Abstract
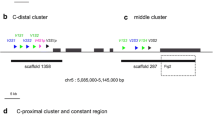
The gene segments encoding antibodies have been studied in many capacities and represent some of the best-characterized gene families in traditional animal disease models (mice and humans). To date, multiple immunoglobulin light chain (IgL) isotypes have been found in vertebrates and it is unclear as to which isotypes might be more primordial in nature. Sequence data emerging from an array of fish genome projects is a valuable resource for discerning complex multigene assemblages in this critical branch point of vertebrate phylogeny. Herein, we have analyzed the genomic organization of medaka (Oryzias latipes) IgL gene segments based on recently released genome data. The medaka IgL locus located on chromosome 11 contains at least three clusters of IgL gene segments comprised of multiple gene assemblages of the kappa light chain isotype. These data suggest that medaka IgL gene segments may undergo both intra- and inter-cluster rearrangements as a means to generate additional diversity. Alignments of expressed sequence tags to concordant gene segments which revealed each of the three IgL clusters are expressed. Collectively, these data provide a genomic framework for IgL genes in medaka and indicate that Ig diversity in this species is achieved from at least three distinct chromosomal regions.






Similar content being viewed by others
References
Bao Y, Wang T, Guo Y, Zhao Z, Li N, Zhao Y et al (2010) The immunoglobulin gene loci in the teleost Gasterosteus aculeatus. Fish Shellfish Immunol 28(1):40–48
Barreto VM, Magor BG (2011) Activation-induced cytidine deaminase structure and functions: a species comparative view. Dev Comp Immunol 35(9):991–1007
Bauer K, Hummel M, Berek C, Paar C, Rosenberger C, Kerzel S, Versmold H, Zemlin M (2007) Homology-directed recombination in IgH variable region genes from human neonates, infants and adults: implications for junctional diversity. Mol Immunol 44(11):2969–77
Belov K, Zenger KR, Hellman L, Cooper DW (2002) Echidna IgA supports mammalian unity and traditional Therian relationship. Mamm Genome 13(11):656–63
Benedict CL, Gilfillan S, Thai TH, Kearney JF (2000) Terminal deoxynucleotidyl transferase and repertoire development. Immunol Rev 175:150–157
Bengtén E, Clem LW, Miller NW, Warr GW, Wilson M (2006) Channel catfish immunoglobulins: repertoire and expression. Dev Comp Immunol 30(1–2):77–92
Chemin G, Tinguely A, Sirac C, Lechouane F, Duchez S, Cogné M, Delpy L (2010) Multiple RNA surveillance mechanisms cooperate to reduce the amount of nonfunctional Ig kappa transcripts. J Immunol 184(9):5009–17
Criscitiello MF, Flajnik MF (2007) Four primordial immunoglobulin light chain isotypes, including lambda and kappa, identified in the most primitive living jawed vertebrates. Eur J Immunol 37(10):2683–2694
Daggfeldt A, Bengtén E, Pilström L (1993) A cluster type organization of the loci of the immunoglobulin light chain in Atlantic cod (Gadus morhua L.) and rainbow trout (Oncorhynchus mykiss Walbaum) indicated by nucleotide sequences of cDNAs and hybridization analysis. Immunogenetics 38(3):199–209
Danilova N, Bussmann J, Jekosch K, Steiner LA (2005) The immunoglobulin heavy-chain locus in zebrafish: identification and expression of a previously unknown isotype, immunoglobulin Z. Nat Immunol 6(3):295–302
Danilova N, Amemiya CT (2009) Going adaptive: the saga of antibodies. Ann N Y Acad Sci 1168:130–155
Das S, Mohamedy U, Hirano M, Nei M, Nikolaidis N (2010) Analysis of the immunoglobulin light chain genes in zebra finch: evolutionary implications. Mol Biol Evol 27(1):113–120
