Abstract
Objective
To investigate the utilization of antiepileptic drugs (AEDs) in Dutch children and compare this with guidelines on the treatment of epilepsy in children.
Method
From the Dutch Interaction Database (IADB.nl) we selected children aged 0–19 years who received at least one prescription for an AED (ATC-code beginning with N03A) between 1997 and 2005. We calculated cumulative incidences and prevalences per 1000 children per year, stratified by age-category, sex and type of AED, and the total number of months each patient received at least one prescription of AEDs, and we counted the number of person-months and AEDs prescribed. For each AED, proportions of person-months were calculated for mono- and combination therapy. Kaplan-Meier survival analysis was used to analyse the duration of AED use, stratified by sex.
Results
The overall prevalence of the prescription of AEDs to children was constant at approximately 4.0 per 1000 children during the years of the study. The overall cumulative incidence from 1997–2005 was 0.67 per 1000 children. When stratified by age category or sex, there were no relevant differences in incidence or prevalence. Valproic acid was the most frequently prescribed drug, followed by carbamazepine and lamotrigine. In 20.3% of all person-months, patients received combination therapy; of these, 34.2% received combination therapy for 3 person-months or less. The older AEDs were prescribed more often as monotherapy than combination therapy, in contrast with the newer AEDs, for which the opposite was true. The 50% survival probability (= time period when 50% of children had stopped using AEDs) was around 2 years, with a significantly lower discontinuation of treatment for girls than boys (P = 0.04).
Conclusion
The overall cumulative incidence of AEDs was determined to be 0.67 per 1000 children, and the prevalence 4.0 per 1000 children. The most frequently prescribed drug is valproic acid, followed by carbamazepine and lamotrigine. The prescribing of AEDs seems to be in conformity with the overall recommendations of the guidelines on the treatment of epilepsy in children.
Similar content being viewed by others
Introduction
Epilepsy is one of the most common neurological disorders. Its incidence is highest during childhood, with a median incidence for children aged 0–14 years of 0.822 per 1000 children [1]. It has also been estimated that 70% of all epilepsy syndromes start between the age of 0 and 19 years [2]. The vast majority of children with epilepsy receive given antiepileptic drug (AED) therapy—for example, in Sweden, 81.0% of epileptic children are treated with AEDs [3]. However, even though many children suffer from epilepsy and are treated AEDs, very little is known about the prescribing and use of AEDs in children, especially on the prescribing of AEDs in combination therapy and the duration of use.
Many AEDs have been developed and registered over the years. Table 1 shows the AEDs available on the Dutch market in 2005 with their approval date and Anatomical Therapeutic Chemical classification (ATC) code. The approval given to these AEDs on these dates was not subject to age restrictions that might be given in the Supplementary Protection Certificates (SPCs), so some of the AEDs may not even be licensed for children.
The Food and Drug Administration (FDA) approved a number of AEDs for the treatment of indications other than epilepsy, such as bipolar disorder, migraine prophylaxis, mania and neuralgia.
Several guidelines for the treatment of epilepsy in children have been published [2, 4], including a Dutch guideline for (pediatric)neurologists and paediatricians in the Netherlands [5]. These guidelines recommend that the diagnosis of ‘epilepsy’ be limited to a specific seizure type and/or epileptic syndrome in order to achieve the best available treatment. The treatment recommendations in the guideline are ordered by type of seizure of the epileptic syndrome; however, the overall conclusion that can be drawn from the guideline is that valproic acid is the most effective drug—and therefore the drug of choice—for most types of epilepsy, followed by carbamazepine and lamotrigine.
We describe here the prescribing patterns of AEDs in Dutch children with an emphasis on type of AED, mono- versus combination therapy of AEDs and the duration of use, and we compare these with guidelines on the treatment of epilepsy in children.
