Abstract
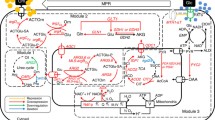
Industrial synthesis of l-carnitine is currently performed by whole-cell biotransformation of industrial waste products, mostly d-carnitine and cronobetaine, through specific bacterial species. No comparable system has been established using eukaryotic microorganisms, even though there is a significant and growing international demand for either the pure compound or carnitine-enriched consumables. In eukaryotes, including the fungus Neurospora crassa, l-carnitine is biosynthesized through a four-step metabolic conversion of trimethyllysine to l-carnitine. In contrast, the industrial yeast, Saccharomyces cerevisiae lacks the enzymes of the eukaryotic biosynthesis pathway and is unable to synthesize carnitine. This study describes the cloning of all four of the N. crassa carnitine biosynthesis genes and the reconstruction of the entire pathway in S. cerevisiae. The engineered yeast strains were able to catalyze the synthesis of l-carnitine, which was quantified using hydrophilic interaction liquid chromatography electrospray ionization mass spectrometry (HILIC-ESI-MS) analyses, from trimethyllysine. Furthermore, the yeast threonine aldolase Gly1p was shown to effectively catalyze the second step of the pathway, fulfilling the role of a serine hydroxymethyltransferase. The analyses also identified yeast enzymes that interact with the introduced pathway, including Can1p, which was identified as the yeast transporter for trimethyllysine, and the two yeast serine hydroxymethyltransferases, Shm1p and Shm2p. Together, this study opens the possibility of using an engineered, carnitine-producing yeast in various industrial applications while providing insight into possible future strategies aimed at tailoring the production capacity of such strains.





Similar content being viewed by others
References
Aouida M, Rubio-Texeira M, Rubio Texeira M, Thevelein JM, Poulin R, Ramotar D (2013) Agp2, a member of the yeast amino acid permease family, positively regulates polyamine transport at the transcriptional level. PLoS One 8:e65717. doi:10.1371/journal.pone.0065717
Berben G, Dumont J, Gilliquet V, Bolle PA, Hilger F (1991) The YDp plasmids: a uniform set of vectors bearing versatile gene disruption cassettes for Saccharomyces cerevisiae. Yeast 7:475–477. doi:10.1002/yea.320070506
Bernal V, Sevilla A, Cánovas M, Iborra JL (2007) Production of L-carnitine by secondary metabolism of bacteria. Microb Cell Fact 6:31. doi:10.1186/1475-2859-6-31
Bieber LL (1988) Carnitine. Annu Rev Biochem 57:261–283. doi:10.1146/annurev.bi.57.070188.001401
Bonafé L, Berger MM, Que YA, Mechanick JI (2014) Carnitine deficiency in chronic critical illness. Curr Opin Clin Nutr Metab Care 17:200–209. doi:10.1097/MCO.0000000000000037
Borum PR, Broquist HP (1977) Purification of S-adenosylmethionine: epsilon-N-L-lysine methyltransferase. The first enzyme in carnitine biosynthesis. J Biol Chem 252:5651–5655
Brass EP (2004) Carnitine and sports medicine: use or abuse? Ann N Y Acad Sci 1033:67–78. doi:10.1196/annals.1320.006
Calabrese V, Giuffrida Stella AM, Calvani M, Butterfield DA (2006) Acetylcarnitine and cellular stress response: roles in nutritional redox homeostasis and regulation of longevity genes. J Nutr Biochem 17:73–88. doi:10.1016/j.jnutbio.2005.03.027
Canelas AB, ten Pierick A, Ras C, Seifar RM, van Dam JC, van Gulik WM, Heijnen JJ (2009) Quantitative evaluation of intracellular metabolite extraction techniques for yeast metabolomics. Anal Chem 81:7379–7389. doi:10.1021/ac900999t
Casey GP, Xiao W, Rank GH (1988) A convenient dominant selection marker for gene transfer in industrial strains of Saccharomyces yeast: SMRI encoded resistance to the herbicide sulfometuron methyl. J Inst Brew 94:93–97. doi:10.1002/j.2050-0416.1988.tb04564.x
Chen Y, Abbate M, Tang L, Cai G, Gong Z, Wei R, Zhou J, Chen X (2014) L-Carnitine supplementation for adults with end-stage kidney disease requiring maintenance hemodialysis: a systematic review and meta-analysis. Am J Clin Nutr 99:408–422. doi:10.3945/ajcn.113.062802