Das S, Nikolaidis N, Klein J, Nei M (2008) Evolutionary redefinition of immunoglobulin light chain isotypes in tetrapods using molecular markers. Proc Natl Acad Sci U S A 105(43):16647–16652
Edelman G, Gally J (1962) The nature of Bence-Jones proteins. Chemical similarities to polypeptide chains of myeloma globulins and normal gammaglobulins. Med Aug 1116(1):207–227
Edholm ES, Wilson M, Bengten E (2011) Immunoglobulin light (IgL) chains in ectothermic vertebrates. Dev Comp Immunol 35(9):906–915
Edholm ES, Wilson M, Sahoo M, Miller NW, Pilström L, Wermenstam NE et al (2009) Identification of Igsigma and Iglambda in channel catfish, Ictalurus punctatus, and Iglambda in Atlantic cod, Gadus morhua. Immunogenetics 61(5):353–370
Feeney AJ (1992) Predominance of VH-D-JH junctions occurring at sites of short sequence homology results in limited junctional diversity in neonatal antibodies. J Immunol 149:222–229
Flajnik MF (2002) Comparative analyses of immunoglobulin genes: surprises and portents. Nat Rev Immunol 2(9):688–698
Francés V et al (1994) A surrogate 15 kDa JC kappa protein is expressed in combination with mu heavy chain by human B cell precursors. EMBO J 24:5937–5943
Gambón-Deza F, Sánchez-Espinel C, Magadán-Mompó S (2010) Presence of an unique IgT on the IGH locus in three-spined stickleback fish (Gasterosteus aculeatus) and the very recent generation of a repertoire of VH genes. Dev Comp Immunol 34(2):114–122
Gambón-Deza F, Sánchez-Espinel C, Mirete-Bachiller S, Magadán-Mompó S (2012) Snakes antibodies. Dev Comp Immunol 38(1):1–9
Gerashchenko V, Su D, Gladyshev N (2010) CUG start codon generates thioredoxin/glutathione reductase isoforms in mouse testes. J Biol Chem 285(7):4595–602
Gerdes T, Wabl M (2002) Physical map of the mouse lambda light chain and related loci. Immunogenetics 54(1):62–5
Ghaffari SH, Lobb CJ (1993) Structure and genomic organization of immunoglobulin light chain in the channel catfish. An unusual genomic organizational pattern of segmental genes. J Immunol 151(12):6900–6912
Ghaffari SH, Lobb CJ (1997) Structure and genomic organization of a second class of immunoglobulin light chain genes in the channel catfish. J Immunol 159(1):250–258
Giudicelli V, Chaume D, Lefranc MP (2004) IMGT/V-QUEST, an integrated software program for immunoglobulin and T cell receptor V-J and V-D-J rearrangement analysis. Nucleic Acids Res 32:W435–W440, Web Server issue
Goecks J, Nekrutenko A, Taylor J, Team G (2010) Galaxy: a comprehensive approach for supporting accessible, reproducible, and transparent computational research in the life sciences. Genome Biol 11(8):R86
Haire RN, Rast JP, Litman RT, Litman GW (2000) Characterization of three isotypes of immunoglobulin light chains and T-cell antigen receptor alpha in zebrafish. Immunogenetics 51(11):915–923
Hansen JD, Landis ED, Phillips RB (2005) Discovery of a unique Ig heavy-chain isotype (IgT) in rainbow trout: implications for a distinctive B cell developmental pathway in teleost fish. Proc Natl Acad Sci USA 102(19):6919–24
Hikima JI, Jung TS, Aoki T (2011) Immunoglobulin genes and their transcriptional control in teleosts. Dev Comp Immunol 35(9):924–936
Hsu E, Criscitiello MF (2006) Diverse immunoglobulin light chain organizations in fish retain potential to revise B cell receptor specificities. J Immunol 177(4):2452–2462
Klein J (1989) Are invertebrates capable of anticipatory immune responses? Scand J Immunol 29(5):499–505
Kumar S, Nei M, Dudley J, Tamura K (2008) MEGA: a biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief Bioinform 9(4):299–306