Methods
Setting and study population
This study was performed with data extracted from the InterAction database (www.IADB.nl), a prescription database containing pharmacy dispensing data from pharmacies located in the northern and eastern parts of the Netherlands [6]. In the period from 1997 to 2005, the IADB.nl covered a population of approximately 500,000 people. The size of this population increased during the study period, but there were no changes in age and gender distribution [7]. The IADB includes all prescriptions, regardless of prescriber, insurance or reimbursement status, with the exception of over-the-counter (OTC) drugs and drugs dispensed during hospitalization. In the Netherlands, people commonly register with one pharmacy and obtain all of their medication from that pharmacy; therefore, a complete medication history of an individual is available in the pharmacy dispensing records.
Children aged 0 to 19 years who from 1997 to 2005 received at least one prescription for an AED (ATC-code N03A*; Table 1) were selected from the IADB.nl.
All AEDs available in the Netherlands were studied, regardless of age restrictions and restrictions in approved indication, because off-label prescription to children of AEDs is possible. Diazepam and a number of other benzodiazepines were not included in the study since they are prescribed mainly for the treatment of prolonged seizures and not for chronic treatment.
Although one might presume that these drugs are most commonly prescribed to children for the treatment of epilepsy, the specific disorder for which the AED was prescribed was not known. Therefore, we classified the probable indication based on the other prescriptions that the child received. When a child received more than one prescription for an antidepressant (ATC-code N06A*) or an antipsychotic drug (ATC-code N05A*), we presumed that the AEDs were prescribed for a bipolar disorder. When propranolol or antimigraine drugs (ATC-code N02C*) were prescribed, we presumed the indication was migraine. When there were fewer than two prescriptions for one of the above mentioned drugs, we presumed epilepsy to be the indication for which the AED was prescribed.
Data analyses
Annual prevalences and cumulative incidences were calculated for all children and stratified by sex and age categories of 5 years. Because of low cumulative incidences, the latter are nearly equal to incidence density figures (number per total person-years). Prevalences and cumulative incidences were also stratified by individual AED (based on ATC-code). We also stratified by type of AED, based on approval date in the USA [8] (newly approved AEDs are indicated in Table 1).
The cumulative incidences of AED prescriptions were defined as the number of children who received initial treatment with an AED, also divided by the total number of children in the population and multiplied by 1000. Initial treatment was defined as the first AED prescription in the IADB.nl, when the child was in the database at least 6 months before the first AED was prescribed or aged 2 years or younger when the first AED was prescribed. When a child received two different AEDs at the date of the initial treatment, both were counted. A child could only once be a first time user, so when at a later time during treatment the prescribed AED was changed or the treatment was resumed after an AED-free period, the child was not counted again as an incident user.
Annual prevalences were calculated by counting the number of children receiving one or more prescriptions for an AED, divided by the total number of children in the population per age and sex group in the respective years, and then multiplied by 1000.
For both the prevalences and cumulative incidences, 95% confidence intervals (CIs) were calculated using the ‘Score with CC’-method, because this method is very suitable for small proportions [9]. When there were no significant differences (P > 0.05) but a trend was suspected, a Chi-square test for trend was conducted.
To investigate combination therapy, the prescriptions of every child were divided into person-months. This was done because pharmacotherapy can change several times during 1 year, therefore counting only children or person-years would result in a loss of data, while counting person-months would preserve more of this information. For each patient we calculated the total number of months they received at least one prescription of AEDs and counted the number of person-months. For every person-month, the number of AEDs and the ATC-codes of the prescribed AEDs were registered.
For every AED, we counted the number of person-months in which the AED was given as monotherapy and the number of person-months in which the AED was given together with at least one other AED (combination therapy). Using these values, we calculated the percentages of the total number of person-months of all children per AED.
For the analyses of combination therapy, we removed clonazepam from the data because this AED is usually not prescribed for chronic treatment, but mainly for the acute treatment of prolonged seizures. Since it was not known when and how much clonazepam was used, person-months could not be calculated.
