Abstract
Introduction
Differentiating inflammatory from malignant head mass in the background of chronic calcific pancreatitis (CCP) is difficult, and there is no investigation which can reliably solve this dilemma. An accurate diagnosis is crucial as the treatment is different for the two cases and a failure to identify malignancy before surgery can be disastrous. We aimed to assess the accuracy of platelet–lymphocyte ratio (PLR) and to compare it with CA 19-9 in determining the nature of pancreatic head mass (PHM).
Materials and methods
Eighty-three patients, who presented with CCP and PHM between 2005 and 2011, were included in the study. Patients identified to have malignancy underwent pancreaticoduodenectomy, while those deemed to have a benign lesion underwent Frey’s procedure. Clinical features of both the groups were compared. CA 19-9 and PLR individually and in combination were compared in both groups. Receiver operating characteristic curves were used to analyze the predictive values of CA 19-9 and PLR individually and together.
Results
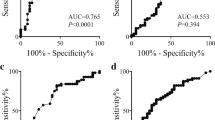
Histologically, 66.3 % had an inflammatory head mass and 33.7 % had a malignant head mass. Significant clinical features which predicted a malignancy included the presence of a head mass in CCP of tropics, older age, jaundice, sudden worsening abdominal pain, gastric outlet obstruction, and significant weight loss. Sensitivity and specificity of CA 19-9 and PLR in diagnosing malignancy were similar (85.5 vs. 81.2 and 96.4 vs. 92.8 %, respectively), on combining CA 19-9 and PLR, there was an improvement in sensitivity (94.5 %).
Conclusion
PLR is at least as good as CA 19-9 as a diagnostic marker to differentiate between malignant and inflammatory head mass in CCP. When used together, PLR improves the predictive value of serum CA 19-9.




Similar content being viewed by others
References
Zografos GN, Bean AG, Bowles M, Williamson RC (1997) Chronic pancreatitis and neoplasia: correlation or coincidence. HPB Surg 10:235–239. doi:10.1155/1997/89374
Bedi MM, Gandhi MD, Jacob G, Lekha V, Venugopal A, Ramesh H (2009) CA 19-9 to differentiate benign and malignant masses in chronic pancreatitis: is there any benefit? Indian J Gastroenterol 28:24–27. doi:10.1007/s12664-009-0005-4
Perumal S, Palaniappan R, Pillai SA, Velayutham V, Sathyanesan J (2013) Predictors of malignancy in chronic calcific pancreatitis with head mass. World J Gastrointest Surg 5(4):97–103. doi:10.4240/wjgs.v5.i4.97
Mohan V, Premalatha G, Pitchumoni CS (2003) Tropical chronic pancreatitis: an update. J Clin Gastroenterol 36:337–346. doi:10.1097/00004836-200304000-00012
Falconi M, Casetti L, Salvia R, Sartori N, Bettini R, Mascetta G et al (2000) Pancreatic head mass, how can we treat it? Chronic pancreatitis: surgical treatment. JOP 1:154–161
Schlieman MG, Ho HS, Bold RJ (2003) Utility of tumour markers in determining resectability of pancreatic cancer. Arch Surg 138:951–956. doi:10.1001/archsurg.138.9.951
Daram DR (2006) CA 19-9: not a magic marker for pancreatic cancer. South Med J 99:205. doi:10.1097/01.smj.0000204525.62861.85
Nouts A, Levy P, Voitot H, Bernades P (1998) Diagnostic value of serum CA 19-9 antigen in chronic pancreatitis and pancreatic adenocarcinoma. Gastroenterol Clin Biol 22:152–159. doi:GCB-03-1998-22-2-0399-8320-101019-ART72
Minghini A, Weireter LJ, Perry RR (1998) Specificity of elevated CA 19-9 levels in chronic pancreatitis. Surgery 124:103–105. doi:10.1016/S0039-6060(98)70082-0
Jamieson NB, Glen P, McMillan DC, McKay CJ, Foulis AK, Carter R et al (2005) Systemic inflammatory response predicts outcome in patients undergoing resection for ductal adenocarcinoma of the head of pancreas. Br J Cancer 92:21–23. doi:10.1038/sj.bjc.6602305
Salazar-Onfray F, Lopez MN, Mendoza-Naranjo A (2007) Paradoxical effects of cytokines in tumor immune surveillance and tumor escape. Cytokine Growth Factor Rev 18:171–182. doi:10.1016/j.cytogfr.2007.01.015
