Abstract
Introduction
The relationship between silent cerebral infarction (SCI) and the integrity of cognitive function is unknown. We intended to investigate whether cognitive impairment is associated with gray matter volume (GMV) in the SCI patients.
Methods
Sixty-two patients with SCI and 62 age- and gender-matched healthy controls (HC) were evaluated with P300 test, Montreal Cognitive Assessment (MoCA) test, Hamilton Anxiety Scale (HAMA), and Hamilton Depression Scale (HDRS). Whole brain high-resolution T1-weighted images were processed with SPM12b software and analyzed by voxel-based morphometry (VBM). Correlation analysis was performed between the GMV and the scores of MoCA Scale, P300 latency, P300 amplitude, HAMA, HDRS, age, and educational level.
Results
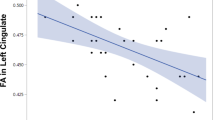
The brains of the SCI patients have a significant reduction in GMV in the left superior and inferior frontal gyrus, left superior temporal gyrus, right middle temporal gyrus, and bilateral hippocampus gyrus (p < 0.01, FDR correction). No significant increase of GMV was detected. The GMV of their frontal and temporal lobes is positively correlated with the score of MoCA scale and P300 amplitude (r ≥ 0.62, p < 0.01). The GMV of frontal, temporal, and hippocampus is negatively correlated with P300 latency (r ≤ −0.71, p < 0.05). No significant correlation between the GMV of abnormal brain regions and another two clinical characteristics was found.
Conclusion
SCI patients have impaired cognitive function and reduced GMV compared to the HC subjects. The neuropathological basis of such cognitive deficits in SCI patients might be a reduced GMV.

Similar content being viewed by others
References
Zhao J, Tang H, Sun J, Wang B, Chen S (2012) Analysis of cognitive dysfunction with silent cerebral infarction: a prospective study in Chinese patients. Metab Brain Dis 27(1):17–22
Larner AJ (2012) Screening utility of the Montreal Cognitive Assessment (MoCA): in place of-or as well as-the MMSE? Int Psychogeriatr 24(3):391–396
Das RR, Seshadri S, Beiser AS, Kelly-Hayes M, Au R, Himali JJ, Kase CS, Benjamin EJ, Polak JF, O'Donnell CJ, Yoshita M, D'Agostino RB Sr, DeCarli C, Wolf PA (2008) Prevalence and correlates of silent cerebral infarcts in the Framingham offspring study. Stroke 39(11):2929–2935
Swartz RH, Stuss DT, Gao F, Black SE (2008) Independent cognitive effects of atrophy and diffuse subcortical and thalamico-cortical cerebrovascular disease in dementia. Stroke 39(3):822–830
Poggesi A, Pantoni L, Inzitari D, Fazekas F, Ferro J, O'Brien J, Hennerici M, Scheltens P, Erkinjuntti T, Visser M, Langhorne P, Chabriat H, Waldemar G, Wallin A, Wahlund A (2011) 2001-2011: a decade of the LADIS (Leukoaraiosis And DISability) study: what have we learned about white matter changes and small-vessel disease? Cerebrovasc Dis 32(6):577–588
Pantoni L, Poggesi A, Inzitari D (2007) The relation between white-matter lesions and cognition. Curr Opin Neurol 20(4):390–397
Verdelho A, Madureira S, Ferro JM, Basile AM, Chabriat H, Erkinjuntti T, Fazekas F, Hennerici M, O’Brien J, Pantoni L, Salvadori E, Scheltens P, Visser MC, Wahlund LO, Waldemar G, Wallin A, Inzitariet D (2007) Differential impact of cerebral white matter changes, diabetes, hypertension and stroke on cognitive performance among non-disabled elderly. The LADIS study. J Neurol Neurosurg Psychiatry 78(12):1325–1330
Mechelli A, Price CJ, Friston KJ, Ashburner J (2005) Voxel-based morphometry of the human brain: methods and applications. Curr Med Imaging Rev 1(2):105–113
Matsuda H, Mizumura S, Nemoto K, Yamashita F, Imabayashi E, Sato N, Asadaet T (2012) Automatic voxel-based morphometry of structural MRI by SPM8 plus diffeomorphic anatomic registration through exponentiated lie algebra improves the diagnosis of probable Alzheimer Disease. Am J Neuroradiol 33(6):1109–1114
Hayakawa YK, Sasaki H, Takao H, Mori H, Hayashi N, Kunimatsu A, Aoki S, Ohtomo K (2013) Structural brain abnormalities in women with subclinical depression, as revealed by voxel-based morphometry and diffusion tensor imaging. J Affect Disord 144(3):263–268
Kok A (2001) On the utility of P3 amplitude as a measure of processing capacity. Psychophysiology 38:557–577
Himani A, Tandon OP, Bhatia MS (1999) A study of P300-event related evoked potential in the patients of major depression. Indian J Physiol Pharmacol 43(3):367–372
Frodl T, Meisenzahl EM, Muller D, Holder J, Juckel G, Möller HJ, Hegerl U (2002) P300 subcomponents and clinical symptoms in schizophrenia. Int J Psychophysiol 43(3):237–246
Polich J (2004) Clinical application of the P300 event-related brain potential. Phys Med Rehabil Clin N Am 15:133–161
Polich J, Corey-Bloom J (2005) Alzheimer's disease and P300: review and evaluation of task and modality. Curr Alzheimer Res 2(5):515–525
