2) docetaxel in patients with advanced non-small-cell lung cancer. Cancer Chemother Pharmacol 48:356–360
National Cancer Institute—Common Toxicity Criteria, version 2.0: the Japan Clinical Oncology Group version (1999) Gan To Kagaku Ryoho 26:1084–1144
Non-small Cell Lung Cancer Collaborative Group (1995) Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. BMJ 311:899–909
Oshita F, Kurata T, Kasai T, Fakuda M, Yamamoto N, Ohe Y, Tamura T, Eguchi K, Shinkai T, Saijo N (1995) Prospective evaluation of the feasibility of cisplatin-based chemotherapy for elderly lung cancer patients with normal organ functions. Jpn J Cancer Res 86:1198–1202
Segawa Y, Watanabe K, Hiraki S, Tominaga K, Hayashi I, Harada M, Yamakido M, Ueda N, Kashimura I, Niitani H (2000) Phase I study of docetaxel and cisplatin for patients with previously untreated metastatic non-small-cell lung cancer: a Japanese cooperative study. Int J Clin Oncol 5:308–315
Sobue T (2001) Lung cancer. Gan To Kagaku Ryoho 28:163–167
Taguchi T, Furue H, Niitani H, Ishitani K, Kanamaru R, Hasegawa K, Ariyoshi Y, Noda K, Furuse K, Fukuoka M (1994) Phase I clinical trial of RP56976 (docetaxel) a new anticancer drug. Gan To Kagaku Ryoho 21:1997–2005
Takigawa N, Segawa Y, Kishino D, Fujiwara K, Ida M, Eguchi K (2000) Combination chemotherapy consisting of ifosfamide and vindesine for non-small cell lung cancer in the elderly. Nippon Kokyuki Gakkai Zasshi 38:273–277
The Elderly Lung Cancer Vinorelbine Italian Study group (1999) Effects of vinorelbine on quality of life and survival of elderly patients with advanced non-small-cell lung cancer. J Natl Cancer Inst 91:66–72
Yoshimi I, Ohshima A, Ajiki W, Tsukuma H, Sobue T (2003) A comparison of trends in the incidence rate of lung cancer by histological type in the Osaka Cancer Registry, Japan and in the Surveillance, Epidemiology and End Results Program, USA. Jpn J Clin Oncol 33:98–104
Acknowledgement
This study was supported in part by a grant from the Ministry of Health, Labor and Welfare of Japan (no. 9-25).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takigawa, N., Segawa, Y., Kishino, D. et al. Clinical and pharmacokinetic study of docetaxel in elderly non-small-cell lung cancer patients. Cancer Chemother Pharmacol 54, 230–236 (2004). https://doi.org/10.1007/s00280-004-0826-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-004-0826-x