Kaplan-Meier analyses were conducted to examine the duration of use of all children and stratified by sex. The start of the therapy was defined the same as for the cumulative incidence rates. To determine the day on which the therapy was ended, we calculated the number of days of use for every prescription from the daily dosage and the prescribed number of units (tablets, etc.). When there were 90 days or more between the last day of use of one prescription and a subsequent prescription (if any), the therapy was defined to be ended. The number of days between the initial treatment and the last day of use before the end was counted. When a child was in the database 90 days or less after the end of the treatment, the case was defined as censored.
The results revealed that the data had a log-normal distribution and many censored cases (50%). Consequently, mean, median and CIs could not be calculated. To compare two curves, we used the ‘Generalized accelerated failure time’-model to determine significant (P < 0.05) differences.
This statistical analysis was conducted using R ver. 2.5.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Overall utilization
From the IADB.nl we identified 1527 children [791 boys (51.8%) and 736 girls (48.2%)] aged 0–19 years who had received at least one AED prescription between 1997 and 2005, with a total of 182,201 prescriptions during this period. All AEDs mentioned in Table 1 were prescribed, except for zonisamide.
We estimated that AEDs had been prescribed for epilepsy in at least 80% of these children, while approximately 10% had received the drugs for mood disorders and 5% for migraine.
The total number of children in the population covered by the IADB.nl is shown in Table 2, together with the yearly cumulative incidences. Throughout the study period, the cumulative incidence remained constant at around 0.67 per 1000 children. There were no significant differences between boys and girls or between age categories (data not shown).
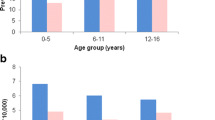
Figure 1 shows the overall prevalence, which was constant around 4.0 per 1000 children throughout the study period. There were no significant differences both sexes or between age-categories (data not shown).
Figure 1 also shows the prevalence’s of the old and new AEDs separately. This graph reveals that the prevalence of the new AEDs was significantly lower than that of the old AEDs, that between 1997 and 2003 there was no significant difference between the prevalence of old AEDs and the overall prevalence and that for both groups of AEDs there were no significant differences between successive years and no significant trends.
Utilization of individual AEDs
As shown in Fig. 2, the AED with the highest prevalence was valproic acid, followed by carbamazepine and lamotrigine. The prevalence of valproic acid was constant during all years, while that of carbamazepine and lamotrigine increased slightly; however, there were no significant differences between successive years, and both trends were not significant. In 1997, vigabatrin had a higher prevalence than lamotrigine, but during the following years the prevalence dropped rapidly, although there were no significant differences between successive years and no significant trend. The prevalence of phenobarbital was constant at around 0.25 per 1000 children. Levetiracetam was introduced on the Dutch market in 2000, and its prevalence increased (although not significant) from 0.04 in 2001 to 0.29 in 2005. For other AEDs, prevalences were around 0.2 per 1000 children or lower (data not shown).
Combination therapy
The study population accounted for a total of 19,829 person-months of AED use during the whole study period (1997–2005). In 20.3% of these months, two or more AEDs were prescribed, with a maximum of four AEDs per person-month. A total of 239 children (22.8%) were prescribed two or more AEDs for at least one person-month, and only nine children (0.9%) were prescribed four AEDs in 1 month.
The number of person-months in which two or more AEDs were prescribed varied from 1 to 103, with 34.2% of the children receiving combination therapy for 3 person-months or less.
In 2000–2005, which is the period that most of the new AEDs were on the Dutch market, therapy with an AED was given in 15,303 person-months. Figure 3 shows the percentages of person-months for every AED in which it was given as monotherapy or in combination therapy. Valproic acid was the most highly prescribed AED in both mono- and combination therapy (51.5 and 14.7%, respectively), followed by carbamazepine in monotherapy (15.2%) and lamotrigine in combination therapy (12.4% of all person-months vs. 7.7% for carbamazepine). Valproic acid, carbamazepine and the other old AEDs (phenobarbital, phenytoin and ethosuximide) were prescribed more often in monotherapy than in combination therapy, while the opposite was true for lamotrigine and other new AEDs (higher prescribing percentages in combination than in monotherapy.