Lefranc MP (2011) IMGT, the International ImMunoGeneTics Information System. Cold Spring Harb protocol 6:595–603
Litman GW, Anderson MK, Rast JP (1999) Evolution of antigen binding receptors. Annu Rev Immunol 17:109–47
Lossos IS, Tibshirani R, Narasimhan B, Levy R (2000) The inference of antigen selection on Ig genes. J Immunol 165(9):5122–5126
Lu G, Moriyama E (2004) Vector NTI, a balanced all-in-one sequence analysis suite. Brief Bioinform 5(4):378–88
Lundqvist ML, Middleton DL, Radford C, Warr GW, Magor KE (2006) Immunoglobulins of the non-galliform birds: antibody expression and repertoire in the duck. Dev Comp Immunol 30(1–2):93–100
Magadán-Mompó S, Sánchez-Espinel C, Gambón-Deza F (2011) Immunoglobulin heavy chains in medaka (Oryzias latipes). BMC Evol Biol 11:165
Marchalonis JJ, Schluter SF (1990) On the relevance of invertebrate recognition and defence mechanisms to the emergence of the immune response of vertebrates. Scand J Immunol 32(1):13–20
Marchalonis JJ, Kaveri S, Lacroix-Desmazes S, Kazatchkine MD (2002) Natural recognition repertoire and the evolutionary emergence of the combinatorial immune system. FASEB J 16(8):842–8
Marianes AE, Zimmerman AM (2011) Targets of somatic hypermutation within immunoglobulin light chain genes in zebrafish. Immunology 132(2):240–255
Market E, Papavasiliou FN (2003) V(D)J recombination and the evolution of the adaptive immune system. PLoS Biol 1(1):E16
Mårtensson L, Keenan A, Licence S (2007) The pre-B-cell receptor. Curr Opin Immunol 2:137–142
Milne I, Bayer M, Cardle L, Shaw P, Stephen G, Wright F et al (2010) Tablet—next generation sequence assembly visualization. Bioinformatics 26(3):401–402
Nei M, Rooney AP (2005) Concerted and birth-and-death evolution of multigene families. Annu Rev Genet 39:121–152
Partula S, Schwager J, Timmusk S, Pilström L, Charlemagne J (1996) A second immunoglobulin light chain isotype in the rainbow trout. Immunogenetics 45(1):44–51
Pham CH, Park KS, Kim BC, Kim HN, Gu MB (2011) Construction and characterization of Japanese medaka (Oryzias latipes) hepatic cDNA library and its implementation to biomarker screening in aquatic toxicology. Aquat Toxicol 105(3–4):569–75
Pilström L (2002) The mysterious immunoglobulin light chain. Dev Comp Immunol 26(2):207–215
Rangel R et al (2005) Assembly of the kappa preB receptor requires a V kappa-like protein encoded by a germline transcript. J Biol Chem 18:17807–17814
Rast JP, Smith LC, Loza-Coll M, Hibino T, Litman GW (2006) Genomic insights into the immune system of the sea urchin. Science 314(5801):952–6
Rosnet O, Blanco-Betancourt C, Grivel K, Richter K, Schiff C (2004) Binding of free immunoglobulin light chains to VpreB3 inhibits their maturation and secretion in chicken B cells. J Biol Chem 11:10228–10236
Sablitzky F, Wildner G, Rajewsky K (1985) Somatic mutation and clonal expansion of B cells in an antigen-driven immune response. EMBO J 4(2):345–50
Sasado T, Tanaka M, Kobayashi K, Sato T, Sakaizumi M, Naruse K (2010) The National BioResource Project Medaka (NBRP Medaka): an integrated bioresource for biological and biomedical sciences. Exp Anim 59(1):13–23
Smith LC, Davidson EH (1992) The echinoid immune system and the phylogenetic occurrence of immune mechanisms in deuterostomes. Immunol Today 13(9):356–62
Solovyev V, Kosarev P, Seledsov I, Vorobyev D (2006) Automatic annotation of eukaryotic genes, pseudogenes and promoters. Genome Biol 7(Suppl1):S10
Stabenau A, McVicker G, Melsopp C, Proctor G, Clamp M, Birney E (2004) The Ensembl core software libraries. Genome Res 14(5):929–33
