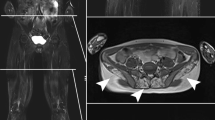
Abstract
Objective
To determine the etiologies of myonecrosis in oncology patients and to assess interobserver variability in interpreting its MRI features.
Materials and methods
Pathology records in our tertiary cancer hospital were searched for proven myonecrosis, and MRIs of affected regions in those patients were identified. MRI reports that suggested myonecrosis were also identified. Each MRI was reviewed independently by 2 out of 6 readers to assess anatomical site, size, and signal intensities of muscle changes, and the presence of the previously reported stipple sign (enhancing foci within a region defined by rim enhancement). The stipple sign was assessed again, weeks after a training session. Cohen kappa and percentage agreement were calculated. Medical records were reviewed for contemporaneous causes of myonecrosis.
Results
MRI reports in 73 patients suggested the diagnosis of myonecrosis; pathological proof was available in another 2. Myonecrosis was frequently associated with radiotherapy (n = 34 patients, 45 %); less frequent causes included intraoperative immobilization, trauma, therapeutic embolization, ablation therapy, exercise, and diabetes. Myonecrosis usually involved the lower extremity, the pelvis, and the upper extremity; mean size was 13.0 cm. The stipple sign was observed in 55–100 % of patients at first assessment (κ = 0.09–0.42; 60–80 % agreement) and 55–100 % at second (κ = 0.0–0.58; 72–90 % agreement). Enhancement surrounded myonecrosis in 55–100 % patients (κ = 0.03–0.32; 58–70 % agreement).
Conclusion
Myonecrosis in oncology patients usually occurred after radiotherapy, and less commonly after intraoperative immobilization, trauma, therapeutic embolization, ablation therapy, exercise, or diabetes. Although interobserver variability for MRI features of myonecrosis exists (even after focused training), a combination of findings facilitates diagnosis and conservative management.




Similar content being viewed by others
References
Horton WB, Taylor JS, Ragland TJ, Subauste AR. Diabetic muscle infarction: a systematic review. BMJ Open Diabetes Res Care. 2015;3(1):e000082.
Trujillo-Santos AJ. Diabetic muscle infarction. Diabetes Care. 2003;26:211–5.
Goswami P, Baruah MP. The role of MRI in diagnosis of diabetic muscle infarction: an underdiagnosed entity. Int J Endocrinol Metab. 2011;9:353–5.
May DA, Disler DG, Jones EA, Balkissoon AA, Manaster BJ. Abnormal signal intensity in skeletal muscle at MR imaging: Patterns, pearls, and pitfalls. Radiographics. 2000;20:295–315.
Cheng YC, Lan HH, Shih CH, Chen CC, Lee SK. Magnetic resonance imaging of rhabdomyolysis: Muscle necrosis versus ischemia. J Radiol Sci. 2013;38:143–8.
Lu CH, Tsang YM, Yu CW, Wu MZ, Hsu CY, Shih TT. Rhabdomyolysis: magnetic resonance imaging and computed tomography findings. J Comput Assist Tomogr. 2007;31:368–74.
Viera AJ, Garrett JM. Understanding interobserver agreement: the kappa statistic. Fam Med. 2005;37:360–3.
Kattapuram TM, Suri R, Rosol MS, Rosenberg AE, Kattapuram SV. Idiopathic and diabetic skeletal muscle necrosis: evaluation by magnetic resonance imaging. Skeletal Radiol. 2005;34:203–9.
Jelinek JS, Murphey MD, Aboulafia AJ, Dussault RG, Kaplan PA, Snearly WN. Muscle infarction in patients with diabetes mellitus: MR imaging findings. Radiology. 1999;211:241–7.
Huang BK, Monu JUV, Doumanian J. Diabetic myopathy: MRI patterns and current trends. Am J Roentgenol. 2010;195:198–204.
Vanholder R, Sever MS, Erek E, Lameire N. Rhabdomyolysis. J Am Soc Nephrol. 2000;11:1553–61.
Hamann PD, Cooper RG, McHugh NJ, Chinoy H. Statin-induced necrotizing myositis—a discrete autoimmune entity within the “statin-induced myopathy spectrum”. Autoimmun Rev. 2013;12:1177–81.
Melli G, Chaudhry V, Cornblath DR. Rhabdomyolysis: an evaluation of 475 hospitalized patients. Medicine (Baltimore). 2005;84:377–85.
Khan FY. Rhabdomyolysis: a review of the literature. Neth J Med. 2009;67:272–83.
Theodorou DJ, Theodorou SJ, Kakitsubata Y. Skeletal muscle disease: patterns of MRI appearances. Br J Radiol. 2012;85(1020):e1298–308.
Burn CC, Pritchard JC, Whay HR. Effects of prevalence on observer agreement: a subjective assessment of working donkey behavior as an example. Proceedings of Measuring Behavior 2008. Maastricht, The Netherlands. 2008.
Velcheti V, Gilstrap E, Bradley J, Govindan R. Radiation-induced myonecrosis presenting as a subcutaneous mass after combined modality therapy for non-small cell lung cancer. J Thorac Oncol. 2007;2:875–6.
