Abstract
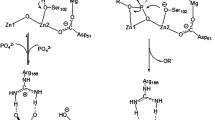
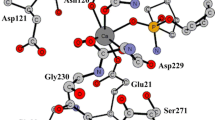
Phosphoserine phosphatase (PSP) utilizes one Mg2+ ion to catalyze the hydrolysis of phospho-l-serine. The displacement of Mg2+ by Ca2+ results in the loss of activity. The reaction mechanisms for the enzyme with both Mg2+ and Ca2+ bound were investigated using hybrid density functional theory. A large quantum chemical model abstracted from the X-ray crystal structure was employed in the calculations. Our calculations shed new insight into the catalytic mechanism of the natural enzyme and its lack of activity by Ca2+ substitution. For the catalytic reaction, our calculations showed that the whole reaction proceeds through two steps, namely dephosphorylation and phosphate hydrolysis. The associated barriers for these two steps are calculated to be 11.9 and 12.0 kcal mol−1, respectively. The Mg-bound Asp11 residue functions as a nucleophile to attack the phosphorus moiety, in concomitant with the departure of the leaving group, which takes a proton from the neutral Asp13 residue. In the subsequent step, the newly formed anionic Asp13 residue activates a water molecule to perform the reverse attack on the phosphoryl intermediate, affording the phosphate product. The substitution of Mg2+ by Ca2+ results in different metal coordination fashion, in which the Asp167 residue changes from bidentate to monodentate and a second water molecule becomes ligated to Ca2+. The calculated barriers for the hydrolysis are ca 8 kcal mol−1 higher than those in the native enzyme, which reconciles with the fact that Ca2+ inhibits the activity of PSP. Several possible reasons are discussed.









Similar content being viewed by others
References
Wang W, Cho Ho S, Kim R, Jancarik J, Yokota H, Nguyen HH, Grigoriev Igor V, Wemmer DE, Kim S-H (2002) J Mol Biol 319:421–431
Kim H-Y, Heo Y-S, Kim JH, Park MH, Moon J, Kim E, Kwon D, Yoon J, Shin D, Jeong E, Park SY, Lee TG, Jeon YH, Ro S, Cho JM, Hwang KY (2002) J Biol Chem 277:46651–46658
Wang W, Kim R, Jancarik J, Yokota H, Kim S-H (2001) Structure 9:65–71
Wolosker H, Sheth KN, Takahashi M, Mothet J-P, Brady RO Jr, Ferris CD, Snyder SO (1999) Proc Natl Acad Sci USA 96:721–725
Dunlop DS, Neidle A (1997) Biochem Biophys Res Commun 235:26–30
Matsui T, Sekiguchi M, Hashimoto A, Tomita U, Nishikawa T, Wada K (1995) J Neurochem 65:454–458
Berger AJ, Dieudonné S, Ascher P (1998) J Neurophysiol 80:3336–3340
Sugiura N, Patel RG, Corriveau RA (2001) J Biol Chem 276:14257–14263
Wood PL (1995) Life Sci 57:301–310
Paudice P, Gemignani A, Raiteri M (1998) Eur J Neurosci 10:2934–2944
Veeranna, Shetty KT (1991) Neurochem Res 15:1203–1210
Hawkinson JE, Acosta-Burruel M, Ta ND, Wood PL (1997) Eur J Pharmacol 337:315–324
Collet J-F, Stroobant V, Van Schaftingen E (1999) J Biol Chem 274:33985–33990
Collet J-F, Stroobant V, Pirard M, Delpierre G, Van Schaftingen E (1998) J Biol Chem 273:14107–14112
MacLennan DH, Clarke DM, Loo TW, Skerjanc IS (1992) Acta Physiol Scand Suppl 607:141–150
Lingrel JB, Kuntzweiler T (1994) J Biol Chem 269:19659–19662
Cho H, Wang W, Kim R, Yokota H, Damo S, Kim S-H, Wemmer D, Kustu S, Yan D (2001) Proc Natl Acad Sci USA 98:8525–8530
Peeraer Y, Rabijns A, Collet J-F, Van Schaftingen E, De Ranter C (2004) Eur J Biochem 271:3421–3427
