Abstract
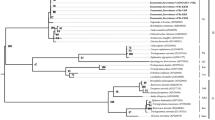
Wolbachia is a symbiont intensively studied due to its ability to interfere with their host’s reproduction, and it has been recently proposed as an alternative tool to control insect pests or vectors of diseases. The Asian citrus psyllid Diaphorina citri is an important pest of citrus since it vectors the bacterium that causes the "Huanglongbing" disease in citrus. The frequency and diversity of Wolbachia associated with D. citri is unknown, limiting the utilization of Wolbachia as an alternative strategy for insect management. Thus, we aimed to determine the natural rate of infection, to characterize the Wolbachia strains associated with this psyllid by "multilocus sequencing typing” (MLST) and wsp analysis, and to verify the association of the symbiont to particular genotypes of the host. Analysis indicated Wolbachia infects 100 % of all specimens tested from all 15 sampled populations. MLST revealed the occurrence of five new sequence types (STs) of Wolbachia, while analysis based on the wsp sequences indicated only four different types of Wolbachia. ST-173 was predominant, while the remaining STs were population specific. Analysis of the host–symbiont relationship did not reveal any particular association of Wolbachia and haplotypes or a decrease in nucleotide diversity of D. citri in populations in which more than one ST was recorded. The consequences of the diversity of STs reported are still unknown, but the fact that Wolbachia infection is fixed and that there is one ST with a broad distribution highlights the use of this symbiont as an alternative strategy to control D. citri.




Similar content being viewed by others
Reference
Aksoy S, Weiss B, Attardo G (2008) Paratransgenesis applied for the control of tsetse transmitted sleeping sickness. Advances in Experimental and Medical Biology 627:35–48
Ammar ED, Shatters RG, Lynch C, Hall DG (2012) Detection and relative titer of Candidatus liberibacter asiaticus in the salivary glands and alimentary canal of Diaphorina citri (Hemiptera: psyllidae) vector of Citrus Huanglongbing disease. Annals of the Entomological Society of America 104:526–533
Anderson CL, Karr TL (2001) Wolbachia: evolutionary novelty in a rickettsial bacteria. BMC Evolutionary Biology 1
Arthofer W, Riegler M, Avtzis DN, Stauffer C (2009) Evidence for low-titre infections in insect symbiosis: wolbachia in the bark beetle Pityogenes chalcographus (Coleoptera, Scolytinae). Environ Microbiol 11:1923–1933
Arthofer W, Riegler M, Schneider D, Krammer M, Miller WJ, Stauffer C (2009) Hidden Wolbachia diversity in field populations of the European cherry fruit fly, Rhagoletis cerasi (Diptera, Tephritidae). Mol Ecol 18:3816–3830
Arthofer W, Riegler M, Schuler H, Schneider D, Moder K, Miller WJ, Stauffer C (2011) Allele Intersection Analysis: A novel tool for multi locus sequence assignment in multiply infected hosts. Plos One 6
Atyame CM, Duron O, Tortosa P, Pasteur N, Fort P, Weill M (2011) Multiple Wolbachia determinants control the evolution of cytoplasmic incompatibilities in Culex pipiens mosquito populations. Mol Ecol 20:286–298
Baldo L, Lo N, Werren JH (2005) Mosaic nature of the Wolbachia surface protein. J Bacteriol 187:5406–5418
Baldo L, Hotopp JCD, Jolley KA, Bordenstein SR, Biber SA, Choudhury RR, Hayashi C, Maiden MCJ, Tettelin H, Werren JH (2006) Multilocus sequence typing system for the endosymbiont Wolbachia pipientis. Appl Environ Microbiol 72:7098–7110
Bass C, Field LM (2011) Gene amplification and insecticide resistance. Pest Management Science 67:886–890
Baumann P (2005) Biology of bacteriocyte-associated endosymbionts of plant sap-sucking insects. Annu Rev Microbiol 59:155–189
Benlarbi M, Ready PD (2003) Host-specific Wolbachia strains in widespread populations of Phlebotomus perniciosus and P. papatasi (Diptera: psychodidae), and prospects for driving genes into these vectors of Leishmania. Bulletin of Entomological Research 93:383–391
Bian G, Xu Y, Lu P, Xie Y, Xi Z (2010) The endosymbiotic bacterium Wolbachia induces resistance to Dengue virus in Aedes aegypti. PLoS Pathog 6:e1000833
