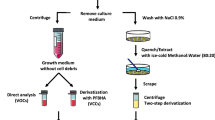
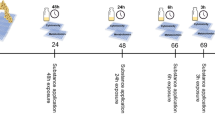
Abstract
3,4-Methylenedioxymethamphetamine (MDMA, ecstasy) is a well-known hepatotoxic drug. Although its toxicity has been thoroughly studied at high concentrations, there is still insufficient knowledge on possible alterations of cell function at subtoxic concentrations, which are in fact more representative concentrations of intoxication scenarios. In this study, a gas chromatography–mass spectrometry (GC–MS) metabolomics approach was used to investigate the metabolic changes in primary mouse hepatocytes (PMH) exposed to two subtoxic concentrations of MDMA (LC01 and LC10) for 24 h. Metabolomic profiling of both intracellular metabolites and volatile metabolites in the extracellular medium of PMH was performed. Multivariate analysis showed that the metabolic pattern of cells exposed to MDMA discriminates from the controls in a concentration-dependent manner. Exposure to LC10 MDMA induces a significant increase in some intracellular metabolites, including oleic acid and palmitic acid, and a decrease in glutamate, aspartate, 5-oxoproline, fumarate, malate, phosphoric acid, α-ketoglutarate and citrate. Extracellular metabolites such as acetophenone, formaldehyde, pivalic acid, glyoxal and 2-butanone were found significantly increased after exposure to MDMA, compared to controls, whereas 4-methylheptane, 2,4-dimethyl-1-heptene, nonanal, among others, were found significantly decreased. The panel of discriminatory metabolites is mainly involved in tricarboxylic acid (TCA) cycle, fatty acid metabolism, glutamate metabolism, antioxidant defenses and possibly changes in the liver enzyme machinery. Overall, these results highlight the potential of the intra- and extracellular metabolome to study alterations triggered by subtoxic concentrations of MDMA in hepatic cell functions, which represents a more realistic appraisal of early toxicity events posed by exposure to this drug. In addition, these results also revealed some metabolites that may be used as potential biomarkers indicative of early events in the hepatotoxicity induced by MDMA.





Similar content being viewed by others
References
Abordo EA, Minhas HS, Thornalley PJ (1999) Accumulation of alpha-oxoaldehydes during oxidative stress: a role in cytotoxicity. Biochem Pharmacol 58(4):641–648
Aickin M, Gensler H (1996) Adjusting for multiple testing when reporting research results: the Bonferroni vs Holm methods. Am J Public Health 86(5):726–728
Antolino-Lobo I, Meulenbelt J, van den Berg M, van Duursen MB (2011) A mechanistic insight into 3,4-methylenedioxymethamphetamine (“ecstasy”)-mediated hepatotoxicity. Vet Q 31(4):193–205. https://doi.org/10.1080/01652176.2011.642534
Araujo AM, Carvalho M, Carvalho F, Bastos ML, Guedes de Pinho P (2017) Metabolomic approaches in the discovery of potential urinary biomarkers of drug-induced liver injury (DILI). Crit Rev Toxicol 47(8):633–649. https://doi.org/10.1080/10408444.2017.1309638
Araujo AM, Moreira N, Lima AR et al (2018) Analysis of extracellular metabolome by HS-SPME/GC–MS: optimization and application in a pilot study to evaluate galactosamine-induced hepatotoxicity. Toxicol Lett 295:22–31. https://doi.org/10.1016/j.toxlet.2018.05.028
Arbo MD, Melega S, Stober R et al (2016) Hepatotoxicity of piperazine designer drugs: up-regulation of key enzymes of cholesterol and lipid biosynthesis. Arch Toxicol 90(12):3045–3060. https://doi.org/10.1007/s00204-016-1665-3
Atayan Y, Cagin YF, Erdogan MA, Harputluoglu MM, Bilgic Y (2015) Ecstasy induced acute hepatic failure. Case reports. Acta Gastroenterol Belg 78(1):53–55