Peeraer Y, Rabijns A, Verboven C, Collet J-F, Van Schaftingen E, De Ranter C (2003) Acta Cryst D59:971–977
Rinaldo-Matthis A, Rampazzo C, Reichard P, Bianchi V, Nordlund P (2002) Nat Struct Biol 9:779–787
Hisano T, Hata Y, Fujii T, Liu JQ, Kurihara T, Esaki N, Soda K (1996) J Biol Chem 271:20322–20330
Toyoshima C, Nakasako M, Nomura H, Ogawa H (2000) Nature 405:647–655
Lahiri SD, Zhang GF, Dunaway-Mariano D, Allen KN (2003) Science 299:2067–2071
Allegrini S, Scaloni A, Ferrara L, Pesi R, Pinna P, Sgarrella F, Camici M, Eriksson S, Tozzi MG (2001) J Biol Chem 276:33526–33532
Re S, Jung J, Ten-no S, Sugita Y (2009) Chem Phys Lett 480:284–288
Re S, Imai T, Jung J, Ten-no S, Sugita Y (2011) J Comput Chem 32:260–270
Dudev T, Lim C (2007) Acc Chem Res 40:85–93
Babu CS, Dudev T, Casareno R, Cowan JA, Lim C (2003) J Am Chem Soc 125:9318–9328
Dudev T, Lim C (2004) J Phys Chem B 108:4546–4557
Becke AD (1993) J Chem Phys 98:1372–1377
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Himo F, Guo J-D, Rinaldo-Matthis A, Nordlund P (2005) J Phys Chem B 109:20004–20008
Himo F, Siegbahn PEM (2003) Chem Rev 103:2421–2456
Noodleman L, Lovell T, Han W-G, Li J, Himo F (2004) Chem Rev 104:459–508
Siegbahn PEM, Borowski T (2006) Acc Chem Res 39:729–738
Himo F (2006) Theo Chem Acc 116:232–240
Ramos MJ, Fernandes PA (2008) Acc Chem Res 41:689–698
Chen S-L, Fang W-H, Himo F (2007) J Phys Chem B 111:1253–1255
Liao R-Z, Yu J-G, Himo F (2011) J Inorg Biochem 105:927–936
Liao R-Z, Yu J-G, Raushel FM, Himo F (2008) Chem Eur J 14:4287–4292
Liao R-Z, Yu J-G, Himo F (2009) Inorg Chem 48:1442–1448
Chen S-L, Fang W-H, Himo F (2009) J Inorg Biochem 103:274–281
Yang L, Liao R-Z, Yu J-G, Liu R-Z (2009) J Phys Chem B 113:6505–6510
Liao R-Z, Himo F, Yu J-G, Liu R-Z (2009) Eur J Inorg Chem 20:2967–2972
Liao R-Z, Himo F, Yu J-G, Liu R-Z (2010) J Inorg Biochem 104:37–46
Liao R-Z, Himo F, Yu J-G (2010) J Phys Chem B 114:2533–2540
Liao R-Z, Himo F, Yu J-G (2010) Inorg Chem 49:6883–6888
Leopoldini M, Russo N, Toscano M (2007) J Am Chem Soc 129:7776–7784
Abashkin YG, Burt SK, Collins JR, Cachau RE, Russo N, Erickson JW (1996) In: Russo N, Salahub DR (eds) Metal-ligand interactions: structure and reactivity, Nato Science Series. Kluwer, Dordrecht
Olsen L, Anthony J, Ryde U, Adolph H-W, Hemmingsen L (2003) J Phys Chem B 107:2366–2375
Marino T, Russo N, Toscano M (2005) J Am Chem Soc 127:4242–4253
Leopoldini M, Russo N, Toscano M (2006) J Phys Chem B 110:1063–1072
Zhang ZY (1998) Crit Rev Biochem Mol Biol 33:1–52
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, revision C02. Gaussian, Inc., Wallingford
Zhao Y, Truhlar DG (2004) J Phys Chem A 108:6908–6918
Zhao Y, Truhlar DG (2008) Acc Chem Res 41:157–167
Barone V, Cossi M (1998) J Phys Chem A 102:1995–2001
Cammi R, Mennucci B, Tomasi J (1999) J Phys Chem A 103:9100–9108
Klamt A, Schüürmann G (1993) J Chem Soc Perkin Trans 2:799–805
Tomasi J, Mennucci B, Cammi R (2005) Chem Rev 105:2999–3093
Hu P, Zhang Y (2006) J Am Chem Soc 128:1272–1278
Senn HM, Thiel S, Thiel W (2005) J Chem Theory Comput 1:494–505
Senn HM, Kästner J, Breidung J, Thiel W (2009) Can J Chem 87:1322–1337
Sousa SF, Fernandes PA, Ramos MJ (2009) Chem Eur J 15:4243–4247
Sousa SF, Fernandes PA, Ramos MJ (2007) J Am Chem Soc 129:1378–1385