Duration of use
A total of 812 patients could be used for survival analyses (analyses of the duration of AED use). Their overall discontinuation of treatment is shown in Fig. 4a, and has a median of approximately 2 years. Figure 4b shows the discontinuation of treatment for boys (48.6%) and girls (51.4%) separately; the discontinuation of treatment of girls is significantly shorter.
Discussion
Principal findings
During the period 1997–2005 the average year-prevalence of AED-prescribing in Dutch children aged 0–19 years was 4.0 per 1000 children and the cumulative incidence was 0.67 per 1000 children per year. Valproic acid was the most frequently prescribed AED, followed by carbamazepine and lamotrigine. The older AEDs were prescribed more as monotherapy than combination therapy, while the opposite was true for the new AEDs. The prescribing of AEDs during the study period seems to be in conformity with the overall recommendations of the guidelines on treatment of epilepsy in children.
The 50% survival probability of children to whom AEDs are prescribed is around 2 years. This is in conformity with the guidelines, for they advise that treatment with an AED should be discontinued when a child is seizure-free for 1–2 years.
Strengths and weaknesses
This study was conducted with data from the IADB.nl, which is a prescription dispensing database; consequently, there was no information on the actual use of the dispensed medicines by the children. Therefore, one limitation to our study is that we investigated dispensing rather than actual drug use, with the assumption that dispensing data are representative for the AEDs used in the population. The strength of this database is that it contains data on a large portion of the Dutch population for a large period of time and that dispensing records are gathered prospectively.
Another limitation is that the actual indication for which AEDs were prescribed was not actually known, but estimated from other drugs prescribed. As such, the estimation is only a rough one, but there was no other possibility to classify the indication.
The IADB.nl does not contain information on the use of OTC medication, but this did not represent a limitation for this study, since in the Netherlands none of the AEDs is available as an OTC. The IADB.nl also does not contain prescriptions prescribed during hospitalization, but as the majority of children are prescribed AEDs in an outpatient setting, this will have no effect on the results.
The number of patients included in this study was small due to the low cumulative incidence and prevalence. This limited our results since survival analyses could not be performed for most individual AEDs because there were 30 patients 30 or less, and a Kaplan–Meier analysis is not reliable [10].
The use of person-months to investigate combination therapy is a helpful approach to obtain insight in how often combination therapy is prescribed. The drawback of counting person-months is that when a patient is prescribed two AEDs consecutively, the number of AEDs counted in 1 person-month is two, even though these AEDs are not used together. Thus, the percentages of combination therapy are probably overestimated in this study. On the other hand, the guidelines recommend that, when there is a change in AEDs, the patient should not switch medication abruptly from one day to the other but that both AEDs should be used during a transition period [2, 4, 5]. To prevent this overestimation, one could use person-days, but this approach generates too much data to handle. Therefore, we accept the possibility of an overestimation due to the use of person-months, but assume that it is small and that person-months are a useful tool in analysing combination therapy.
Results compared to those reported in the literature
The prevalences and cumulative incidences that we found correspond with prevalences and incidences reported on the prescription of AEDs to children in Denmark, where the average prevalence for children 5–14 years old is approximately 5 per 1000 person-years [11], and the average incidence for children <16 years is around 0.5 per 1000 person-years [12]. In the USA, the prevalence was reported to be 17.5 per 1000 children in 2000 [13]. This prevalence is remarkably higher than those in the Netherlands and Denmark, probably because in the USA AEDs are prescribed for psychiatric disorders (68%) more often than in the Netherlands (10% [13]) and Denmark (18% [12]).
A recent study using data on children aged 0–18 years from 1997–2005 that were extracted from the General Practice Research Database (GPRD) of the UK reported a prevalence of AEDs of approximately 8 per 1000 person-years and an average incidence of around 0.7 per 1000 person-years [14]. Thus, the incidence in the UK is similar to that reported for the Netherlands, but the prevalence is higher, suggesting that in the UK, children are treated for a longer period of time than in Denmark and the Netherlands. Our study shows that in the Netherlands, children who receive their first AED prescription have a 50% survival probability of around 2 years. To our knowledge, this survival probability is not known for children in the UK who receive one or more AED prescriptions; therefore, this hypothesis can not be tested.