Chern MK, Pietruszko R (1995) Human aldehyde dehydrogenase E3 isozyme is a betaine aldehyde dehydrogenase. Biochem Biophys Res Commun 213:561–568. doi:10.1006/bbrc.1995.2168
Davis RH, de Serres FJ (1970) Metabolism of amino acids and amines part A. Methods Enzymol 17:79–143. doi:10.1016/0076-6879(71)17168-17176
DiNicolantonio JJ, Lavie CJ, Fares H, Menezes AR, O’Keefe JH (2013) L-carnitine in the secondary prevention of cardiovascular disease: systematic review and meta-analysis. Mayo Clin Proc 88:544–551. doi:10.1016/j.mayocp.2013.02.007
Dunn WA, Rettura G, Seifter E, Englard S (1984) Carnitine biosynthesis from γ-butyrobetaine and from exogenous protein-bound 6-N-trimethyl-L-lysine by the perfused guinea pig liver. Effect of ascorbate deficiency on the in situ activity of γ-butyrobetaine hydroxylase. J Biol Chem 259:10764–10770
Franken J, Bauer FF (2010) Carnitine supplementation has protective and detrimental effects in Saccharomyces cerevisiae that are genetically mediated. FEMS Yeast Res 10:270–281
Franken J, Kroppenstedt S, Swiegers JH, Bauer FF (2008) Carnitine and carnitine acetyltransferases in the yeast Saccharomyces cerevisiae: a role for carnitine in stress protection. Curr Genet 53:347–360
Ghaddar K, Krammer E-M, Mihajlovic N, Brohée S, André B, Prévost M (2014) Converting the yeast arginine Can1 permease to a lysine permease. J Biol Chem 289:7232–7246. doi:10.1074/jbc.M113.525915
Gietz RD, Sugino A (1988) New yeast-Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 74:527–534
Henderson JW, Brooks A (2010) Improved amino acid methods using Agilent ZORBAX Eclipse Plus C18 columns for a variety of Agilent LC instrumentation and separation goals. Agilent Technologies, Wilmington, DE. http://www.chem.agilent.com/Library/applications/5990-4547EN.pdf
Henderson LM, Nelson PJ, Henderson L (1982) Mammalian enzymes of trimethyllysine conversion to trimethylaminobutyrate. Fed Proc 41:2843–2847
Kang W-K, Lee B-U, Park Y, Koh E-S, Ju E-S, Lee J, Kim H, Choi H (2010) A microorganism of Enterobacteriaceae genus harbouring genes associated with L-carnitine biosynthesis and method of producing l-carnitine using the microorganism U.S. Patent 7718414
Kikonyogo A, Pietruszko R (1996) Aldehyde dehydrogenase from adult human brain that dehydrogenates γ-aminobutyraldehyde: purification, characterization, cloning and distribution. Biochem J 316(Pt 1):317–324
Kurys G, Shah PC, Kikonygo A, Reed D, Ambroziak W, Pietruszko R (1993) Human aldehyde dehydrogenase. cDNA cloning and primary structure of the enzyme that catalyzes dehydrogenation of 4-aminobutyraldehyde. Eur J Biochem 218:311–320
LaBadie J, Dunn WA, Aronson NN (1976) Hepatic synthesis of carnitine from protein-bound trimethyl-lysine. Lysosomal digestion of methyl-lysine-labelled asialo-fetuin. Biochem J 160:85–95
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948. doi:10.1093/bioinformatics/btm404
Lin SW, Chen JC, Hsu LC, Hsieh CL, Yoshida A (1996) Human γ-aminobutyraldehyde dehydrogenase (ALDH9): cDNA sequence, genomic organization, polymorphism, chromosomal localization, and tissue expression. Genomics 34:376–380. doi:10.1006/geno.1996.0300
Longo N, di San A, Filippo C, Pasquali M (2006) Disorders of carnitine transport and the carnitine cycle. Am J Med Genet C Semin Med Genet 142C:77–85. doi:10.1002/ajmg.c.30087
McCluskey K, Wiest A, Plamann M (2010) The Fungal Genetics Stock Center: a repository for 50 years of fungal genetics research. J Biosci 35:119–126
Meyer H-P, Robins KT (2005) Large scale bioprocess for the production of optically pure L-carnitine. Monatsh Chem 136:1269–1277. doi:10.1007/s00706-005-0330-y
Monschau N, Stahmann KP, Sahm H, McNeil JB, Bognar AL (1997) Identification of Saccharomyces cerevisiae GLY1 as a threonine aldolase: a key enzyme in glycine biosynthesis. FEMS Microbiol Lett 150:55–60