Redvanly RD, Hudgins PA, Gussack GS, et al. CT of muscle necrosis following radiation therapy in a patient with head and neck malignancy. Am J Neuroradiol. 1992;13:220–2.
Becker M, Schroth G, Zbaren P, et al. Long-term changes induced by high-dose irradiation of the head and neck region: Imaging findings. Radiographics. 1997;17:5–26.
Terezakis SA, Heron DE, Lavigne RF, Diehn M, Loo Jr BW. What the diagnostic radiologist needs to know about radiation oncology. Radiology. 2011;261:30–44.
Ahmad FU, Madhavan K, Trombly R, Levi AD. Anterior thigh compartment syndrome and local myonecrosis after posterior spine surgery on a Jackson table. World Neurosurg. 2012;78:553.e5–8.
Bostanjian D, Anthone GJ, Hamoui N, Crookes PF. Rhabdomyolysis of gluteal muscles leading to renal failure: A potentially fatal complication of surgery in the morbidly obese. Obes Surg. 2003;13:302–5.
Brown JA. Lateral gluteal myonecrosis in a patient undergoing partial nephrectomy in the modified flank position. Urology. 2003;61:462.
Ward E, Munk PL, Rashid F, Torreggiani WC. Musculoskeletal interventional radiology: radiofrequency ablation. Radiol Clin North Am. 2008;46:599–610.
Rybak LD. Fire and ice: thermal ablation of musculoskeletal tumors. Radiol Clin North Am. 2009;47:455–69.
Seror O. Ablative therapies: advantages and disadvantages of radiofrequency, cryotherapy, microwave and electroporation methods, or how to choose the right method for an individual patient? Diagn Interv Imaging. 2015;96:617–24.
Robinson D, Yassin M, Nevo Z. Cryotherapy of musculoskeletal tumors-from basic science to clinical results. Technol Cancer Res Treat. 2004;3:371–5.
Volkmer D, Sichlau M, Rapp TB. The use of radiofrequency ablation in the treatment of musculoskeletal tumors. J Am Acad Orthop Surg. 2009;17:737–43.
Kanso F, Nahon P, Blaison D, Trinchet JC, Beaugrand M, Seror O, et al. Diaphragmatic necrosis after radiofrequency ablation of hepatocellular carcinoma: a successful surgical repair. Clin Res Hepatol Gastroenterol. 2013;37:e59–63.
Liu HJ, Chen TS, Lee RC, Ho DM, Lin JT, Chu LS, et al. Abdominal wall necrosis following transcatheter arterial chemoembolization for hepatocellular carcinoma. Zhonghua Yi Xue Za Zhi (Taipei). 2000;63:838–43.
Suzuki T, Shindo M, Kataoka Y, Kobayashi I, Nishimaki H, Yamamoto S, et al. Clinical characteristics of pelvic fracture patients with gluteal necrosis resulting from transcatheter arterial embolization. Arch Orthop Trauma Surg. 2005;125:448–52.
Yasumura K, Ikegami K, Kamohara T, Nohara Y. High incidence of ischemic necrosis of the gluteal muscle after transcatheter angiographic embolization for severe pelvic fracture. J Trauma. 2005;58:985–90.
Chen TY, Yeh YW, Kuo SC, Chen CY, Lin TP, Chang CC. Diphenhydramine dependence through deep intramuscular injection resulting in myonecrosis and prolonged QT interval. J Clin Pharm Ther. 2014;39:325–7.
Zurmeyer S, Fotopoulou C, Braicu E, Schlichting U, Sehouli J. Clostridium septicum can cause distant myonecrosis in patients with ovarian cancer. Anticancer Res. 2013;33:1585–9.
Jain V, Kiran P, Dhal A. Limb salvage after clostridial myonecrosis of upper limb caused by intramuscular injection. J Hand Surg Eur Vol. 2013;38:444–6.
O’Dwyer HM, Al-Nakshabandi NA, Al-Muzahmi K, Ryan A, O’Connell JX, Munk PL. Calcific myonecrosis: keys to recognition and management. Am J Roentgenol. 2006;187:67–76.
Ryu KN, Bae DK, Park YK, Lee JH. Calcific tenosynovitis associated with calcific myonecrosis of the leg: Imaging features. Skeletal Radiol. 1996;25:273–5.
Tuncay IC, Demirors H, Isiklar ZU, Agildere M, Demirhan B, Tandogan RN. Calcific myonecrosis. Int Orthop. 1999;23:68–70.
Acknowledgements
The authors thank Ramon Sosa and Sumar Hayan for their assistance with data management.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research was funded in part through the NIH/NCI Cancer Center Support Grant P30 CA008748.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Cunningham, J., Sharma, R., Kirzner, A. et al. Acute myonecrosis on MRI: etiologies in an oncological cohort and assessment of interobserver variability. Skeletal Radiol 45, 1069–1078 (2016). https://doi.org/10.1007/s00256-016-2389-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-016-2389-4