Abstract
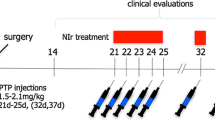
We have shown previously that near-infrared light (NIr), when applied at the same time as a parkinsonian insult (e.g. 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MPTP), reduces behavioural deficits and offers neuroprotection. Here, we explored whether the timing of NIr intervention—either before, at the same time or after the MPTP insult—was important. Mice received MPTP injections (total of 50 mg/kg) and, at various stages in relation to these injections, extracranial application of NIr. Locomotor activity was tested with an open-field test, and brains were processed for immunohistochemistry. Our results showed that regardless of when NIr was applied in relation to MPTP insult, behavioural impairment was reduced by a similar magnitude. The beneficial effect of NIr was fast-acting (within minutes) and long-lasting (for several days). There were more dopaminergic cells in the NIr-treated MPTP groups than in the MPTP group; there was no clear indication that a particular combination of NIr treatment and MPTP injection resulted in a higher cell number. In summary, irrespective of whether it was applied before, at the same time as or after MPTP insult, NIr reduced both behavioural and structural measures of damage by a similar magnitude. There was a broad therapeutic time window of NIr application in relation to the stage of toxic insult, and the NIr was fast-acting and long-lasting.





Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- LED:
-
Light-emitting diode
- MG:
-
Medial geniculate nucleus
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- NIr:
-
Near-infrared light
- PaG:
-
Periaqueductal grey matter
- PBS:
-
Phosphate-buffered saline
- Red:
-
Red nucleus
- SNc:
-
Substantia nigra pars compacta
- SNr:
-
Substantia nigra pars reticulata
- TH:
-
Tyrosine hydroxylase
- VTA:
-
Ventral tegmental area
- 6OHDA:
-
6-Hydroxydopamine
References
Aravamuthan BR, Bergstrom DA, French RA et al (2008) Altered neuronal activity relationships between the pedunculopontine nucleus and motor cortex in a rodent model of Parkinson’s disease. Exp Neurol 213:268–280. doi:10.1016/j.expneurol.2008.05.023
Begum R, Powner MB, Hudson N et al (2013) Treatment with 670 nm light up regulates cytochrome C oxidase expression and reduces inflammation in an age-related macular degeneration model. PLoS One 8:e57828. doi:10.1371/journal.pone.0057828
Bezard E, Yue Z, Kirik D, Spillantini MG (2013) Animal models of Parkinson’s disease: limits and relevance to neuroprotection studies. Mov Disord 28:61–70. doi:10.1002/mds.25108
Blesa J, Phani S, Jackson-Lewis V, Przedborski S (2012) Classic and new animal models of Parkinson’s disease. J Biomed Biotechnol 2012:845618. doi:10.1155/2012/845618
Bové J, Perier C (2012) Neurotoxin-based models of Parkinson’s disease. Neuroscience 211:51–76. doi:10.1016/j.neuroscience.2011.10.057
Chung H, Dai T, Sharma SK et al (2012) The nuts and bolts of low-level laser (light) therapy. Ann Biomed Eng 40:516–533. doi:10.1007/s10439-011-0454-7
Darlot F, Moro C, El Massri N et al (2015) Near-infrared light is neuroprotective in a monkey model of Parkinson’s disease. Ann Neurol. doi:10.1002/ana.24542
El Massri N, Johnstone DM, Peoples CL et al (2015) The effect of different doses of near infrared light on dopaminergic cell survival and gliosis in MPTP-treated mice. Int J Neurosci. doi:10.3109/00207454.2014.994063
Gonzalez-Lima F, Barrett DW (2014) Augmentation of cognitive brain functions with transcranial lasers. Front Syst Neurosci 8:36. doi:10.3389/fnsys.2014.00036
Gkotsi D, Begum R, Salt T et al (2014) Recharging mitochondrial batteries in old eyes. Near infra-red increases ATP. Exp Eye Res 122:50–53. doi:10.1016/j.exer.2014.02.023
Hamblin MR, Demidova TN (2006) Mechanisms of low level light therapy. In: Mechanisms for low-light therapy. In: Proc. of SPIE 6140 (The International Society for Optical Engineering)
Jankovic J, Poewe W (2012) Therapies in Parkinson’s disease. Curr Opin Neurol 25:433–447. doi:10.1097/WCO.0b013e3283542fc2
Johnstone D, Coleman K, Moro C et al (2014a) The potential of light therapy in Parkinson’s disease. Chronophysiol Ther 4:1–14. doi:10.2147/CPT.S57180
Johnstone DM, El Massri N, Moro C et al (2014b) Indirect application of near infrared light induces neuroprotection in a mouse model of parkinsonism—an abscopal neuroprotective effect. Neuroscience 274:93–101. doi:10.1016/j.neuroscience.2014.05.023
Johnstone DM, Moro C, Stone J et al (2016) Turning on lights to stop neurodegeneration: the potential of near infrared light therapy in Alzheimer’s and Parkinson’s disease. Front Neurosci. doi:10.3389/fnins.2015.00500
Liang HL, Whelan HT, Eells JT, Wong-Riley MTT (2008) Near-infrared light via light-emitting diode treatment is therapeutic against rotenone- and 1-methyl-4-phenylpyridinium ion-induced neurotoxicity. Neuroscience 153:963–974. doi:10.1016/j.neuroscience.2008.03.042
Moro C, Torres N, El Massri N et al (2013) Photobiomodulation preserves behaviour and midbrain dopaminergic cells from MPTP toxicity: evidence from two mouse strains. BMC Neurosci 14:40. doi:10.1186/1471-2202-14-40
