Abstract
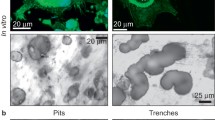
Advanced glycation end products (AGEs) accumulate in bone extracellular matrix as people age. Previous studies have shown controversial results regarding the role of in situ AGEs accumulation in osteoclastic resorption. To address this issue, this study cultured human osteoclast cells directly on human cadaveric bone slices from different age groups (young and elderly) to warrant its relevance to in vivo conditions. The cell culture was terminated on the 3rd, 7th, and 10th day, respectively, to assess temporal changes in the number of differentiated osteoclasts, the number and size of osteoclastic resorption pits, the amount of bone resorbed, as well as the amount of matrix AGEs released in the medium by resorption. In addition, the in situ concentration of matrix AGEs at each resorption pit was also estimated based on its AGEs autofluorescent intensity. The results indicated that (1) osteoclastic resorption activities were significantly correlated with the donor age, showing larger but shallower resorption pits on the elderly bone substrates than on the younger ones; (2) osteoclast resorption activities were not significantly dependent on the in situ AGEs concentration in bone matrix, and (3) a correlation was observed between osteoclast activities and the concentration of AGEs released by the resorption. These results suggest that osteoclasts tend to migrate away from initial anchoring sites on elderly bone substrate during resorption compared to younger bone substrates. However, such behavior is not directly related to the in situ concentration of AGEs in bone matrix at the resorption sites.






Similar content being viewed by others
References
Ji JD, Woo JH, Choi SJ et al (2009) Advanced glycation end-products (ages): a novel therapeutic target for osteoporosis in patients with rheumatoid arthritis. Med Hypotheses 2:201–202
Valcourt U, Merle B, Gineyts E et al (2007) Non-enzymatic glycation of bone collagen modifies osteoclastic activity and differentiation. J Biol Chem 8:5691–5703
Wautier JL, Paton RC, Wautier MP et al (1981) Increased adhesion of erythrocytes to endothelial cells in diabetes mellitus and its relation to vascular complications. N Engl J Med 5:237
Stevens VJ, Rouzer CA, Monnier VM et al (1978) Diabetic cataract formation: potential role of glycosylation of lens crystallins. Proc Natl Acad Sci USA 6:2918–2922
McCann VJ, Davis RE (1979) Glycosylated hemoglobin concentrations in patients with diabetic neuropathy. Acta Diabetol Lat 3:205–209
Viberti GC, Pickup JC, Jarrett RJ et al (1979) Effect of control of blood glucose on urinary excretion of albumin and β2 microglobulin in insulin-dependent diabetes. N Engl J Med 12:638–641
Takagi M, Kasayama S, Yamamoto T et al (1997) Advanced glycation endproducts stimulate interleukin-6 production by human bone-derived cells. J Bone Miner Res 3:439–446
Dyer DG, Blackledge JA, Thorpe SR et al (1991) Formation of pentosidine during nonenzymatic browning of proteins by glucose. Identification of glucose and other carbohydrates as possible precursors of pentosidine in vivo. J Biol Chem 18:11654–11660
Tang SY, Vashishth D (2010) Non-enzymatic glycation alters microdamage formation in human cancellous bone. Bone 1:148–154
Karim L, Tang SY, Sroga GE et al (2013) Differences in non-enzymatic glycation and collagen cross-links between human cortical and cancellous bone. Osteoporos Int 9:2441–2447
McCarthy AD, Etcheverry SB, Bruzzone L et al (1997) Effects of advanced glycation end-products on the proliferation and differentiation of osteoblast-like cells. Mol Cell Biochem 1–2:43–51
McCarthy AD, Etcheverry SB, Bruzzone L et al (2001) Non-enzymatic glycosylation of a type i collagen matrix: Effects on osteoblastic development and oxidative stress. BMC cell biology 2:16
Mercer N, Ahmed H, Etcheverry SB et al (2007) Regulation of advanced glycation end product (age) receptors and apoptosis by ages in osteoblast-like cells. Mol Cell Biochem 1–2:87–94
Alikhani Z, Alikhani M, Boyd C et al (2007) Advanced glycation end products stimulate osteoblast apoptosis via the map kinase and cytosolic apoptotic pathways. Bone 2:345–353