Bove JM (2006) Huanglongbing: a destructive, newly-emerging, century-old disease of citrus. Journal of Plant Pathology 88:7–37
Braig HR, Zhou WG, Dobson SL, O’Neill SL (1998) Cloning and characterization of a gene encoding the major surface protein of the bacterial endosymbiont Wolbachia pipientis. J Bacteriol 180:2373–2378
Brownlie JC, Adamski M, Slatko B, McGraw EA (2007) Diversifying selection and host adaptation in two endosymbiont genomes. BMC Evol Biol 7:12
Buchner P (1965) Endosymbiosis of animals with plant microorganisms. Wiley, Hoboken
Chafee ME, Funk DJ, Harrison RG, Bordenstein SR (2010) Lateral phage transfer in obligate intracellular bacteria (Wolbachia): verification from natural populations. Mol Biol Evol 27:501–505
Charlat S, Duplouy A, Hornett EA, Dyson EA, Davies N, Roderick GK, Wedell N, Hurst GDD (2009) The joint evolutionary histories of Wolbachia and mitochondria in Hypolimnas bolina. BMC Evol Biol 9:64
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659
Coutinho-Abreu I, Zhu KY, Ramalho-Ortigao M (2010) Transgenesis and paratransgenesis to control insect-borne diseases and future challenges. Parasitoly International 59:1–8
Curtis CF, Sinkins SP (1998) Wolbachia as a possible means of driving genes into populations. Parasitology 116:S111–S115
Dedeine F, Vavre F, Fleury F, Loppin B, Hochberg ME, Bouletreau M (2001) Removing symbiotic Wolbachia bacteria specifically inhibits oogenesis in a parasitic wasp. Proc Natl Acad Sci U S A 98:6247–6252
Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Durvasula RV, Gumbs A, Panackal A, Kruglov O, Aksoy S, Merrifield RB, Richards FF, Beard CB (1997) Prevention of insect-borne disease: an approach using transgenic symbiotic bacteria. Proc Natl Acad Sci U S A 94:3274–3278
Durvasula RV, Gumbs A, Panackal A, Kruglov O, Taneja J, Kang AS (1999) Expression of a functional antibody fragment in the gut of Rhodnius prolixus via transgenic bacterial symbiont Rhodococcus rhodnii. Medical and Veterinary Entomology 13:115–119
Engelstadter J, Hammerstein P, Hurst GDD (2007) The evolution of endosymbiont density in doubly infected host species. J Evol Biol 20:685–695
Ewing B, Green P (1998) Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Research 8:186–194
Ewing B, Hillier L, Wendl MC, Green P (1998) Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Research 8:175–185
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567
Godfray HCJ (2010) An insect-endosymbiont conundrum. Heredity 104:237–238
Guidolin AS (2011) Diversidade genética de Diaphorina citri Kuwayama, 1908 (Hemiptera: Psyllidae) e caracterização molecular das linhagens de Wolbachia associadas. Dissertation, University of São Paulo—Escola Superior de Agricultura "Luiz de Queiroz.
Harris HL, Brennan LJ, Keddie BA, Braig HR (2010) Bacterial symbionts in insects: balancing life and death. Symbiosis 51:37–53
Hedges LM, Brownlie JC, O’Neill SL, Johnson KN (2008) Wolbachia and virus protection in insects. Science 322:702
Hoffmann AA, Montgomery BL, Popovici J, Iturbe-Ormaetxe I, Johnson PH, Muzzi F, Greenfield M, Durkan M, Leong YS, Dong Y, Cook H, Axford J, Callahan AG, Kenny N, Omodei C, McGraw EA, Ryan PA, Ritchie SA, Turelli M, O’Neill SL (2011) Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 476:454–457
Hosokawa T, Koga R, Kikuchi Y, Meng XY, Fukatsu T (2010) Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci U S A 107:769–774
Hughes GL, Ren XX, Ramirez JL, Sakamoto JM, Bailey JA, Jedlicka AE, Rasgon JL (2011) Wolbachia infections in Anopheles gambiae cells: Transcriptomic characterization of a novel host-symbiont interaction. Plos Pathogens 7
Hurst GDD, Jiggins FM, von der Schulenburg JHG, Bertrand D, West SA, Goriacheva II, Zakharov IA, Werren JH, Stouthamer R, Majerus MEN (1999) Male-killing Wolbachia in two species of insect. Proc R Soc London, Ser B 266:735–740
Inoue H, Ohnishi J, Ito T, Tomimura K, Miyata S, Iwanami T, Ashihara W (2009) Enhanced proliferation and efficient transmission of Candidatus liberibacter asiaticus by adult Diaphorina citri after acquisition feeding in the nymphal stage. Ann Appl Biol 155:29–36