Moro C, El Massri N, Torres N et al (2014) Photobiomodulation inside the brain: a novel method of applying near-infrared light intracranially and its impact on dopaminergic cell survival in MPTP-treated mice. J Neurosurg 120:670–683. doi:10.3171/2013.9.JNS13423
Olanow CW, Kieburtz K, Schapira AHV (2008) Why have we failed to achieve neuroprotection in Parkinson’s disease? Ann Neurol 64(Suppl 2):S101–S110. doi:10.1002/ana.21461
Oueslati A, Lovisa B, Perrin J et al (2015) Photobiomodulation suppresses alpha-synuclein-induced toxicity in an AAV-based rat genetic model of Parkinson’s disease. PLoS One 10:e0140880. doi:10.1371/journal.pone.0140880
Paxinos G, Franklin K (2001) The mouse brain in stereotaxic coordinates, 2nd edn. Academic Press, Millbrae
Peoples C, Spana S, Ashkan K et al (2012) Photobiomodulation enhances nigral dopaminergic cell survival in a chronic MPTP mouse model of Parkinson’s disease. Parkinsonism Relat Disord 18:469–476. doi:10.1016/j.parkreldis.2012.01.005
Purushothuman S, Nandasena C, Johnstone DM et al (2013) The impact of near-infrared light on dopaminergic cell survival in a transgenic mouse model of parkinsonism. Brain Res 1535:61–70. doi:10.1016/j.brainres.2013.08.047
Quirk BJ, Torbey M, Buchmann E et al (2012) Near-infrared photobiomodulation in an animal model of traumatic brain injury: improvements at the behavioral and biochemical levels. Photomed Laser Surg 30:523–529. doi:10.1089/pho.2012.3261
Reinhart F, El Massri N, Darlot F et al (2014) 810 nm near-infrared light offers neuroprotection and improves locomotor activity in MPTP-treated mice. Neurosci Res. doi:10.1016/j.neures.2014.11.005
Reinhart F, El Massri N, Darlot F et al (2015) Evidence for improved behaviour and neuroprotection after intracranial application of near infrared light in a hemi-parkinsonian rat model. J Neurosurg 27:1–13, PMID: 26613166
Rojas J, Gonzaalez-Lima F (2011) Low-level light therapy of the eye and brain. Eye Brain 3:49–67
Schapira AHV, Olanow CW, Greenamyre JT, Bezard E (2014) Slowing of neurodegeneration in Parkinson’s disease and Huntington’s disease: future therapeutic perspectives. Lancet 384:545–555. doi:10.1016/S0140-6736(14)61010-2
Schober A (2004) Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell Tissue Res 318:215–224. doi:10.1007/s00441-004-0938-y
Shaw VE, Spana S, Ashkan K et al (2010) Neuroprotection of midbrain dopaminergic cells in MPTP-treated mice after near-infrared light treatment. J Comp Neurol 518:25–40. doi:10.1002/cne.22207
Shaw VE, Peoples C, Spana S et al (2012) Patterns of cell activity in the subthalamic region associated with the neuroprotective action of near-infrared light treatment in MPTP-treated mice. Parkinsons Dis 2012:296875. doi:10.1155/2012/296875
Trimmer PA, Schwartz KM, Borland MK et al (2009) Reduced axonal transport in Parkinson’s disease cybrid neurites is restored by light therapy. Mol Neurodegener 4:26. doi:10.1186/1750-1326-4-26
Valencia M, Chavez M, Artieda J et al (2014) Abnormal functional connectivity between motor cortex and pedunculopontine nucleus following chronic dopamine depletion. J Neurophysiol 111:434–440. doi:10.1152/jn.00555.2013
Wallace BA, Ashkan K, Heise CE et al (2007) Survival of midbrain dopaminergic cells after lesion or deep brain stimulation of the subthalamic nucleus in MPTP-treated monkeys. Brain 130:2129–2145. doi:10.1093/brain/awm137
Whelan HT, DeSmet KD, Buchmann EV et al (2008) Harnessing the cell’s own ability to repair and prevent neurodegenerative disease. SPIE Newsroom 24:1–3
Ying R, Liang HL, Whelan HT et al (2008) Pretreatment with near-infrared light via light-emitting diode provides added benefit against rotenone- and MPP+-induced neurotoxicity. Brain Res 1243:167–173. doi:10.1016/j.brainres.2008.09.057
Acknowledgments
We are forever grateful to Michael J. Fox Foundation, Credit Agricole Sud Rhones Alpes, Fondation Philanthropique Edmond J. Safra, France Parkinson and the French National Research Agency (ANR Carnot Institute), Tenix corp and Salteri family for funding this work. D.M.J. is an Early Career Fellow of the NHMRC, Australia. J.S. was supported by the Foote Foundation and Sir Zelman Cowen Universities Fund; he is Director of CSCM Pty Ltd.
Author contributions
C.M., D.M.J., J.S., A.L.B. and J.M. are full-time members of staff at their respective institutions, while F.R. and N.E.M. are postgraduate students. All authors contributed to the analysis of the data and of the reading and writing of the manuscript. F.R., C.M., N.E.M., A.L.B. and J.M. contributed to the experimental work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper, except for the funding provided by the organisations mentioned in the Acknowledgements.
Rights and permissions
About this article
Cite this article
Reinhart, F., El Massri, N., Johnstone, D.M. et al. Near-infrared light (670 nm) reduces MPTP-induced parkinsonism within a broad therapeutic time window. Exp Brain Res 234, 1787–1794 (2016). https://doi.org/10.1007/s00221-016-4578-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4578-8