The prevalences of individual AEDs, as shown in Fig. 2, are in conformity with the guidelines that recommend valproic acid as the AED of first choice for most seizure types and epileptic syndromes [2, 4, 5]. Carbamazepine is also effective in many seizure types, but may worsen others and is therefore, not advised as often as valproic acid [2, 4, 5]. Consequently, it has the second highest prevalence. Lamotrigine also has a broad antiepileptic spectrum and fewer side-effects than valproic acid and carbamazepine. However, because physicians have less experience with the prescribing of these newer AEDs and because they are more expensive [15], lamotrigine is advised as a second-choice AED [4, 5]; therefore, it has a lower prevalence than valproic acid and carbamazepine. Of the new AEDs, lamotrigine is prescribed the most, which is in agreement with data from the UK [14] and Denmark [11]. The decrease in the prevalence of vigabatrin after 1997, also mentioned by Ackers et al., is probably the result of reports that vigabatrin can cause irreversible visual field defects [16]. The prevalence of phenobarbital is constant and higher than that of the new AEDs, even though this old AED has more severe side-effects than the new AEDs [17]. It has also been prescribed mostly to the youngest age category (0–4 years), likely due to its unique position of being recommended for the treatment of neonatal seizures [5, 18]. Levetiracetam has on the Dutch market for a few years, and the increase in prevalence indicates its popularity among prescribers.
In our study, valproic acid and carbamazepine were the most prescribed AEDs in monotherapy in terms of person-months. This result is in conformity with the overall recommendations of the guidelines that recommend that monotherapy is the preferred treatment over combination therapy, with valproic acid and carbamazepine being the AEDs of first choice in monotherapy [2, 4, 5]. Also in agreement with the guidelines—that new AEDs should be used as add-on therapy to an old AED—are the higher percentages we found for the new AEDs in combination therapy than in monotherapy[2, 4, 5]. The percentages of pregabalin in our study were close to zero due to its approval only in July 2004; consequently, it was on the Dutch market for only 5 months of the total study period of 108 months.
Our finding that 70% of the children discontinue treatment after 9 years is in agreement with the finding that 64% of the Dutch children discontinue treatment after 5 years [19]. The reason for the significant difference in the discontinuation of treatment of boys and girls is not known, but it may possibly be due to a re-consideration in the prescribing of an AED when girls start using contraceptives since many AEDs interact with oral contraceptives [20].
Conclusion
The results of this study show that valproic acid, carbamazepine and lamotrigine were the mostly frequently prescribed AEDs for children aged 0–19 years between 1997 and 2005. This prevalence is in conformity with the overall recommendations of the guidelines for the treatment of epilepsy in children. After 2 years, the medication was stopped in 50% of all children who were prescribed an AED. In combination therapy older AEDs are combined with newer AEDs, but valproic acid is still the most highly prescribed AED.
To our knowledge, this is the first study that has analysed the prescribing AEDs in combination therapy and the duration of their use.