Sousa SF, Fernandes PA, Ramos MJ (2005) J Mol Struct: THEOCHEM 729:125–129
Sousa SF, Fernandes PA, Ramos MJ (2005) J Biol Inorg Chem 10:3–10
Sousa SF, Fernandes PA, Ramos MJ (2005) Biophys J 88:483–494
Sousa SF, Fernandes PA, Ramos MJ (2007) J Comput Chem 28:1160–1168
Ryde U (1999) Biophys J 77:2777–2787
Tamames B, Sousa SF, Tamames J, Fernandes PA, Ramos MJ (2007) Proteins: Struct, Funct, Bioinf 69:466–475
Robert V, Lemercier G (2006) J Am Chem Soc 128:1183–1187
Szeto MWY, Mujika JI, Zurek J, Mulholland AJ, Harvey JN (2009) J Mol Struct: THEOCHEM 898:106–114
Torrent M, Musaev DG, Morokuma K (2001) J Phys Chem B 105:322–327
Gherman BF, Baik MH, Lippard SJ, Friesner RA (2004) J Am Chem Soc 126:2978–2990
Voegtli WC, Khidekel N, Baldwin J, Ley BA, Bollinger JM, Rosenzweig AC (2000) J Am Chem Soc 122:3255–3261
Rosenzweig AC, Nordlund P, Takahara PM, Frederick CA, Lippard SJ (1995) Chem Biol 2:409–418
Whittington DA, Lippard S (2001) J Am Chem Soc 123:827–838
Dunietz BD, Beachy MD, Cao YX, Whittington DA, Lippard SJ, Friesner RA (2000) J Am Chem Soc 122:2828–2839
Paterová J, Heyda J, Jungwirth P, Shaffer CJ, Révész Á, Zins EL, Schröder D (2011) J Phys Chem A 115:6813–6819
Demsar A, Kosmrlj J, Petricek S (2002) J Am Chem Soc 124:3951–3958
Lemercier G, Mulliez E, Brouca-Cabarrecq C, Dahan F, Tuchagues JP (2004) Inorg Chem 43:2105–2113
Kuzelka J, Spingler B, Lippard SJ (2002) Inorg Chim Acta 337:212–222
Rardin RL, Bino A, Poganiuch P, Tolman WB, Liu S, Lippard SJ (1990) Angew Chem Int Ed 29:812–814
Baffert C, Collomb MN, Deronzier A, Kjaergaard-Knudsen S, Latour JM, Lund KH, McKenzie CJ, Mortensen M, Nielsen L, Thorup N (2003) J Chem Soc, Dalton Trans 9:1765–1772
Pursche D, Triller MU, Reddig N, Rompel A, Krebs BZ (2003) Anorg Allg Chem 629:24–28
Feig AL, Masschelein A, Bakac A, Lippard SJ (1997) J Am Chem Soc 119:334–342
Ducháčková L, Schröder D, Roithová J (2011) Inorg Chem 50:3153–3158
LeCloux DD, Barrios AM, Mizoguchi TJ, Lippard SJ (1998) J Am Chem Soc 120:9001–9014
Calvaresi M, Garavelli M, Bottoni A (2008) Proteins 73:527–538
Bounaga S, Laws AP, Galleni M, Page MI (1998) Biochem J 331:703–711
Åqvist J, Kolmodin K, Florian J, Warshel A (1999) Chem Biol 6:R71–R80
Klähn M, Rosta E, Warshel A (2006) J Am Chem Soc 128:15310–15323
Pelmenschikov V, Blomberg MRA, Siegbahn PEM (2002) J Biol Inorg Chem 7:284–298
Pelmenschikov V, Siegbahn PEM (2002) Inorg Chem 41:5659–5666
Siegbahn PEM (2004) J Biol Inorg Chem 9:577–590
Siegbahn PEM (2011) ChemPhysChem 12:3274–3280
Acknowledgments
We appreciate Dr. Sven de Marothy (from Stockholm University) for providing xyzviewer to create all the figures. This work was supported by grants from the National Natural Science Foundation of China (grant nos. 20733002, 20873008, 21073014, and 21203042), the Fundamental Research Funds for the Central Universities (Grant No. HIT. NSRIF. 2013057), and Major State Basic Research Development Programs (grant nos. 2004CB719903 and 2002CB613406).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, L., Liao, RZ., Ding, WJ. et al. Why calcium inhibits magnesium-dependent enzyme phosphoserine phosphatase? A theoretical study. Theor Chem Acc 131, 1275 (2012). https://doi.org/10.1007/s00214-012-1275-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-012-1275-y