Abstract
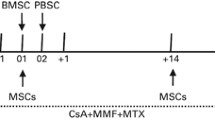
Human leukocyte antigen haploidentical hematopoietic stem-cell transplantation (haplo-HSCT) is associated with an increased risk of graft failure and severe graft-versus-host disease (GVHD). Mesenchymal stromal cells (MSCs) have been shown to support in vivo normal hematopoiesis and to display potent immunesuppressive effects. We cotransplanted the culture-expanded third-party donor-derived umbilical cord MSCs (UC-MSCs) in 50 people with refractory/relapsed hematologic malignancy undergoing haplo-HSCT with myeloablative conditioning. We observed that all patients given MSCs showed sustained hematopoietic engraftment without any adverse UC-MSC infusion-related reaction. The median times to neutrophil >0.50 × 109/L and platelet >20 × 109/L engraftment were 12.0 and 15.0 days, respectively. We did not observe an increase in severe acute GVHD (aGVHD) and extensive chronic GVHD (cGVHD), too. Grade II–IV aGVHD was observed in 12 of 50 (24.0 %) patients. cGVHD was observed in 17 of 45 (37.7 %) patients and was extensive in 3 patients. Additionally, only five patients (10.0 %) experienced relapse at a median time to progression of 192 days. The probability that patients would attain progression-free survival at 2 years was 66.0 %. The results indicate that this new strategy is effective in improving donor engraftment and reducing severe GVHD, which will provide a feasible option for the therapy of high-risk hematologic malignancy.

Similar content being viewed by others
References
Tabbara IA, Zimmerman K, Morgan C, Nahleh Z (2002) Allogeneic hematopoietic stem cell transplantation: complications and results. Arch Intern Med 162(14):1558–1566
Rizzieri DA, Koh LP, Long GD, Gasparetto C, Sullivan KM, Horwitz M, Chute J, Smith C, Gong JZ, Lagoo A, Niedzwiecki D, Dowell JM, Waters-Pick B, Liu C, Marshall D, Vredenburgh JJ, Gockerman J, Decastro C, Moore J, Chao NJ (2007) Partially matched nonmyeloablative allogenetic transplantation: clinical outcome and immune reconstitution. J Clin Oncol 25(6):690–697
Bethge WA, Haegele M, Faul C, Lang P, Schumm M, Bornhauser M, Handgretinger R, Kanz L (2006) Haploidentical allogeneic hematopoietic cell transplantation in adult with reduced-intensity conditioning and CD3/CD19 depletion: fast engraftment and low toxicity. Exp Hematol 34(12):1746–1752
Angelopoulou M, Novelli E, Grove JE (2003) Cotransplantation of human mesenchymal stem cells enhances humanmyelopoiesis and megakaryocytopoiesis in NOD/SCID mice. Exp Hematol 31(5):413–420
Le Blanc K, Rasmusson I, Sundberg B, Gotherstrom C, Hassan M, Uzunel M, Ringdén O (2004) Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 363(9419):1439–1441
Anker I‘t A, Noort WA, Scherjon SA, van der Kleijburg-Keur C, Kruisselbrink AB, van Bezooijen RL, Beekhuizen W, Willemze R, Kanhai HH, Fibbe WE (2003) Mesenchymal stem cells in human second-trimester bone marrow, liver, lung, and spleen exhibit a similar immunophenotype but a heterogeneous multilineage differentiation potential. Haematologica 88(8):845–852
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S, Gianni AM (2002) Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99(10):3838–3843
In’t Anker PS, Noort WA, Scherjon SA, Kleijburg-van der Keur C, Kruisselbrink AB, van Bezooijen RL, Beekhuizen W, Willemze R, Kanhai HH, Fibbe WE (2003) Mesenchymal stem cells in human secondtrimester bone marrow, liver, lung, and spleen exhibit a similar immunophenotype but a heterogeneous multilineage differentiation potential. Haematologica 88(8):845–852