Stanke M, Steinkamp R, Waack S, Morgenstern B (2004) AUGUSTUS: a web server for gene finding in eukaryotes. Nucleic Acids Res 32:W309–312, Web Server issue
Takeda H (2008) Draft genome of the medaka fish: a comprehensive resource for medaka developmental genetics and vertebrate evolutionary biology. Dev Growth Differ 50(Suppl 1):S157–S166
Takeda H, Shimada A (2010) The art of medaka genetics and genomics: what makes them so unique? Annu Rev Genet 44:217–241
Teng G, Papavasiliou FN (2007) Immunoglobulin somatic hypermutation. Annu Rev Genet 41:107–120
Wang X, Parra E, Miller D (2012) A VpreB3 homologue in a marsupial, the gray short-tailed opossum, Monodelphis domestica. Immunogenetics 8:647–652
Wheeler DL, Church DM, Lash AE, Leipe DD, Madden TL, Pontius JU, Schuler GD, Schriml LM, Tatusova TA, Wagner L, Rapp BA (2001) Database resources of the National Center for Biotechnology Information. Nucleic Acids Res 29(1):11–6
Yang F, Waldbieser GC, Lobb CJ (2006) The nucleotide targets of somatic mutation and the role of selection in immunoglobulin heavy chains of a teleost fish. J Immunol 176(3):1655–1667
Zhang M, Srivastava G, Lum L (2004) The pre-B cell receptor and its function during B cell development. Cell Mol Immunol 2:89–94
Zimmerman AM, Romanowski KE, Maddox BJ (2011) Targeted annotation of immunoglobulin light chain (IgL) genes in zebrafish from BAC clones reveals kappa-like recombining/deleting elements within IgL constant regions. Fish Shellfish Immunol 31(5):697–703
Zimmerman AM, Yeo G, Howe K, Maddox BJ, Steiner LA (2008) Immunoglobulin light chain (IgL) genes in zebrafish: Genomic configurations and inversional rearrangements between (V(L)-J(L)-C(L)) gene clusters. Dev Comp Immunol 32(4):421–434
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary File 1
Table: Medaka full length cDNA libraries. The table includes the number of clones. (PDF 10 kb)
Supplementary File 2
Nucleotide sequences of IGL loci described in the medaka. Individual VLJLCL clusters in medaka IGL1, IGL2, and IGL3 regions, their annotation, and corresponding sequences in a GenBank formats are depicted. (PDF 272 kb)
Supplementary File 3
Phylogenetic analyses of medaka VL segments. An unrooted tree with representative amino acid VL sequences from each VL family and other fish VL sequences constructed using a pairwise deletion algorithm, JTT matrix, and differences by sites activated with gamma parameter 2.5. (PDF 79 kb)
Supplementary File 4
An example of a RSS-J-C coding EST. The alternative translation initiation codon CUG, shown here as CTG, is proposed to precede and open reading frame which originates a J-CL protein. The start codon (CTG) and stop codon (TGA) are shown in red. (PDF 67 kb)
Supplementary File 5
Table: Assignment of VL, JL segments and CL exons in 29 medaka IgL coding ESTs. (PDF 110 kb)
Supplementary File. 6
Analysis of VLJLCL joining in four medaka ESTs (olsp42k18, olsp40b17, olki63m21, and olki48c02). The figure depicts the joining region of alignments of germline VL, JL, CL segments. Although neither N nor P nucleotides were found imprecision in the V-J junction was found suggestive of joining regions contributing to antibody diversity in the medaka model. The four sequences are representative of all 29 ESTs analyzed. (PDF 40 kb)
Rights and permissions
About this article
Cite this article
Magadán-Mompó, S., Zimmerman, A.M., Sánchez-Espinel, C. et al. Immunoglobulin light chains in medaka (Oryzias latipes). Immunogenetics 65, 387–396 (2013). https://doi.org/10.1007/s00251-013-0678-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-013-0678-9