Moses SBG, Otero RRC, Pretorius IS (2005) Domain engineering of Saccharomyces cerevisiae exoglucanases. Biotechnol Lett 27:355–362. doi:10.1007/s10529-005-1014-8
Ramsay RR, Gandour RD, van der Leij FR (2001) Molecular enzymology of carnitine transfer and transport. Biochim Biophys Acta 1546:21–43
Servillo L, Giovane A, Cautela D, Castaldo D, Balestrieri ML (2014) Where does N(ε)-trimethyllysine for the carnitine biosynthesis in mammals come from? PLoS One 9:e84589. doi:10.1371/journal.pone.0084589
Shanahan C (2011) L-carnitine: getting noticed in a challenging market place. Retrieved from http://www.frost.com/reg/blog-display.do?id=28527
Siebert PD, Chenchik A (1993) Modified acid guanidinium thiocyanate-phenol-chloroform RNA extraction method which greatly reduces DNA contamination. Nucleic Acids Res 21:2019–2020
Sikorski RS, Hieter P (1989) A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 122:19–27
Stein R, Englard S (1981) The use of a tritium release assay to measure 6-N-trimethyl-L-lysine hydroxylase activity: synthesis of 6-N-[3-3H]trimethyl-DL-lysine. Anal Biochem 116:230–236
Swiegers JH, Dippenaar N, Pretorius IS, Bauer FF (2001) Carnitine-dependent metabolic activities in Saccharomyces cerevisiae: three carnitine acetyltransferases are essential in a carnitine-dependent strain. Yeast 18:585–595. doi:10.1002/yea.712
Swiegers JH, Vaz FM, Pretorius IS, Wanders RJA, Bauer FF (2002) Carnitine biosynthesis in Neurospora crassa: identification of a cDNA coding for epsilon-N-trimethyllysine hydroxylase and its functional expression in Saccharomyces cerevisiae. FEMS Microbiol Lett 210:19–23
Tein I, Bukovac SW, Xie ZW (1996) Characterization of the human plasmalemmal carnitine transporter in cultured skin fibroblasts. Arch Biochem Biophys 329:145–155. doi:10.1006/abbi.1996.0203
Van Roermund CW, Elgersma Y, Singh N, Wanders RJ, Tabak HF (1995) The membrane of peroxisomes in Saccharomyces cerevisiae is impermeable to NAD(H) and acetyl-CoA under in vivo conditions. EMBO J 14:3480–3486
Vaz FM, Wanders RJA (2002) Carnitine biosynthesis in mammals. Biochem J 361:417–429
Vaz FM, van Gool S, Ofman R, Ijlst L, Wanders RJ (1998) Carnitine biosynthesis: identification of the cDNA encoding human γ-butyrobetaine hydroxylase. Biochem Biophys Res Commun 250:506–510. doi:10.1006/bbrc.1998.9343
Vaz FM, van Gool S, Ofman R, Ijlst L, Wanders RJ (1999) Carnitine biosynthesis. Purification of γ-butyrobetaine hydroxylase from rat liver. Adv Exp Med Biol 466:117–124
Vaz FM, Fouchier SW, Ofman R, Sommer M, Wanders RJ (2000) Molecular and biochemical characterization of rat γ-trimethylaminobutyraldehyde dehydrogenase and evidence for the involvement of human aldehyde dehydrogenase 9 in carnitine biosynthesis. J Biol Chem 275:7390–7394
Vaz FM, Ofman R, Westinga K, Back JW, Wanders RJ (2001) Molecular and biochemical characterization of rat ε-N-trimethyllysine hydroxylase, the first enzyme of carnitine biosynthesis. J Biol Chem 276:33512–33517. doi:10.1074/jbc.M105929200
Vogel HJ (1956) A convenient growth medium for Neurospora (Medium N). Microbial Genet Bull 13:42–43
Winston F, Dollard C, Ricupero-Hovasse SL (1995) Construction of a set of convenient Saccharomyces cerevisiae strains that are isogenic to S288C. Yeast 11:53–55. doi:10.1002/yea.320110107
Winter SC (2003) Treatment of carnitine deficiency. J Inherit Metab Dis 26:171–180
Acknowledgments
Jaco Franken was supported by an NRF NPPD postdoctoral fellowship (National Research Foundation of SA, grant number 78691 (http://www.nrf.ac.za/)).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 2392 kb)
Rights and permissions
About this article
Cite this article
Franken, J., Burger, A., Swiegers, J.H. et al. Reconstruction of the carnitine biosynthesis pathway from Neurospora crassa in the yeast Saccharomyces cerevisiae . Appl Microbiol Biotechnol 99, 6377–6389 (2015). https://doi.org/10.1007/s00253-015-6561-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6561-x