Lee OK, Kuo TK, Chen WM, Lee KD, Hsieh SL, Chen TH (2004) Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood 103(5):1669–1675
Romanov YA, Svintsitskaya VA, Smirnov VN (2003) Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. Stem Cells 21(1):105–110
Rao MS, Matton MP (2001) Stem cells and aging: expanding the possibilities. Mech Ageing Dev 122(7):713–734
Lu LL, Liu YJ, Yang SG, Zhao QJ, Wang X, Gong W, Gong W, Han ZB, Xu ZS, Lu YX, Liu D, Chen ZZ, Han ZC (2006) Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica 91(8):1017–1026
Mickelson E, Smith A, McKinney S, Anderson G, Hansen JA (1993) A comparative study of HLA-DRB1 typing by standard serology and hybridization of non-radioactive sequence-specific oligonucleotide probes to PCR-amplified DNA. Tissue Antigens 41(2):86–93
Petersdorf EW, Smith AG, Mickelson EM, Martin PJ, Hansen JA (1991) Ten HLA-DR4 alleles defined by sequence polymorphisms within the DRB1 first domain. Immunogenetics 33(4):267–275
Copelan EA, Biggs JC, Szer J, Thompson JM, Crilley P, Brodsky I, Klein JL, Kapoor N, Harman GS, Avalos BR (1993) Allogeneic bone marrow transplantation for acute myelogenous leukemia, acute lymphocytic leukemia, and multiple myeloma following preparation with busulfan and cyclophosphamide (BuCy2). Semin Oncol 20(4 suppl 4):33–38
Socié G, Clift RA, Blaise D, Devergie A, Ringden O, Martin PJ, Remberger M, Deeg HJ, Ruutu T, Michallet M, Sullivan KM, Chevret S (2001) Busulfan plus cyclophosphamide compared with total-body irradiation plus cyclophosphamide before marrow transplantation for myeloid leukemia: long-term follow-up of 4 randomized studies. Blood 98(13):3569–3574
Blume KG, Kopecky KJ, Henslee-Downey JP, Forman SJ, Stiff PJ, LeMaistre CF, Appelbaum FR (1993) A prospective randomized comparison of total body irradiation-etoposide versus busulfan-cyclophosphamide as preparatory regimens for bone marrow transplantation in patients with leukemia who were not in first remission: a Southwest Oncology Group study. Blood 81(8):2187–2193
Koç ON, Gerson SL, Cooper BW, Dyhouse SM, Haynesworth SE, Caplan AI, Lazarus HM (2000) Rapid hematopoietic recovery after co-infusion of autologous culture-expanded human mesenchymal stem cells (hMSCs) and PBPCs in breast cancer patients receiving high dose chemotherapy. J Clin Oncol 18(2):307–316
Storb R, Deeg HJ, Whitehead J, Appelbaum F, Beatty P, Bensinger W, Buckner CD, Clift R, Doney K, Farewell V (1986) Methotrexate and cyclosporine compared with cyclosporine alone for prophylaxis of acute graft versus host disease after marrow transplantation for leukemia. N Engl J Med 314(12):729–735
Atkinson K, Horowitz MM, Gale RP, Lee MB, Rimm AA, Bortin MM (1989) Consensus among bone marrow transplanters for diagnosis, grading and treatment of chronic graft-versus-host disease. Bone Marrow Transplant 4(3):247–254
Goodrich JM, Bowden RA, Fisher L, Keller C, Schoch G, Meyers JD (1993) Ganciclovir prophylaxis to prevent cytomegalovirus disease after allogeneic marrow transplant. Ann Intern Med 118(3):173–178
Lazarus HM, Koc ON, Devine SM, Curtin P, Maziarz RT, Holland HK, Shpall EJ, McCarthy P, Atkinson K, Cooper BW, Gerson SL, Laughlin MJ, Loberiza FR Jr, Moseley AB, Bacigalupo A (2005) Cotransplantation of HLA-identical sibling culture-expanded mesenchymal stem cells and hematopoietic stem cells in hematologic malignancy patients. Biol Blood Marrow Transplant 11(5):389–398
Handgretinger R, Chen X, Pfeiffer M, Mueller I, Feuchtinger T, Hale GA, Lang P (2007) Feasibility and outcome of reduced intensity conditioning in haploidentical transplantation. Ann N Y Acad Sci 1106:279–289
