Abstract
Purpose
Malignant pleural mesothelioma (MPM) is a disease with poor prognosis despite multimodal therapy but there is variation in survival between patients. Prognostic information is therefore potentially valuable in managing patients, particularly in the context of clinical trials where patients could be stratified according to risk. Therefore we have evaluated the prognostic ability of parameters derived from baseline 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography (18F-FDG PET/CT).
Methods
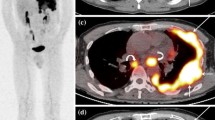
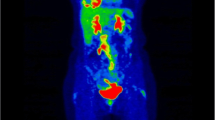
In order to determine the relationships between metabolic activity and prognosis we reviewed all 18F-FDG PET/CT scans used for pretreatment staging of MPM patients in our institution between January 2005 and December 2011 (n = 60) and measured standardised uptake values (SUV) including mean, maximum and peak values, metabolic tumour volume (MTV) and total lesion glycolysis (TLG). Overall survival (OS) or time to last censor was recorded, as well as histological subtypes.
Results
Median follow-up was 12.7 months (1.9–60.9) and median OS was 14.1 months (1.9–54.9). By univariable analysis histological subtype (p = 0.013), TLG (p = 0.024) and MTV (p = 0.038) were significantly associated with OS and SUVmax was borderline (p = 0.051). On multivariable analysis histological subtype and TLG were associated with OS but at borderline statistical significance (p = 0.060 and 0.058, respectively). No statistically significant differences in any PET parameters were found between the epithelioid and non-epithelioid histological subtypes.
Conclusion
18F-FDG PET/CT parameters that take into account functional volume (MTV, TLG) show significant associations with survival in patients with MPM before adjusting for histological subtype and are worthy of further evaluation to determine their ability to stratify patients in clinical trials.

Similar content being viewed by others
References
Brown T, Darnton A, Fortunato L, Rushton L, British Occupational Cancer Burden Study Group. Occupational cancer in Britain. Respiratory cancer sites: larynx, lung and mesothelioma. Br J Cancer 2012;107:S56–70.
Muers MF, Stephens RJ, Fisher P, Darlison L, Higgs CM, Lowry E, et al. Active symptom control with or without chemotherapy in the treatment of patients with malignant pleural mesothelioma (MS01): a multicentre randomised trial. Lancet 2008;371:1685–94.
Cao C, Yan TD, Bannon PG, McCaughan BC. A systematic review of extrapleural pneumonectomy for malignant pleural mesothelioma. J Thorac Oncol 2010;5:1692–703.
Juweid ME, Cheson BD. Positron-emission tomography and assessment of cancer therapy. N Engl J Med 2006;354:496–507.
Scott AM. Current status of positron emission tomography in oncology. Int Med J 2001;31:27–36.
Bénard F, Sterman D, Smith RJ, Kaiser LR, Albelda SM, Alavi A. Prognostic value of FDG PET imaging in malignant pleural mesothelioma. J Nucl Med 1999;40:1241–5.
Flores RM. The role of PET in the surgical management of malignant pleural mesothelioma. Lung Cancer 2005;49:S27–32.
Erasmus JJ, Truong MT, Smythe WR, Munden RF, Marom EM, Rice DC, et al. Integrated computed tomography-positron emission tomography in patients with potentially resectable malignant pleural mesothelioma: staging implications. J Thorac Cardiovasc Surg 2005;129:1364–70.
Schneider DB, Clary-Macy C, Challa S, Sasse KC, Merrick SH, Hawkins R, et al. Positron emission tomography with f18-fluorodeoxyglucose in the staging and preoperative evaluation of malignant pleural mesothelioma. J Thorac Cardiovasc Surg 2000;120:128–33.
Sharif S, Zahid I, Routledge T, Scarci M. Does positron emission tomography offer prognostic information in malignant pleural mesothelioma? Interact Cardiovasc Thorac Surg 2011;12:806–11.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med 2009;50:122S–50S.
Uto F, Shiba E, Onoue S, Yoshimura H, Takada M, Tsuji Y, et al. Phantom study on radiotherapy planning using PET/CT–delineation of GTV by evaluating SUV. J Radiat Res 2010;51:157–64.
Boucek J, Francis RJ, Green AJ. Automated approach to identification and quantitation of tumour volumes in chemotherapy monitoring using FDG PET. J Nucl Med 2005;46:464P.
Lang-Lazdunski L, Bille A, Lal R, Crane P, McLean E, Landau D, et al. Pleurectomy/decortication is superior to extrapleural pneumonectomy in the multimodality management of patients with malignant pleural mesothelioma. J Thorac Oncol 2012;7:737–43.
