Abstract
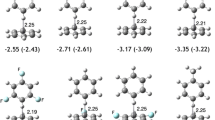
Intermolecular interactions between phenol and benzene molecules with the T-shaped geometries could be diverse. They can be pursued via two structural series: one with benzene standing above phenol using one or two C–H bonds as the anchoring point and the other with phenol standing on top of benzene with either C–H or O–H bond as the leg. In this work, structure and interaction properties of these species are investigated at the DFT M06-2X/6-311++G(2d,2p) level of theory without and with the counterpoise correction. A total of twelve distinct isomers have been identified, eight of which were unveiled for the first time. It is found that π–π interactions play essential roles in stabilizing these conformations, while C–H/π, C–H···O van der Waals interactions, and C–H···O and O–H/π hydrogen bonds are also involved and positively contribute to the stability of these species. Our energy decomposition analysis shows that the driving force for the formation of these complexes arises from attractive electrostatic, exchange, polarization, and dispersion terms, balanced by the repulsion term. The dispersion effect plays a dominant role, but the electrostatic and exchange terms are also markedly significant. Their close relationships between one another have also been disclosed.




Similar content being viewed by others
References
Salonen LM, Ellermann M, Diederich F (2011) Angew Chem Int Ed 50:4808–4842
Meyer EA, Castellano RK, Diederich F (2003) Angew Chem Int Ed 42:1210–1250
Schneider H-J (2009) Angew Chem Int Ed 48:3924–3977
Sinnokrot MO, Sherrill CD (2006) J Phys Chem A 110:10656–10668 (and references therein)
Hunter CA, Sanders JKM (1990) J Am Chem Soc 112:5525–5534
van der Avoird A, Podeszwa R, Szalewicz K, Leforestier C, van Harrevelt R, Bunker PR, Schnell M, von Helden G, Meijer G (2010) Phys Chem Chem Phys 12:8219–8240
Tsuzuki S, Uchimaru T, Sugawara K, Mikami M (2002) J Chem Phys 117:11216–11221
Tsuzuki S, Honda K, Uchimaru T, Mikami M, Tanabe K (2002) J Am Chem Soc 124:104–112
Sÿpirko V, Engkvist O, Soldan P, Selzle HL, Schlag EW, Hobza P (1999) J Chem Phys 111:572–582
Smith T, Slipchenko LV, Gordon MS (2008) J Phys Chem A 112:5286–5294
Sinnokrot MO, Valeev EF, Sherrill CD (2002) J Am Chem Soc 124:10887–10893
Singh NJ, Min SK, Kim DY, Kim KS (2009) J Chem Theory Comput 5:515–529
Sherrill CD (2013) Acc Chem Res 46:1020–1028
Schnell M, Erlekam U, Bunker PR, von Helden G, Grabow J-U, Meijer G, van der Avoird A (2013) Angew Chem Int Ed 52:5180–5183
Schnell M, Erlekam U, Bunker PR, von Helden G, Grabow J-U, Meijer G, van der Avoird A (2013) Phys Chem Chem Phys 15:10207–10223
Sanders JM (2010) J Phys Chem A 114:9205–9211
Řezáč J, Hobza P (2008) J Chem Theory Comput 4:1835–1840
Ding Y, Mei Y, Zhang JZ, Tao FM (2008) J Comput Chem 29:275–279
Sinnokrot MO, Sherrill CD (2004) J Phys Chem A 108:10200–10207
Wheeler SE, Houk KN (2009) Mol Phys 107:749–760
Wheeler SE, Houk KN (2008) J Am Chem Soc 130:10854–10855
Wheeler SE (2013) Acc Chem Res 46:1029–1038
Wheeler SE (2011) J Am Chem Soc 133:10262–10274
Watt M, Hardebeck LKE, Kirkpatrick CC, Lewis M (2011) J Am Chem Soc 133:3854–3862
Sinnokrot MO, Sherrill CD (2004) J Am Chem Soc 126:7690–7697
Sinnokrot MO, Sherrill CD (2003) J Phys Chem A 107:8377–8379
Ringer AL, Sinnokrot MO, Lively RP, Sherrill CD (2006) Chem Eur J 12:3821–3828
Ringer AL, Sherrill CD (2009) J Am Chem Soc 131:4574–4575
Raju RK, Bloom JWG, An Y, Wheeler SE (2011) ChemPhysChem 12:3116–3130
Lee EC, Hong BH, Lee JY, Kim JC, Kim D, Kim Y, Tarakeshwar P, Kim KS (2005) J Am Chem Soc 127:4530–4537
Hohenstein EG, Duan J, Sherrill CD (2011) J Am Chem Soc 133:13244–13247