Ayala A, Munoz MF, Arguelles S (2014) Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev 2014:360438. https://doi.org/10.1155/2014/360438
Barbosa DJ, Capela JP, Feio-Azevedo R, Teixeira-Gomes A, Bastos Mde L, Carvalho F (2015) Mitochondria: key players in the neurotoxic effects of amphetamines. Arch Toxicol 89(10):1695–1725. https://doi.org/10.1007/s00204-015-1478-9
Berben L, Sereika SM, Engberg S (2012) Effect size estimation: methods and examples. Int J Nurs Stud 49(8):1039–1047. https://doi.org/10.1016/j.ijnurstu.2012.01.015
Boxler MI, Liechti ME, Schmid Y, Kraemer T, Steuer AE (2017) First time view on human metabolome changes after a single intake of 3,4-methylenedioxymethamphetamine in healthy placebo-controlled subjects. J Proteome Res 16(9):3310–3320. https://doi.org/10.1021/acs.jproteome.7b00294
Capela JP, Carmo H, Remiao F, Bastos ML, Meisel A, Carvalho F (2009) Molecular and cellular mechanisms of ecstasy-induced neurotoxicity: an overview. Mol Neurobiol 39(3):210–271. https://doi.org/10.1007/s12035-009-8064-1
Carvalho M, Carvalho F, Bastos ML (2001) Is hyperthermia the triggering factor for hepatotoxicity induced by 3,4-methylenedioxymethamphetamine (ecstasy)? An in vitro study using freshly isolated mouse hepatocytes. Arch Toxicol 74(12):789–793
Carvalho M, Carvalho F, Remiao F, de Lourdes Pereira M, Pires-das-Neves R, de Lourdes Bastos M (2002) Effect of 3,4-methylenedioxymethamphetamine (“ecstasy”) on body temperature and liver antioxidant status in mice: influence of ambient temperature. Arch Toxicol 76(3):166–172. https://doi.org/10.1007/s00204-002-0324-z
Carvalho M, Pontes H, Remiao F, Bastos ML, Carvalho F (2010) Mechanisms underlying the hepatotoxic effects of ecstasy. Curr Pharm Biotechnol 11(5):476–495
Carvalho M, Carmo H, Costa VM et al (2012) Toxicity of amphetamines: an update. Arch Toxicol 86(8):1167–1231. https://doi.org/10.1007/s00204-012-0815-5
Catala A (2009) Lipid peroxidation of membrane phospholipids generates hydroxy-alkenals and oxidized phospholipids active in physiological and/or pathological conditions. Chem Phys Lipids 157(1):1–11. https://doi.org/10.1016/j.chemphyslip.2008.09.004
Chan EC, Pasikanti KK, Nicholson JK (2011) Global urinary metabolic profiling procedures using gas chromatography-mass spectrometry. Nat Protoc 6(10):1483–1499. https://doi.org/10.1038/nprot.2011.375
Cunha-Oliveira T, Rego AC, Oliveira CR (2013) Oxidative stress and drugs of abuse: an update. Mini Rev Org Chem 10(4):321–334. https://doi.org/10.2174/1570193X113106660026
da Silva DD, Silva E, Carmo H (2014) Combination effects of amphetamines under hyperthermia—the role played by oxidative stress. J Appl Toxicol 34(6):637–650. https://doi.org/10.1002/jat.2889
Elliott SP (2005) MDMA and MDA concentrations in antemortem and postmortem specimens in fatalities following hospital admission. J Anal Toxicol 29(5):296–300
El-Tawil OS, Abou-Hadeed AH, El-Bab MF, Shalaby AA (2011) d-Amphetamine-induced cytotoxicity and oxidative stress in isolated rat hepatocytes. Pathophysiology 18(4):279–285. https://doi.org/10.1016/j.pathophys.2011.04.001
EMCDDA (2016) Recent changes in Europe’s MDMA/ecstasy market: results from an EMCDDA trendspotter study. In. http://www.emcdda.europa.eu/. Accessed May 03, 2018
Goudarzi M, Kalantari H, Rezaei M (2017) Glyoxal toxicity in isolated rat liver mitochondria. Hum Exp Toxicol. https://doi.org/10.1177/0960327117715900
Gowing LR, Henry-Edwards SM, Irvine RJ, Ali RL (2002) The health effects of ecstasy: a literature review. Drug Alcohol Rev 21(1):53–63. https://doi.org/10.1080/09595230220119363