Kaira K, Serizawa M, Koh Y, Takahashi T, Hanaoka H, Oriuchi N, et al. Relationship between 18F-FDG uptake on positron emission tomography and molecular biology in malignant pleural mesothelioma. Eur J Cancer 2012;48:1244–54.
Lee ST, Ghanem M, Herbertson RA, Berlangieri SU, Byrne AJ, Tabone K, et al. Prognostic value of 18F-FDG PET/CT in patients with malignant pleural mesothelioma. Mol Imaging Biol 2009;11:473–9.
Genestreti G, Moretti A, Piciucchi S, Tiseo M, Bersanelli M, Scarlattei M, et al. Prognostic value of 18F-FDG standard uptake value by integrated PET/CT in the staging of malignant pleural mesothelioma. Technol Cancer Res Treat 2012;11:163–7.
Nowak AK, Francis RJ, Phillips MJ, Millward MJ, van der Schaaf AA, Boucek J, et al. A novel prognostic model for malignant mesothelioma incorporating quantitative FDG-PET imaging with clinical parameters. Clin Cancer Res 2010;16:2409–17.
Francis RJ, Byrne MJ, van der Schaaf AA, Boucek JA, Nowak AK, Phillips M, et al. Early prediction of response to chemotherapy and survival in malignant pleural mesothelioma using a novel semiautomated 3-dimensional volume-based analysis of serial 18F-FDG PET scans. J Nucl Med 2007;48:1449–58.
Melton GB, Lavely WC, Jacene HA, Schulick RD, Choti MA, Wahl RL, et al. Efficacy of preoperative combined 18-fluorodeoxyglucose positron emission tomography and computed tomography for assessing primary rectal cancer response to neoadjuvant therapy. J Gastrointest Surg 2007;11:961–9.
Larson SM, Erdi Y, Akhurst T, Mazumdar M, Macapinlac HA, Finn RD, et al. Tumor treatment response based on visual and quantitative changes in global tumor glycolysis using PET-FDG imaging. The visual response score and the change in total lesion glycolysis. Clin Positron Imaging 1999;2:159–71.
Schaefer NG, Veit-Haibach P, Soyka JD, Steinert HC, Stahel RA. Continued pemetrexed and platin-based chemotherapy in patients with malignant pleural mesothelioma (MPM): value of 18F-FDG-PET/CT. Eur J Radiol 2012;81:e19–25.
Veit-Haibach P, Schaefer NG, Steinert HC, Soyka JD, Seifert B, Stahel RA. Combined FDG-PET/CT in response evaluation of malignant pleural mesothelioma. Lung Cancer 2010;67:311–7.
Lee HY, Hyun SH, Lee KS, Kim BT, Kim J, Shim YM, et al. Volume-based parameter of 18F-FDG PET/CT in malignant pleural mesothelioma: prediction of therapeutic response and prognostic implications. Ann Surg Oncol 2010;17:2787–94.
Kadota K, Kachala SS, Nitadori J, Suzuki K, Dunphy MP, Sima CS, et al. High SUVmax on FDG-PET indicates pleomorphic subtype in epithelioid malignant pleural mesothelioma: supportive evidence to reclassify pleomorphic as nonepithelioid histology. J Thorac Oncol 2012;7:1192–7.
Genestreti G, Moretti A, Piciucchi S, Giovannini N, Galassi R, Scarpi E, et al. FDG PET/CT response evaluation in malignant pleural mesothelioma patients treated with talc pleurodesis and chemotherapy. J Cancer 2012;3:241–5.
Pleural mesothelioma. In: Edge SB, Byrd DR, Compton CC, et al., editors. AJCC cancer staging manual. 7th ed. New York: Springer; 2010. p. 271–7.
Acknowledgments
We thank Paul Bassett (BSc, MSc, FRSS) for statistical advice and analysis. The authors acknowledge support from the NIHR Biomedical Research Centre of Guys & St Thomas’ NHS Trust in partnership with Kings College London and the King’s College London and University College London Comprehensive Cancer Imaging Centre funded by the CRUK and EPSRC in association with the MRC and DoH (England).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klabatsa, A., Chicklore, S., Barrington, S.F. et al. The association of 18F-FDG PET/CT parameters with survival in malignant pleural mesothelioma. Eur J Nucl Med Mol Imaging 41, 276–282 (2014). https://doi.org/10.1007/s00259-013-2561-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2561-1