Abstract
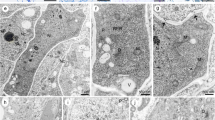
The ultrastructure of post-fertilization development of the red alga Callophyllis linearis (Kyl.) Abb. & Norr. of the Kallymeniaceae showed that the Kallymeniaceae are more closely related to the Cryptomeniales than to the Gigartinales. The auxiliary cell produces multinucleate gonimoblast initials which divide to produce generative gonimoblast cells. These cells undergo repeated cleavages to form clusters of carpospore initials, which finally differentiate into carpospores. Carpospore differentiation is simple and occurs in three developmental stages. In the first stage, young carpospores are avacuolate, contain a large nucleus, have few undeveloped plastids and exhibit incipient dictyosome activity. During the intermediate stage, plastids develop their internal thylakoid system, starch granules are formed and dictyosomes continue to produce vesicles that contribute carpospore wall material. The formation of fibrous vacuoles originates from the fibrous vacuole associated organelles (FVAOs). In the final stage, mature carpospores form cored vesicles. They also contain well-developed plastids, fibrous vacuoles, numerous starch granules and are surrounded by a two-layered carpospore wall.






Similar content being viewed by others
References
Abbott IA, Hollenberg GI (1976) Marine algae of California. Stanford University Press, Stanford
Bold HW, Wynne MJ (1985) Introduction to the algae—structure and reproduction, 2nd edn. Prentice-Hall, Englewood Cliffs, N.J.
Broadwater S, Scott J (1982) Ultrastructure of early development in the female reproductive system of Polysiphonia harveyi Bailey (Ceramiales, Rhodophyta). J Phycol 18:427–441
Broadwater S, Scott J (1983) Fibrous vacuole associated organelles (FVAOs) in the Florideophyceae: a new interpretation of the “appareil cinétique”. Phycologia 22:225–233
Chamberlain AH, Evans LV (1973) Aspects of spore production in the red alga Ceramium. Protoplasma 76:139–159
Delivopoulos SG (2000) Ultrastructure of spermatiogenesis in the red alga Osmundea spectabilis var. spectabilis (Rhodomelaceae, Ceramiales, Rhodophyta). Phycologia 39:517–526
Delivopoulos SG (2002) Ultrastructure of tetrasporogenesis in the red alga Osmundea spectabilis var. spectabilis (Rhodomelaceae: Ceramiales: Rhodophyta). Mar Biol 140:921–934
Delivopoulos SG (2003) Ultrastructure of post-fertilization development in the red alga Nienburgia andersoniana (Delesseriaceae, Ceramiales, Rhodophyta). Bot J Linn Soc 142:289–299
Delivopoulos S, Diannelidis BE (1991a) Ultrastructure of carposporophyte development in the red alga Ceramium strictum (Rhodophyta, Ceramiales). Microbios 65:71–80
Delivopoulos S, Diannelidis BE (1991b) Ultrastructure of carpospore differentiation in the red alga Ceramium strictum Greville and Harvey (Ceramiales, Ceramiaceae). Ann Sci Nat Bot 13(II):9–21
Delivopoulos S, Diannelidis BE (1991c) Ultrastructure of carpospore differentiation in the red alga Alsidium corallinum C. Agardh. (Ceramiales, Rhodomelaceae). Ann Sci Nat Bot 13(II):23–31
Delivopoulos S, Kugrens P (1984) Ultrastructure of carposporogenesis in the parasitic red alga Faucheocolax attenuata Setch. (Rhodymeniales, Rhodymeniaceae). Am J Bot 71:1245–1259
Delivopoulos S, Kugrens P (1985) Ultrastructure of carposporophyte development in the red alga Gloiosiphonia verticillaris (Cryptonemiales, Gloiosiphoniaceae). Am J Bot 72:1926–1938
Delivopoulos SG, Polne-Fuller M, Diannelidis BE (1999) Ultrastructural study of the differentiated blade of Porphyra perforata (Rhodophyta). Nova Hedwigia 68:65–74
Gori P (1980) Ultrastructure of carpospores in Gastroclonium clavatum Ardissone (Rhodymeniales). Protoplasma 103:263–271
Hommersand MH, Ott DW (1970) Development of the carposporophyte of Kallymenia reniformis (Turner) J. Agardh. J Phycol 6:322–331