Hakim M, Broza YY, Barash O et al (2012) Volatile organic compounds of lung cancer and possible biochemical pathways. Chem Rev 112(11):5949–5966. https://doi.org/10.1021/cr300174a
Hayton S, Maker GL, Mullaney I, Trengove RD (2017) Untargeted metabolomics of neuronal cell culture: a model system for the toxicity testing of insecticide chemical exposure. J Appl Toxicol 37(12):1481–1492. https://doi.org/10.1002/jat.3498
Heijne WH, Kienhuis AS, van Ommen B, Stierum RH, Groten JP (2005) Systems toxicology: applications of toxicogenomics, transcriptomics, proteomics and metabolomics in toxicology. Expert Rev Proteom 2(5):767–780. https://doi.org/10.1586/14789450.2.5.767
Hewit NJ, Lechón MJG, Houston JB et al (2007) Primary hepatocytes: current understanding of the regulation of metabolic enzymes and transporter proteins, and pharmaceutical practice for the use of hepatocytes in metabolism, enzyme induction, transporter, clearance and hepatotoxicity studies. Drug Metab Rev 39(1):159–234. https://doi.org/10.1080/03602530601093489
Kell DB, Brown M, Davey HM, Dunn WB, Spasic I, Oliver SG (2005) Metabolic footprinting and systems biology: the medium is the message. Nat Rev Microbiol 3(7):557–565. https://doi.org/10.1038/nrmicro1177
Kumar A, Bachhawat AK (2012) Pyroglutamic acid: throwing light on a lightly studied metabolite. Curr Sci 102:288–297
Lange JN, Wood KD, Knight J, Assimos DG, Holmes RP (2012) Glyoxal formation and its role in endogenous oxalate synthesis. Adv Urol 2012:819202. https://doi.org/10.1155/2012/819202
Lange-Brock N, Berg T, Muller AR et al (2002) Acute liver failure following the use of ecstasy (MDMA). Z Gastroenterol 40(8):581–586. https://doi.org/10.1055/s-2002-33416
Leon Z, Garcia-Canaveras JC, Donato MT, Lahoz A (2013) Mammalian cell metabolomics: experimental design and sample preparation. Electrophoresis 34(19):2762–2775. https://doi.org/10.1002/elps.201200605
Lima AR, Araujo AM, Pinto J et al (2018a) Discrimination between the human prostate normal and cancer cell exometabolome by GC–MS. Sci Rep 8(1):5539. https://doi.org/10.1038/s41598-018-23847-9
Lima AR, Araujo AM, Pinto J et al (2018b) GC–MS-based endometabolome analysis differentiates prostate cancer from normal prostate cells. Metabolites. https://doi.org/10.3390/metabo8010023
Lindon JC, Holmes E, Nicholson JK (2006) Metabonomics techniques and applications to pharmaceutical research & development. Pharm Res 23(6):1075–1088. https://doi.org/10.1007/s11095-006-0025-z
Lu SC (2013) Glutathione synthesis. Biochim Biophys Acta 1830(5):3143–3153. https://doi.org/10.1016/j.bbagen.2012.09.008
Ma S, Chan KW, Lee TK et al (2008) Aldehyde dehydrogenase discriminates the CD133 liver cancer stem cell populations. Mol Cancer Res 6(7):1146–1153. https://doi.org/10.1158/1541-7786.MCR-08-0035
Mashima T, Seimiya H, Tsuruo T (2009) De novo fatty-acid synthesis and related pathways as molecular targets for cancer therapy. Br J Cancer 100(9):1369–1372. https://doi.org/10.1038/sj.bjc.6605007
Mastrangelo A, Ferrarini A, Rey-Stolle F, Garcia A, Barbas C (2015) From sample treatment to biomarker discovery: a tutorial for untargeted metabolomics based on GC-(EI)-Q-MS. Anal Chim Acta 900:21–35. https://doi.org/10.1016/j.aca.2015.10.001
Milroy CM (2011) “Ecstasy” associated deaths: what is a fatal concentration ? Analysis of a case series. Forensic Sci Med Pathol 7(3):248–252. https://doi.org/10.1007/s12024-010-9220-7
Mlakar A, Spiteller G (1996) Previously unknown aldehydic lipid peroxidation compounds of arachidonic acid. Chem Phys Lipids 79(1):47–53