Abbreviations
- AEDs:
-
Antiepileptic drugs
- ATC:
-
Anatomical Therapeutic Chemical classification
- FDA:
-
Food and Drug Administration
- GPRD:
-
General practice research database
- IADB.nl:
-
InterAction database of the Netherlands
- SPC:
-
Supplementary protection certificate
References
Kotsopoulos IAW, van Merode T, Kessels FGH, de Krom MCTFM, Knottnerus JA (2002) Systematic review and meta-analysis of incidence studies of epilepsy and unprovoked seizures. Epilepsia 43:1402–1409
Scottish Intercollegiate Guidelines Network (SIGN) (2005) Diagnosis and management of epilepsy in children and young people—a national clinical guideline. Available at www.sign.ac.uk/pdf/sign81.pdf. Accessed 30 July 2007
Larsson K, Eeg-Olofsson O (2006) A population based study of epilepsy in children from a Swedish county. Eur J Peadiatr Neurol 10:107–113
National Institute for Clinical Excellence (NICE) (2004). Diagnosis and management of epilepsy in children and young people. Available at http://www.nice.org.uk/CG020NICEguideline. Accessed 30 July 2007
Nederlandse Vereniging voor Neurologie (NVN) (2006) Epilepsy—guidelines for diagnose and treatment (in Dutch). Available at http://www.neurologie.nl/uploads/136/454/richtlijn_epilepsie_definief_2.pdf. Accessed 30 July 2007
Schirm E, Monster TBM, de Vries R, van den Berg PB, de Jong-van den Berg LTW, Tobi H (2004) How to estimate the population that is covered by community pharmacies? An evaluation of two methods using drug utilisation information. Pharmacoepidemiol Drug Saf 13:173–179
Lub R, Denig P, van den Berg PB, Hoogenberg K, de Jong-van den Berg LTW (2006) The impact of new insights and revised practice guidelines on prescribing drugs in the treatment of type 2 diabetes mellitus. Br J Clin Pharmacol 62:660–665
French JA, Kanner AM et al (2004) Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new onset epilepsy. Neurology 62:1252–1260
Tobi H, van den Berg PB, de Jong-van den Berg LTW (2005) Small proportions: what to report for confidence intervals? Pharmacoepidemiology and drug safety 14:239–247
Statsoft—Electronic Statistics Textbook. Available at http://www.statsoft.com/textbook/stsurvan.html#lrequired. Accessed 27 July 2007
Rochat P, Hallas J, Gaist D, Friis ML (2001) Antiepileptic drug utilization: a Danish prescription database analysis. Acta Neurol Scand 104:6–11
Tsiropoulos I, Gichangi A, Andersen M, Bjerrum L, Gaist D, Hallas J (2006) Trends in utilization of antiepileptic drugs in Denmark. Acta Neurol Scand 113:405–411
Zito JM, Safer DJ, Gardner JF, Soeken K, Ryu J (2006) Anticonvulsant treatment fir psychiatric and seizure indications among youths. Psychiatr Serv 57:681–685
Ackers R, Murray ML, Besag FM, Wong ICK (2007) Prioritizing children’s medicines for research: a pharmacoepidemiological study of antiepileptic drugs. Br J Clin Pharmacol 63:687–697
College voor Zorgverzekeringen (2007) Farmacotherapeutic Compass (in Dutch). Available at http://www.fk.cvz.nl. Accessed 24 July 2007
Eke T, Talbot JF, Lawden MC (1997) Severe persistent visual field constriction associated with vigabatrin. Br Med J 314:180–181
Heller AJ, Chesterman P, Elwes RD, Crawford P, Chadwick D, Johnson AL, Reynolds EH (1995) Phenobarbitone, phenytoin, carbamazepine, or sodium valproate for newly diagnosed adult epilepsy: a randomised comparative monotherapy trail. J Neurol Neurosurg Psychiatry 58:44–50
Painter MJ, Scher MS, Stein AD, Armatti S, Wang Z, Gardiner JC, Paneth N, Minnigh B, Alvin J (1997) Phenobarbital compared with Phenytoin for the treatment of Neonatal Seizures. N Eng J Med 341:485–489
Arts WFM, Brouwer OF, Peters ACB, Stroink H, Peeters EAJ, Schmitz PIM, van Donselaar CA, Geerts, AT (2004) Course and prognosis of childhood epilepsy: 5-year follow-up of the Dutch study of epilepsy in childhood. Brain 127:1774–1784
Zupanc ML (2006) Antiepileptic drugs and hormonal contraceptives in adolescent women with epilepsy. Neurology 66(S3):S37–S45
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
van de Vrie-Hoekstra, N.W., de Vries, T.W., van den Berg, P.B. et al. Antiepileptic drug utilization in children from 1997–2005—a study from the Netherlands. Eur J Clin Pharmacol 64, 1013–1020 (2008). https://doi.org/10.1007/s00228-008-0480-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-008-0480-z