Konrad-Hawkins E (1974) Golgi vesicles of uncommon morphology and wall formation in the red alga, Polysiphonia. Protoplasma 80:1–14
Kugrens P (1974) Light and electron microscope studies of the development and liberation of Janczewskia gardneri Setch spermatia (Rhodophyta). Phycologia 13:295–306
Kugrens P (1980) Electron microscopic observations on the differentiation and release of spermatia in the marine red alga Polysiphonia hendryi (Ceramiales, Rhodomelaceae). Am J Bot 67:519–528
Kugrens P, Delivopoulos SG (1986) Ultrastructure of the carposporophyte and carposporogenesis in the parasitic red alga Plocamiocolax pulvinata Setch. (Gigartinales, Plocamiaceae). J Phycol 22:8-21
Kugrens P, West JA (1973) The ultrastructure of carpospore differentiation in the parasitic red alga Levringiella gardneri (Setch) Kylin. Phycologia 12:163–173
Kugrens P, West JA (1974) The ultrastructure of carposporogenesis in the marine hemiparasitic red alga Erythrocystis saccata. J Phycol 10:139–147
Kylin H (1928) Entwicklungsgeschichtliche Florideen-studien. Acta Univ Lund 24(4):1–127
Kylin H (1930) Über die Entwicklungsgeschichte der Florideen. Acta Univ Lund 26(6):1–104
Kylin H (1932) Die Florideenordnung Gigartinales. Acta Univ Lund 28(8):1–88
Kylin H (1956) Die Gattungen der Rhodophyceen. CWK Gleerups, Lund
Pueschel CM (1992) An ultrastructural survey of the diversity of crystalline proteinaceous inclusions in red algal cells. Phycologia 31:489–499
Ramm-Anderson SM, Wetherbee R (1982) Structure and development of the carposporophyte of Nemalion helminthoides (Nemalionales, Rhodophyta). J Phycol 1:133–141
Triemer RE, Vasconcelos AC (1977) The ultrastructure of carposporogenesis in Caloglossa leprieurii (Delesseriaceae, Ceramiales). Am J Bot 64:825–834
Tripodi G (1971) The fine structure of the cystocarp in the red alga Polysiphonia sertularioides (Grat.) J. Ag. J Submicr Cytol 3:71–79
Tripodi G (1974) Ultrastructural changes during carpospore formation in the red alga Polysiphonia. J Submicr Cytol 6:275–286
Tripodi G, Gargiulo GM (1984) Relationships among membranes in plastids of the red alga Nitophyllum punctatum (Stackh) Grev. Protoplasma 119:55–61
Tsekos I (1982) Plastid development and floridean starch grain formation during carposporogenesis in the red alga Gigartina teedii. Cryptog Algol 3:91–103
Tsekos I, Schnepf E (1983) The ultrastructure of carposporogenesis in Gigartina teedii (Roth) Lamour. (Gigartinales, Rhodophyceae): auxiliary cell, cystocarpic plant. Flora 173:81–96
Wetherbee R (1978) Differentiation and continuity of the Golgi apparatus during carposporogenesis in Polysiphonia (Rhodophyta). Protoplasma 95:347–360
Wetherbee R (1980) Postfertilization development in the red alga Polysiphonia 1. Proliferation of the carposporophyte. J Ultrastruct Res 70:259–274
Wetherbee R, West JA (1977) Golgi apparatus of unique morphology during early carposporogenesis in a red alga. J Ultrastruct Res 58:119–133
Wetherbee R, Wynne MJ (1973) The fine structure of the nucleus and nuclear association of developing carposporangia in Polysiphonia novae-angliae (Rhodophyta). J Phycol 9:402–407
Wetherbee R, Janda DM, Bretherton GA (1984) The structure, composition and distribution of proteinaceous crystalloids in vegetative cells of the red alga Wrangelia plumosa. Protoplasma 119:135–140
Acknowledgements
I gratefully acknowledge Dr. Ursula Luetz-Meindl for her critical review and constructive suggestions for improving the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by O. Kinne, Oldendorf/Luhe
Rights and permissions
About this article
Cite this article
Delivopoulos, S.G. Ultrastructure of post-fertilization development in the red alga Callophyllis linearis (Kallymeniaceae, Cryptonemiales, Rhodophyta). Marine Biology 144, 665–674 (2004). https://doi.org/10.1007/s00227-003-1235-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-003-1235-0