Turetsky BI, Dress EM, Braff DL, Calkins ME, Green MF, Greenwood TA, Gur RE, Gur RC, Lazzeroni LC, Nuechterlein KH, Radant AD, Seidman LJ, Siever LJ, Silverman JM, Sprock J, Stone WS, Sugar CA, Swerdlow NR, Tsuang DW, Tsuang MT, Light G (2015) The utility of P300 as a schizophrenia endophenotype and predictive biomarker: clinical and socio-demographic modulators in COGS-2. Schizophr Res 163(1-3):53–62
Ehlers MR, López Herrero C, Kastrup A, Hildebrandt H (2014) The P300 in middle cerebral artery strokes or hemorrhages: Outcome predictions and source localization. Clin Neurophysiol PMID: 25465358
Meador KJ, Loring DW, Davis HC, Patel BR, Adams RJ, Hammond EJ (1989) Cholinergic and serotoninergic effects on the P3 potential and recent memory. J Clin Exp Neuropsychol 11:252–260
Smith ME, Halgren E, Sokolik M, Baudena P, Musolino A, Liegeois-Chauvel C, Chauvel P (1990) The intracranial topography of the P3 event-related potential elicited during auditory oddball. Electroencephalogr Clin Neurophysiol 76(3):235–48
David E, Linden J (2005) The P300: where in the brain is it produced and what does it tell us? Neuroscientist 11:563–576
Li J, Zhang Z, Shang H (2012) A meta-analysis of voxel-based morphometry studies on unilateral refractory temporal lobe epilepsy. Epilepsy Res 98(2-3):97–103
Kopelman M, Lasserson D, Kingsley D, Bello F, Rush C, Stanhope N, Stevens T, Goodman G, Heilpern G, Kendall B, Colchester A (2001) Structural MRI volumetric analysis in patients with organic amnesia, 2: correlations with anterograde memory and executive tests in 40 patients. J Neurol Neurosurg Psychiatry 71(1):23–28
Duarte A, Ranganath C, Winward L, Hayward D, Knight RT (2004) Dissociable neural correlates for familiarity and recollection during the encoding and retrieval of pictures. Brain Res Cogn Brain Res 18(3):255–272
Yonelinas AP, Kroll NE, Quamme JR, Lazzara MM, Sauve MJ, Widaman KF, Knight RT (2002) Effects of extensive temporal lobe damage or mild hypoxia on recollection and familiarity. Nat Neurosci 5(11):1236–1241
Duarte A, Hayasaka S, Du A, Schuff N, Jahng G, Kramer J, Miller B, Weineret M (2006) Volumetric correlates of memory and executive function in normal elderly, mild cognitive impairment and Alzheimer's disease. Neurosci Lett 406(1-2):60–65
Chee MW, Chen KH, Zheng H, Chan KP, Isaac V, Sim SK, Chuah LY, Schuchinsky M, Fischl B, Ng TP (2009) Cognitive function and brain structure correlations in healthy elderly East Asians. Neuroimage 46(1):257–269
Phillips ML, Drevets WC, Rauch SL, Lane R (2003) Neurobiology of emotion perception I: the neural basis of normal emotion perception. Biol Psychiatry 54(5):504–514
Shiga T, Owada K, Hoshino T, Nagahara H, Shiratori K (2008) An analysis of risk factors for asymptomatic cerebral infarction. Intern Med 47(1):1–6
Romero JR, Beiser A, Seshadri S, Benjamin EJ, Polak JF, Vasan RS, DeCarli C, Wolf PA (2009) Carotid artery atherosclerosis, MRI indices of brain ischemia, aging, and cognitive impairment: the Framingham study. Stroke 40(5):1590–1596
van Oijen M, de Jong FJ, Witteman JC, Hofman A, Koudstaal PJ, Breteler MM (2007) Atherosclerosis and risk for dementia. Ann Neurol 61(5):403–410
Périco CA-M, Skaf CR, Yamada A, Duran F, Buchpiguel CA, Castro CC, Soares JC, Busatto GF (2005) Relationship between regional cerebral blood flow and separate symptom clusters of major depression: a single photon emission computed tomography study using statistical parametric mapping. Neurosci Lett 384(3):265–270
Acknowledgements
This work was supported by the Study Guide project of Hubei Provincial Health Department (2011 JX5C12).
Ethical Standards and Patient Consent
We declare that all human and animal studies have been approved by the First Affiliated Hospital of Yangtze University, China, and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We declare that all patients gave informed consent prior to inclusion in this study.
Conflict of Interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Wei Luo and Xun Jiang contributed equally to this work.
This article has been retracted by the authors due to the fact that it was published without permission and knowledge of the key authors of the study who published the same data in a different article (Yang T, Zhang L, Xiang M, Luo W, Huang J, Li M, Xiong X, Wang H. "Cognitive impairment and gray matter volume abnormalities in silent cerebral infarction" NeuroReport 2015 26(15):890-5.) The authors of both articles have reached an agreement to retract this article. The authors apologize to the Editors and readers as well as to all authors of the study.
About this article
Cite this article
Luo, W., Jiang, X., Wei, X. et al. RETRACTED ARTICLE: A study on cognitive impairment and gray matter volume abnormalities in silent cerebral infarction patients. Neuroradiology 57, 783–789 (2015). https://doi.org/10.1007/s00234-015-1535-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-015-1535-3