Mochalski P, Sponring A, King J, Unterkofler K, Troppmair J, Amann A (2013) Release and uptake of volatile organic compounds by human hepatocellular carcinoma cells (HepG2) in vitro. Cancer Cell Int 13(1):72. https://doi.org/10.1186/1475-2867-13-72
Mochalski P, Al-Zoairy R, Niederwanger A, Unterkofler K, Amann A (2014) Quantitative analysis of volatile organic compounds released and consumed by rat L6 skeletal muscle cells in vitro. J Breath Res 8(4):046003. https://doi.org/10.1088/1752-7155/8/4/046003
Moon KH, Upreti VV, Yu LR et al (2008) Mechanism of 3,4-methylenedioxymethamphetamine (MDMA, ecstasy)-mediated mitochondrial dysfunction in rat liver. Proteomics 8(18):3906–3918. https://doi.org/10.1002/pmic.200800215
Moratalla R, Khairnar A, Simola N et al (2017) Amphetamine-related drugs neurotoxicity in humans and in experimental animals: main mechanisms. Prog Neurobiol 155:149–170. https://doi.org/10.1016/j.pneurobio.2015.09.011
Nielsen KL, Telving R, Andreasen MF, Hasselstrom JB, Johannsen M (2016) A metabolomics study of retrospective forensic data from whole blood samples of humans exposed to 3,4-methylenedioxymethamphetamine: a new approach for identifying drug metabolites and changes in metabolism related to drug consumption. J Proteome Res 15(2):619–627. https://doi.org/10.1021/acs.jproteome.5b01023
O’Brien PJ, Siraki AG, Shangari N (2005) Aldehyde sources, metabolism, molecular toxicity mechanisms, and possible effects on human health. Crit Rev Toxicol 35(7):609–662
Owen OE, Kalhan SC, Hanson RW (2002) The key role of anaplerosis and cataplerosis for citric acid cycle function. J Biol Chem 277(34):30409–30412. https://doi.org/10.1074/jbc.R200006200
Perrine SA, Michaels MS, Ghoddoussi F, Hyde EM, Tancer ME, Galloway MP (2009) Cardiac effects of MDMA on the metabolic profile determined with 1H-magnetic resonance spectroscopy in the rat. NMR Biomed 22(4):419–425. https://doi.org/10.1002/nbm.1352
Pizzimenti S, Ciamporcero E, Daga M et al (2013) Interaction of aldehydes derived from lipid peroxidation and membrane proteins. Front Physiol 4:242. https://doi.org/10.3389/fphys.2013.00242
Pluskal T, Castillo S, Villar-Briones A, Oresic M (2010) MZmine 2: modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinform 11:395. https://doi.org/10.1186/1471-2105-11-395
Pontes H, Sousa C, Silva R et al (2008) Synergistic toxicity of ethanol and MDMA towards primary cultured rat hepatocytes. Toxicology 254(1–2):42–50. https://doi.org/10.1016/j.tox.2008.09.009
Portuguese Government (2013) Decreto-Lei n.º 113/2013. Diário da República n.º 151/2013, Série I de 2013-08-07, pp 4709–4739
Reinke H, Asher G (2016) Circadian clock control of liver metabolic functions. Gastroenterology 150(3):574–580. https://doi.org/10.1053/j.gastro.2015.11.043
Robertson DG, Watkins PB, Reily MD (2011) Metabolomics in toxicology: preclinical and clinical applications. Toxicol Sci 120(Suppl 1):S146–S170. https://doi.org/10.1093/toxsci/kfq358
Rui L (2014) Energy metabolism in the liver. Compr Physiol 4(1):177–197. https://doi.org/10.1002/cphy.c130024
Ruiz-Aracama A, Peijnenburg A, Kleinjans J et al (2011) An untargeted multi-technique metabolomics approach to studying intracellular metabolites of HepG2 cells exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. BMC Genom 12:251. https://doi.org/10.1186/1471-2164-12-251
Shangari N, O’Brien PJ (2004) The cytotoxic mechanism of glyoxal involves oxidative stress. Biochem Pharmacol 68(7):1433–1442. https://doi.org/10.1016/j.bcp.2004.06.013