Zhu XF, Wang TC, Zhang RH (2012) Effects of yigu capsule containing serum on the osteoblast differentiation and the expressions of osteoprotegerin and bone morphogenetic protein 2 after treatment by advanced glycation end products in vitro. Zhongguo Zhong Xi Yi Jie He Za Zhi 4:525–529
Franke S, Ruster C, Pester J et al (2011) Advanced glycation end products affect growth and function of osteoblasts. Clin Exp Rheumatol 4:650–660
Hein G, Weiss C, Lehmann G et al (2006) Advanced glycation end product modification of bone proteins and bone remodelling: hypothesis and preliminary immunohistochemical findings. Ann Rheum Dis 1:101–104
Miyata T, Notoya K, Yoshida K et al (1997) Advanced glycation end products enhance osteoclast-induced bone resorption in cultured mouse unfractionated bone cells and in rats implanted subcutaneously with devitalized bone particles. J Am Soc Nephrol 2:260–270
Yamagishi S, Nakamura K, Inoue H (2005) Possible participation of advanced glycation end products in the pathogenesis of osteoporosis in diabetic patients. Med Hypotheses 6:1013–1015
Dong XN, Qin A, Xu J et al (2011) In situ accumulation of advanced glycation end products (ages) in bone matrix and its correlation with osteoclastic bone resorption. Bone 2:174–183
Luevano-Contreras C, Chapman-Novakofski K (2010) Dietary advanced glycation end products and aging. Nutrients 12:1247–1265
Hein G, Wiegand R, Lehmann G et al (2003) Advanced glycation end-products pentosidine and n epsilon-carboxymethyllysine are elevated in serum of patients with osteoporosis. Rheumatology 10:1242–1246
Rumpler M, Würger T, Roschger P et al (2012) Microcracks and osteoclast resorption activity in vitro. Calcif Tissue Int 3:230–238
Chang P-C, Chung M-C, Wang Y-P et al (2012) Patterns of diabetic periodontal wound repair: a study using micro-computed tomography and immunohistochemistry. J Periodontol 5:644–652
Hein GE (2006) Glycation endproducts in osteoporosis—is there a pathophysiologic importance? Clin Chim Acta 1–2:32–36
Schwartz AV, Garnero P, Hillier TA et al (2009) Pentosidine and increased fracture risk in older adults with type 2 diabetes. J Clin Endocrinol Metab 7:2380–2386
Zhou Z, Immel D, Xi CX et al (2006) Regulation of osteoclast function and bone mass by rage. J Exp Med 4:1067–1080
Charoonpatrapong K, Shah R, Robling AG et al (2006) Hmgb1 expression and release by bone cells. J Cell Physiol 2:480–490
Zhou Z, Han JY, Xi CX et al (2008) Hmgb1 regulates rankl-induced osteoclastogenesis in a manner dependent on rage. J Bone Miner Res 7:1084–1096
Burr DB (1993) Remodeling and the repair of fatigue damage. Calcif Tissue Int 53:S75–S80 discussion S80-1
Schaffler MB, Choi K, Milgrom C (1995) Aging and matrix microdamage accumulation in human compact bone. Bone 6:521–525
Barrere F, van Blitterswijk CA, de Groot K (2006) Bone regeneration: molecular and cellular interactions with calcium phosphate ceramics. Int J Nanomed 3:317–332
Hofmann B, Adam A-C, Jacobs K et al (2013) Advanced glycation end product associated skin autofluorescence: a mirror of vascular function? Exp Gerontol 1:38
Vashishth D, Gibson GJ, Khoury JI et al (2001) Influence of nonenzymatic glycation on biomechanical properties of cortical bone. Bone 2:195–201
Acknowledgments
We are grateful of Mr. Evan Veregge for helping process the experimental data and the fellowship support to Ms. Xiao Yang (No. 201306020100) from China Scholarship Council (CSC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Xiao Yang, Chintan Gandhi, Md. Mizanur Rahman, Mark Appleford, Lian-Wen Sun, and Xiaodu Wang have nothing to disclose.
Human and Animal Rights and Informed Consent
The use of human cadaveric bone samples in this study was approved by the Institutional Review Board at the University of Texas at San Antonio.
Rights and permissions
About this article
Cite this article
Yang, X., Gandhi, C., Rahman, M.M. et al. Age-Related Effects of Advanced Glycation End Products (Ages) in Bone Matrix on Osteoclastic Resorption. Calcif Tissue Int 97, 592–601 (2015). https://doi.org/10.1007/s00223-015-0042-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-0042-1