Jaenike J (2009) Coupled population dynamics of endosymbionts within and between hosts. Oikos 118:353–362
Jamnongluk W, Kittayapong P, Baimai V, O’Neill SL (2002) Wolbachia infections of tephritid fruit flies: molecular evidence for five distinct strains in a single host species. Curr Microbiol 45:255–260
Jeyaprakash A, Hoy MA (2000) Long PCR improves Wolbachia DNA amplification: wsp sequences found in 76 % of sixty-three arthropod species. Insect Molecular Biology 9:393–405
Kambris Z, Blagborough AM, Pinto SB, Blagrove MSC, Godfray HCJ, Sinden RE, Sinkins SP (2010) Wolbachia stimulates immune gene expression and inhibits Plasmodium development in Anopheles gambiae. PLoS Pathogens 6(10):e1001143
Kawasaki Y, Ito M, Miura K, Kajimura H (2010) Superinfection of five Wolbachia in the alnus ambrosia beetle, Xylosandrus germanus (Blandford) (Coleoptera: curculionidae). Bulletin of Entomological Research 100:231–239
Lopes SA, Frare GF, Yamamoto PT, Ayres AJ, Barbosa JC (2007) Ineffectiveness of pruning to control citrus Huanglongbing caused by Candidatus liberibacter americanus. Eur J Plant Pathol 119:463–468
Malloch G, Fenton B, Butcher RDJ (2000) Molecular evidence for multiple infections of a new subgroup of Wolbachia in the European raspberry beetle Byturus tomentosus. Mol Ecol 9:77–90
Manjunath KL, Halbert SE, Ramadugu C, Webb S, Lee RF (2008) Detection of ‘Candidatus liberibacter asiaticus’ in Diaphorina citri and its importance in the management of citrus Huanglongbing in Florida. Phytopathology 98:387–396
McGraw EA, Merritt DJ, Droller JN, O’Neill SL (2002) Wolbachia density and virulence attenuation after transfer into a novel host. Proc Natl Acad Sci U S A 99:2918–2923
McMeniman CJ, Lane RV, Cass BN, Fong AWC, Sidhu M, Wang YF, O’Neill SL (2009) Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 323:141–144
Moreira LA, Iturbe-Ormaetxe I, Jeffery JA, Lu GJ, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M, Hugo LE, Johnson KN, Kay BH, McGraw EA, van den Hurk AF, Ryan PA, O’Neill SL (2009) A Wolbachia symbiont in Aedes aegypti limits infection with Dengue, Chikungunya, and Plasmodium. Cell 139:1268–1278
Nunes MDS, Nolte V, Schlotterer C (2008) Nonrandom Wolbachia infection status of Drosophila melanogaster strains with different mtDNA haplotypes. Mol Biol Evol 25:2493–2498
Ochman H, Wilson AC (1987) Evolution in bacteria—evidence for a universal substitution rate in cellular genomes. J Mol Evol 26:74–86
Rancès E, Ye YH, Woolfit M, McGraw EA, O’Neill SL (2012) The relative importance of innate immune priming in Wolbachia-mediated Dengue interference. PLoS Pathog 8(2):e1002548
Reuter M, Keller L (2003) High levels of multiple Wolbachia infection and recombination in the ant Formica exsecta. Mol Biol Evol 20:748–753
Riegler M, Sidhu M, Miller WJ, O’Neill SL (2005) Evidence for a global Wolbachia replacement in Drosophila melanogaster. Curr Biol 15:1428–1433
Rodriguero MS, Lanteri AA, Confalonieri VA (2010) Mito-nuclear genetic comparison in a Wolbachia infected weevil: insights on reproductive mode, infection age and evolutionary forces shaping genetic variation. BMC Evol Biol 10:340
Rottshaefer SM, Lazzaro BP (2012) No effect of Wolbachia on resistance to intracellular infection by pathogenic bacteria in Drosophila melanogaster. PLoS One 7(7):e40500
Rousset F, Vautrin D, Solignac M (1992) Molecular identification of Wolbachia the agent of cytoplasmic incompatibility in Drosophila simulans, and variability in relation with host mitochondrial types. Proc R Soc London, Ser B 247:163–168
Rousset F, Bouchon D, Pintureau B, Juchault P, Solignac M (1992) Endosymbionts responsible for various alterations of asexuality in Arthropods. Proceedings Biological Sciences 250:91–98
Ruang-areerate T, Kittayapong P (2006) Wolbachia transinfection in Aedes aegypti: a potential gene driver of dengue vectors. Proc Natl Acad Sci U S A 103:12534–12539
Sintupachee S, Milne JR, Poonchaisri S, Baimai V, Kittayapong P (2006) Closely related Wolbachia strains within the pumpkin arthropod community and the potential for horizontal transmission via the plant. Microb Ecol 51:294–301