Bellone G, Turletti A, Artusio E, Mareschi K, Carbone A, Tibaudi D et al (1999) Tumor-associated transforming growth factor-beta and interleukin-10 contribute to a systemic Th2 immune phenotype in pancreatic carcinoma patients. Am J Pathol 155:537–547. doi:10.1016/S0002-9440(10)65149-8
Fogar P, Sperti C, Basso D, Sanzari MC, Greco E, Davoli C et al (2006) Decreased total lymphocyte counts in pancreatic cancer: an index of adverse outcome. Pancreas 32:22–28. doi:10.1097/01.mpa.0000188305.90290.50
Miglani RK, Bhateja N, Bhat RS, Kumar KVA (2013) Diagnostic role of platelet Lymphocyte Ratio (PLR) in pancreatic head masses. Indian J Surg 75(1):4–9. doi:10.1007/s12262-012-0443-6
Smith RA, Bosonnet L, Ghaneh P, Sutton R, Evans J, Healey P et al (2008) The platelet–lymphocyte ratio improves the predictive value of serum CA19-9 levels in determining patient selection for staging laparoscopy in suspected periampullary cancer. Surgery 143(5):658–666. doi:10.1016/j.surg.2007.12.014
Suzuki K, Aiura K, Kitagou M, Hoshimoto S, Takahashi S, Ueda M et al (2004) Platelet counts closely correlate with the disease-free survival interval of pancreatic cancer patients. Hepatogastroenterology 51:847–853
Brown KM, Domin C, Aranha GV, Yong S, Shoup M et al (2005) Increased preoperative platelet count is associated with decreased survival after resection for adenocarcinoma of the pancreas. Am J Surg 189:278–282. doi:10.1016/j.amjsurg.2004.11.014
Sata N, Koizumi M, Nagai H (2007) Alcoholic pancreatopathy: a proposed new diagnostic category representing the preclinical stage of alcoholic pancreatic injury. J Gastroenterol 42(Suppl 17):131–134. doi:10.1007/s00535-006-1936-5
Augustine P, Ramesh H (1992) Is tropical pancreatitis premalignant? Am J Gastroenterol 87:1005–1008. doi:10.1007/s00535-006-1936-5
Lesniak RJ, Hohenwalter MD, Taylor AJ (2002) Spectrum of causes of pancreatic calcifications. Am J Roentgenol 178:79–86. doi:10.2214/ajr.178.1.1780079
Kwon HC, Kim SH, Oh SY, Lee S, Lee JH et al (2012) Clinical significance of preoperative neutrophil–lymphocyte versus platelet–lymphocyte ratio in patients with operable colorectal cancer. Biomarkers 17:216–222. doi:10.3109/1354750X.2012.656705
Asher V, Lee J, Innamaa A, Bali A (2011) Preoperative platelet lymphocyte ratio as an independent prognostic marker in ovarian cancer. Clin Transl Oncol 13:499–503. doi:10.1007/s12094-011-0687-9
Azab B, Shah N, Radbel J, Tan P, Bhatt V (2013) Pretreatment neutrophil/lymphocyte ratio is superior to platelet/lymphocyte ratio as a predictor of long-term mortality in breast cancer patients. Med Oncol 30:432. doi:10.1007/s12032-012-0432-4
Raungkaewmanee S, Tangjitgamol S, Manusirivithaya S, Srijaipracharoen S, Thavaramara T (2012) Platelet to lymphocyte ratio as a prognostic factor for epithelial ovarian cancer. J Gynecol Oncol 23:265–273. doi:10.3802/jgo.2012.23.4.265
Seretis C, Seretis F, Lagoudianakis E, Politou M, Gemenetzis G et al (2012) Enhancing the accuracy of platelet to lymphocyte ratio after adjustment for large platelet count: a pilot study in breast cancer patients. Int J Surg Oncol 2012:653608. doi:10.1155/2012/653608
Liu H, Wu Y, Wang Z, Yao Y, Chen F et al (2013) Pretreatment platelet-to-lymphocyte ratio (PLR) as a predictor of response to first-line platinum-based chemotherapy and prognosis for patients with non-small cell lung cancer. J Thorac Dis 5(6):783–789. doi:10.3978/j.issn.2072-1439.2013.12.34
Conflict of interest
The above doctors have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rammohan, A., Cherukuri, S.D., Palaniappan, R. et al. Preoperative Platelet–Lymphocyte Ratio Augments CA 19-9 as a Predictor of Malignancy in Chronic Calcific Pancreatitis. World J Surg 39, 2323–2328 (2015). https://doi.org/10.1007/s00268-015-3087-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-015-3087-4