Bensinger WI, Martin PJ, Storer B, Clift R, Forman SJ, Negrin R, Kashyap A, Flowers ME, Lilleby K, Chauncey TR, Storb R, Appelbaum FR (2001) Transplantation of bone marrow as compared with peripheral-blood cells from HLA-identical relatives in patients with hematologic cancers. N Engl J Med 344(3):175–181
Couban S, Simpson DR, Barnett MJ, Bredeson C, Hubesch L, Howson-Jan K, Shore TB, Walker IR, Browett P, Messner HA, Panzarella T, Lipton JH (2002) A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 100(5):1525–1531
Deeg HJ (2007) How I treat refractory acute GVHD. Blood 109(10):4119–4126
Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE (2006) Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in an onmyeloablative setting. Blood 108(6):2114–2120
Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, Lanino E, Sundberg B, Bernardo ME, Remberger M, Dini G, Egeler RM, Bacigalupo A, Fibbe W, Ringdén O (2008) Developmental Committee of the European Group for Blood and Marrow Transplantation. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 371(9624):1579–1586
Spitzer TR (2005) Haploidentical stem cell transplantation: the always present but overlooked donor. Hematology Am Soc Hematol Educ Program 390–395
Koc ON, Lazarus HM (2001) Mesenchymal stem cells: heading into the clinic. Bone Marrow Transplant 27(3):235–239
Laughlin MJ, Barker J, Bambach B, Koc ON, Rizzieri DA, Wagner JE, Gerson SL, Lazarus HM, Cairo M, Stevens CE, Rubinstein P, Kurtzberg J (2001) Hematopoietic engraftment and survival in adult recipients of umbilical-cord blood from unrelated donors. N Engl J Med 344(24):1815–1822
Rasmusson I, Ringden O, Sundberg B, Le Blanc K (2003) Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation 76(8):1208–1213
Krampera M, Glennie S, Dyson J, Scott D, Laylor R, Simpson E, Dazzi F (2003) Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101(9):3722–3729
Maitra B, Szekely E, Gjini K, Laughlin MJ, Dennis J, Haynesworth SE, Koc ON (2004) Human mesenchymal stem cells support unrelated donor hematopoietic stem cells and suppress T-cell activation. Bone Marrow Transplant 33(6):597–604
Dalton WS, Hazlehurst L, Shain K, Landowski T, Alsina M (2004) Targeting the bone marrow microenvironment in hematologic malignancies. Semin Hematol 41(2 suppl 4):1–5
Lee CK, deMagalhaes-Silverman M, Hohl RJ, Hayashi M, Buatti J, Wen BC, Schlueter A, Strauss RG, Gingrich RD (2003) Donor T-lymphocyte infusion for unrelated allogeneic bone marrow transplantation with CD3+ T-cell-depleted graft. Bone Marrow Transplant 31(2):121–128
Fung HC, Stein A, Slovak M, O’donnell MR, Snyder DS, Cohen S, Smith D, Krishnan A, Spielberger R, Bhatia R, Bhatia S, Falk P, Molina A, Nademanee A, Parker P, Rodriguez R, Rosenthal J, Sweetman R, Kogut N, Sahebi F, Popplewell L, Vora N, Somlo G, Margolin K, Chow W, Smith E, Forman SJ (2003) A long-term follow-up report on allogeneic stem cell transplantation for patients with primary refractory acute myelogenous leukemia: impact of cytogenetic characteristics on transplantation outcome. Biol Blood Marrow Transplant 9(12):766–771
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Wu and Z. Wang contributed equally to this study and should be considered as co-first authors.
Rights and permissions
About this article
Cite this article
Wu, Y., Wang, Z., Cao, Y. et al. Cotransplantation of haploidentical hematopoietic and umbilical cord mesenchymal stem cells with a myeloablative regimen for refractory/relapsed hematologic malignancy. Ann Hematol 92, 1675–1684 (2013). https://doi.org/10.1007/s00277-013-1831-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-013-1831-0