Stouthamer R (1993) The use of sexual versus asexual wasps in biological control. Entomophaga 38:3–6
Subandiyah S, Nikoh N, Tsuyumu S, Somowiyarjo S, Fukatsu T (2000) Complex endosymbiotic microbiota of the citrus psyllid Diaphorina citri (Homoptera: psylloidea). Zoological Science 17:983–989
Sun X, Cui LW, Li ZH (2007) Diversity and phylogeny of Wolbachia infecting Bactrocera dorsalis (Diptera: tephritidae) populations from China. Environmental Entomology 36:1283–1289
Sunnucks P, Hales DF (1996) Numerous transposed sequences of mitochondrial cytochrome oxidase I-II in aphids of the genus Sitobion (Hemiptera: aphididae). Mol Biol Evol 13:510–524
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Teixeira DD, Danet JL, Eveillard S, Martins EC, Junior WCJ, Yamamoto PT, Lopes SA, Bassanezi RB, Ayres AJ, Saillard C, Bove JM (2005) Citrus Huanglongbing in Sao Paulo State, Brazil: PCR detection of the 'Candidatus' liberibacter species associated with the disease. Molecular and Cellular Probes 19:173–179
Teixeira DD, Saillard C, Eveillard S, Danet JL, da Costa PI, Ayres AJ, Bove J (2005) 'Candidatus liberibacter americanus', associated with citrus Huanglongbing (greening disease) in Sao Paulo State, Brazil. Int J Syst Evol Microbiol 55:1857–1862
Teixeira L, Ferreira A, Ashburner M (2008) The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. Plos Biology 6:e1000002
Tiwari S, Mann RS, Rogers ME, Stelinski LL (2011) Insecticide resistance in field populations of Asian citrus psyllid in Florida. Pest Management Science 67:1258–1268
Turelli M, Hoffmann AA, McKechnie SW (1992) Dynamics of cytoplasmic incompatibility and mtDNA variation natural Drosophila simulans populations. Genetics 132:713–723
Veneti Z, Clark ME, Karr TL, Savakis C, Bourtzis K (2004) Heads or tails: host–parasite interactions in the Drosophila–Wolbachia system. Appl Environ Microbiol 70:5366–5372
Wang S, Ghosh AK, Bongio N, Stebbings KA, Lampe DJ, Jacobs-Lorena M (2012) Fighting malaria with engineered symbiotic bacteria from vector mosquitoes. Proc Natl Acad Sci 109:12734–12739
Watanabe M, Miura K, Hunter MS, Wajnberg E (2011) Superinfection of cytoplasmic incompatibility-inducing Wolbachia is not additive in Orius strigicollis (Hemiptera: anthocoridae). Heredity 106:642–648
Weeks AR, Reynolds KT, Hoffmann AA, Mann H (2002) Wolbachia dynamics and host effects: what has (and has not) been demonstrated? Trends in Ecology & Evolution 17:257–262
Wong ZS, Hedges LM, Brownlie JC, Johnson KN (2011) Wolbachia-mediated antibacterial protection and immune gene regulation in Drosophila. PLoS One 6(9):e25430
Yen JH, Barr AR (1971) New hypothesis of the cause of cytoplasmic incompatibility in Culex pipiens L. Nature 232:657–658
Zabalou S, Apostolaki A, Livadaras I, Franz G, Robinson AS, Savakis C, Bourtzis K (2009) Incompatible insect technique: incompatible males from a Ceratitis capitata genetic sexing strain. Entomologia Experimentalis Et Applicata 132:232–240
Zabalou S, Riegler M, Theodorakopoulou M, Stauffer C, Savakis C, Bourtzis K (2004) Wolbachia-induced cytoplasmic incompatibility as a means for insect pest population control. Proc Natl Acad Sci U S A 101:15042–15045
Acknowledgments
Authors greatly appreciate the support of all of those that contributed in sample collection, especially those from Fundecitrus. We are also in debt with two anonymous reviewers which greatly contributed to improve the initial version of this manuscript. Thanks also to Fundecitrus, CNPq/MAPA (578797-2008/9) and FAPESP (process 2004/14215-0) for providing funds for this research.
This manuscript was reviewed by a professional science editor and by a native English-speaking copy editor to improve readability.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 21 kb)
Rights and permissions
About this article
Cite this article
Guidolin, A.S., Cônsoli, F.L. Molecular Characterization of Wolbachia Strains Associated with the Invasive Asian Citrus Psyllid Diaphorina citri in Brazil. Microb Ecol 65, 475–486 (2013). https://doi.org/10.1007/s00248-012-0150-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0150-7