Shangari N, Bruce WR, Poon R, O’Brien PJ (2003) Toxicity of glyoxals—role of oxidative stress, metabolic detoxification and thiamine deficiency. Biochem Soc Trans 31(Pt 6):1390–1393. https://doi.org/10.1042/bst0311390
Singh S, Brocker C, Koppaka V et al (2013) Aldehyde dehydrogenases in cellular responses to oxidative/electrophilic stress. Free Radic Biol Med 56:89–101. https://doi.org/10.1016/j.freeradbiomed.2012.11.010
Sumner LW, Amberg A, Barrett D et al (2007) Proposed minimum reporting standards for chemical analysis Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 3(3):211–221. https://doi.org/10.1007/s11306-007-0082-2
Tapiero H, Mathe G, Couvreur P, Tew KD (2002) II. Glutamine and glutamate. Biomed Pharmacother 56(9):446–457
Taware R, Taunk K, Pereira JAM et al (2017) Investigation of urinary volatomic alterations in head and neck cancer: a non-invasive approach towards diagnosis and prognosis. Metabolomics 13:111. https://doi.org/10.1007/s11306-017-1251-6
Teng S, Beard K, Pourahmad J et al (2001) The formaldehyde metabolic detoxification enzyme systems and molecular cytotoxic mechanism in isolated rat hepatocytes. Chem Biol Interact 130–132(1–3):285–296
Trygg J, Holmes E, Lundstedt T (2007) Chemometrics in metabonomics. J Proteome Res 6(2):469–479. https://doi.org/10.1021/pr060594q
Valente MJ, Araujo AM, Bastos Mde L et al (2016a) Editor’s highlight: characterization of hepatotoxicity mechanisms triggered by designer cathinone drugs (beta-Keto Amphetamines). Toxicol Sci 153(1):89–102. https://doi.org/10.1093/toxsci/kfw105
Valente MJ, Araujo AM, Silva R et al (2016b) 3,4-Methylenedioxypyrovalerone (MDPV): in vitro mechanisms of hepatotoxicity under normothermic and hyperthermic conditions. Arch Toxicol 90(8):1959–1973. https://doi.org/10.1007/s00204-015-1653-z
Voulgaridou GP, Anestopoulos I, Franco R, Panayiotidis MI, Pappa A (2011) DNA damage induced by endogenous aldehydes: current state of knowledge. Mutat Res 711(1–2):13–27. https://doi.org/10.1016/j.mrfmmm.2011.03.006
Wheelock AM, Wheelock CE (2013) Trials and tribulations of ‘omics data analysis: assessing quality of SIMCA-based multivariate models using examples from pulmonary medicine. Mol Biosyst 9(11):2589–2596. https://doi.org/10.1039/c3mb70194h
Worley B, Powers R (2013) Multivariate analysis in metabolomics. Curr Metabolom 1(1):92–107. https://doi.org/10.2174/2213235X11301010092
Yuan L, Kaplowitz N (2009) Glutathione in liver diseases and hepatotoxicity. Mol Aspects Med 30(1–2):29–41. https://doi.org/10.1016/j.mam.2008.08.003
Acknowledgements
This work received financial support from the European Union (FEDER funds POCI/01/0145/FEDER/007728) and National Funds (FCT/MEC, Fundação para a Ciência e a Tecnologia and Ministério da Educação e Ciência) under the Partnership Agreement PT2020 UID/MULTI/04378/2013. The study is a result of the project NORTE-01-0145-FEDER-000024, supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement (DESignBIOtecHealth—New Technologies for three Health Challenges of Modern Societies: Diabetes, Drug Abuse and Kidney Diseases), through the European Regional Development Fund (ERDF). A. M. Araújo thanks to FCT for her PhD fellowship (SFRH/BD/107708/2015) and M. Carvalho also acknowledges FCT through the UID/MULTI/04546/2016 project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Araújo, A.M., Bastos, M.d.L., Fernandes, E. et al. GC–MS metabolomics reveals disturbed metabolic pathways in primary mouse hepatocytes exposed to subtoxic levels of 3,4-methylenedioxymethamphetamine (MDMA). Arch Toxicol 92, 3307–3323 (2018). https://doi.org/10.1007/s00204-018-2314-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2314-9