Arnstein SA, Sherrill CD (2008) Phys Chem Chem Phys 10:2646–2655
Raju RK, Bloom JWG, Wheeler SE (2013) J Chem Theory Comput 9:3479–3490
Lee EC, Kim D, Jurecˇka P, Tarakeshwar P, Hobza P, Kim KS (2007) J Phys Chem A 111:3446–3457
Grimme S, Antony J, Schwabe T, Mück-Lichtenfeld C (2007) Org Biomol Chem 5:741–758
Seo J-I, Kim I, Lee YS (2009) Chem Phys Lett 474:101–106
Nishio M, Hirota M, Umezawa Y (1998) The CH/π interaction: evidence, nature, and consequences. Wiley-VCH, New York
Kwac K, Lee C, Jung Y, Han J, Kwak K, Zheng J, Fayer MD, Cho M (2006) J Chem Phys 125:244508–244516
Nikolova V, Ilieva S, Galabov B, Schaefer HF III (2014) J Org Chem 79:6823–6831
Zheng J, Kwak K, Asbury J, Chen X, Piletic IR, Fayer MD (2005) Science 309:1338–1343
Saggu M, Levinson NM, Boxer SG (2011) J Am Chem Soc 133:17414–17419
Saggu M, Levinson NM, Boxer SG (2012) J Am Chem Soc 134:18986–18997
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215–241
Zhao Y, Truhlar DG (2008) Acc Chem Res 41:157–167
Zhao Y, Truhlar DG (2007) J Chem Theory Comput 3:289–300
Zhao Y, Truhlar DG (2006) J Chem Phys 125:194101
Zhao Y, Schultz NE, Truhlar DG (2006) J Chem Theory Comput 2:364–382
Zhao Y, Schultz NE, Truhlar DG (2005) J Chem Phys 123:161103
Hohenstein EG, Chill ST, Sherrill CD (2008) J Chem Theory Comput 4:1996–2000
Liu Y, Zhao J, Li F, Chen Z (2013) J Comput Chem 34:121–131
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision, A. 02. Gaussian Inc., Wallingford
Johnson ER, Keinan S, Mori-Sánchez P, Contreras-García J, Cohen AJ, Yang W (2010) J Am Chem Soc 132:6498–6506
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, England
Bader RFW (1991) Chem Rev 91:893–928
Zhou P-P, Qiu W-Y (2009) ChemPhysChem 10:1847–1858
Zhou P-P, Qiu W-Y (2009) J Phys Chem A 113:10306–10320
Zhikol OA, Shishkin OV, Lyssenko KA, Leszczynski J (2005) J Chem Phys 122:144104
Quinonero D, Frontera A, Deya PM, Alkorta I, Elguero J (2008) Chem Phys Lett 460:406–410
Lipkowski P, Grabowski SJ, Robinson TL, Leszczynski J (2004) J Phys Chem A 108:10865–10872
Kock U, Popelier PLA (1995) J Phys Chem 99:9747–9754
Ebrahimi A, Habibi-Khorassani M, Gholipour AR, Masoodi HR (2009) Theor Chem Acc 124:115–122
Czyżnikowska Ż, Góra RW, Zaleśny R, Lipkowski P, Jarzembska K, Dominiak P, Leszczynski J (2010) J Phys Chem B 114:9629–9644
Keith TA (2009) AIMALL Version 09.11.29, aim.tkgristmill.com
Lu T, Chen F (2012) J Comput Chem 33:580–592
Su P, Li H (2009) J Chem Phys 131:014102–014115
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su S, Windus TL, Dupuis M, Montgomery JA Jr (1993) J Comput Chem 14:1347–1363
Cohen AJ, Mori-Sánchez P, Yang W (2008) Science 321:792–794
Gibbs GV, Cox DF, Rosso KM (2004) J Phys Chem A 108:7643–7645
Arnold WD, Oldfied E (2000) J Am Chem Soc 122:12835–12841
Fang D, Piquemal J-P, Liu S, Cisneros GA (2014) Theor Chem Acc 133:1484
Liu SB (2013) J Phys Chem A 117:962–965
Liu SB, Schauer CK (2015) J Chem Phys 142:054107
Liu SB (2016) Acta Phys Chim Sin 32:98–118
Acknowledgments
The work was financially supported by the National Natural Science Foundation of China (Grant No. 21403097) and the Fundamental Research Funds for the Central Universities (lzujbky-2014-182).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published as part of the special collection of articles “Festschrift in honour of A. Vela”.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, PP., Yang, X., Zhou, DG. et al. T-shaped phenol–benzene complexation driven by π-involved noncovalent interactions. Theor Chem Acc 135, 100 (2016). https://doi.org/10.1007/s00214-016-1863-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-016-1863-3