Abstract
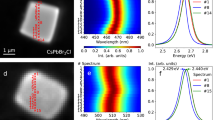
We studied the photoluminescence properties of a sample of SiO2-clathrate Melanophlogite, a crystalline microporous material which is found in nature as a rare mineral. Upon β irradiation, the material displays an intense light emission under near-UV illumination. We studied in detail this optical activity by steady-state and time-resolved photoluminescence measurements as a function of temperature. The spectroscopic properties we find can be ascribed to a population of quasi-free molecules trapped within each of the two different types of cage available in the structure of this clathrate, although the spectroscopic properties of the guest molecules are affected by their interactions with the host matrix. Based on the available data, we attribute the observed photoluminescence to trapped S2 molecules, emitting from their excited 3Σ − u or 3Π u electronic states, depending on the cage they are trapped in and on temperature. Our results have an impact on the fundamental understanding of host–guest interactions characteristic of microporous systems such as clathrates. Indeed, the data highlight that even a relatively weak coupling between quasi-free S2 molecules and the two types of cages provided by the Melanophlogite host has a surprisingly complex influence on the optical properties of the guest.






Similar content being viewed by others
References
Bondybey VE, English JH (1978) S2 fluorescence and excitation spectra in solid Ar. J Chem Phys 69:1865–1868
Bondybey VE, English JH (1980) B3Σ − u predissociation and relaxation processes in matrix isolated S2. J Chem Phys 72:3113–3122
Brewer L, Brabson GD (1966) Ultraviolet fluorescent and absorption spectra of S2 isolated in inert-gas matrices. J Chem Phys 44:3274–3278
Brewer L, Brabson GD, Meyer B (1965) UV absorption spectrum of trapped S2. J Chem Phys 42:1385–1389
Buscarino G, Agnello S, Gelardi FM, Boscaino R (2013) Properties of methyl radical trapped in amorphous SiO2 and in natural SiO2-clathrate Melanophlogite. J Non-Crist Solids 361:9–12
Buscarino G, Alessi A, Agnello S, Boizot B, Gelardi FM, Boscaino R (2014) Isolation of the CH ·3 rotor in a thermally stable inert matrix: first characterization of the gradual transition from classical to quantum behavior at low temperatures. Phys Chem Chem Phys 16:13360–13366
Gaft M, Reisfeld R, Panczer G (2005) Modern luminescence spectroscopy of minerals and materials. Springer, Berlin
Gerasimova VI, Rybaltovskii AO, Chernov PV, Zimmerer G (2002) The influence of silica glass matrix on the spectra of interstitial molecules S2. Glass Phys Chem 28:59–65
Gies H (1983) Studies on clathrasils. III. Crystal structure of melanophlogite, a natural clathrate compound of silica. Z Krist 164:247–257
Gies H, Gerke H, Liebau F (1982) Chemical composition and synthesis of melanophlogite, a clathrate compound of silica. N Jb Miner Mh 3:119–124
Harris DC, Bertolucci MD (1978) Symmetry and Spectroscopy: an introduction to vibrational and electronic spectroscopy. Oxford University Press, Oxford
Kamb B (1965) A clathrate crystalline form of silica. Science 148:232–234
Kolesov BA, Geiger CA (2003) Molecules in the SiO2-clathrate melanophlogite: a single-crystal Raman study. Am Mineral 88:1364–1368
Kortus J, Irmer G, Monecke J, Pederson MR (2000) Influence of cage structures on the vibrational modes and Raman activity of methane. Model Simul Mater Sci Eng 8:403–411
Lakowicz JR (2007) Principles of fluorescence spectroscopy. Springer, Singapore
McGee TH, Weston RE Jr (1978) Collisional quenching of fluorescence from S2(B3Σ − u ). J Chem Phys 68:1736–1743
Nakagawa T, Kihara K, Harada K (2001) The crystal structure of low melanophlogite. Am Mineral 86:1506–1512
Quick CR Jr, Weston RE Jr (1981) Fluorescence excitation spectra, lifetimes, and quenching of S2(B3Σ − u ) in the wavelength region 280–315 nm. J Chem Phys 74:4951–4959
Rebane KK, Rebane LA (1974) Small molecular ions as impurity centers in crystals. Pure Appl Chem 37:161–181
Rolfe J (1968) Emission spectra of S2 −, Se −2 , and SeS− ions in KI crystals. J Chem Phys 49:4193–4197
Shaw DM (1960) The geochemistry of scapolite. Part I. Previous work and general mineralogy. J Petrol 1:218–260
Sidike A, Kusachi I, Kobayashi S, Atobe K, Yamashita N (2008) Photoluminescence spectra of S2 − center in natural and heat-treated scapolites. Phys Chem Miner 35:137–145
Skinner BJ, Appleman DE (1963) Melanophlogite, a cubic polymorph of silica. Am Mineral 48:854–867
Sum AK, Koh CA, Sloan ED (2009) Clathrate hydrates: from laboratory science to engineering practice. Ind Eng Chem Res 48:7457–7465
Weller MT (2000) Where zeolites and oxides merge: semi-condensed tetrahedral frameworks. J Chem Soc, Dalton Trans (23):4227–4240
Yagi T, Iida E, Hirai H, Miyajima N, Kikegawa T, Bunno M (2007) High-pressure behavior of a SiO2 clathrate observed by using various pressure media. Phys Rev B 75:174115
Acknowledgments
We acknowledge the LAMP group (www.unipa.it/lamp) for stimulating discussion and useful advices. We thank G. Napoli for technical assistance in cryogenic measurements. We thank B. Boizot, École Polytechnique, Palaiseau, France, for taking care of β irradiations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Messina, F., Todaro, M., Buscarino, G. et al. Photoluminescence properties of S2 molecule trapped in Melanophlogite. Phys Chem Minerals 43, 171–179 (2016). https://doi.org/10.1007/s00269-015-0785-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-015-0785-2