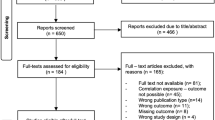
Abstract
Summary
Compared to pH ≥7.0 in Norwegian municipal drinking water, pH <7.0 increased the risk of forearm fractures in the population-based Cohort of Norway (CONOR; n = 127,272). The association was attenuated (p > 0.05) after adjustments for indicators of bacteria and organic matter, which may signify an association between poor drinking water and bone health.
Introduction
The Norwegian population has the highest rate of fractures ever reported. A large variation in fracture rate both between and within countries indicates that an environmental factor, such as the quality of drinking water, could be one of the causes of the disparities. Our aim was to investigate a possible association between pH (an important parameter for water quality) and self-reported forearm fracture and to examine whether other water quality factors could account for this association.
Methods
Using Geographic Information Systems, information on the quality of drinking water was linked to CONOR (n = 127,272; mean age, 50.2 ± 15.8 years), a database comprising ten regional epidemiological health surveys from across the country in the time period 1994–2003.
Results
The highest risk of forearm fracture was found at a pH of around 6.75, with a decreasing risk toward both higher and lower pH values. The increased adjusted odds of forearm fracture in men consuming municipal drinking water with pH <7.0 compared to water with pH ≥7.0 was odds ratio (OR) = 1.19 (95 % CI, 1.14, 1.25), and the corresponding increased odds in women was OR = 1.14 (95 % CI, 1.08, 1.19). This association was attenuated (p > 0.05) after further adjustments for other water quality factors (color grade, intestinal enterococci, and Clostridium perfringens).
Conclusions
Our findings indicate a higher risk of fracture when consuming water of an acidic pH; however, the risk does not only seem to be due to the acidity level per se, but also to other aspects of water quality associated with pH.


Similar content being viewed by others
References
Ismail AA, Pye SR, Cockerill WC, Lunt M et al (2002) Incidence of limb fracture across Europe: results from the European Prospective Osteoporosis Study (EPOS). Osteoporos Int 13:565–571
Lofthus CM, Frihagen F, Meyer HE, Nordsletten L, Melhuus K, Falch JA (2008) Epidemiology of distal forearm fractures in Oslo, Norway. Osteoporos Int 19:781–786
Chrischilles E, Shireman T, Wallace R (1994) Costs and health effects of osteoporotic fractures. Bone 15:377–386
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA III, Berger M (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739
Omsland TK, Ahmed LA, Gronskag A, Schei B, Emaus N, Langhammer A, Joakimsen RM, Jorgensen L, Sogaard AJ, Gjesdal CG, Meyer HE (2011) More forearm fractures among urban than rural women: the NOREPOS study based on the Tromso study and the HUNT study. J Bone Miner Res 26:850–856
Sanders KM, Nicholson GC, Ugoni AM, Seeman E, Pasco JA, Kotowicz MA (2002) Fracture rates lower in rural than urban communities: the Geelong Osteoporosis Study. J Epidemiol Community Health 56:466–470
Søgaard AJ, Gustad TK, Bjertness E, Tell GS, Schei B, Emaus N, Meyer HE (2007) Urban–rural differences in distal forearm fractures: Cohort Norway. Osteoporos Int 18:1063–1072
Flaten TP (2004) An investigation of the chemical composition of Norwegian drinking water and its possible relationships with the epidemiology of some diseases. Dissertation, Norwegian University of Science and Technology
Flaten TP (1991) A nation-wide survey of the chemical composition of drinking water in Norway. Sci Total Environ 102:35–73
Norwegian Institute of Public Health (2004) [Watersupply ABC: chapter C, watersources and catchment]. Drinking water quality and hygiene [in Norwegian]. Available at http://www.fhi.no/eway/default.aspx?pid=233&trg=MainArea_5661&MainArea_5661=5631:0:15,3030:1:0:0:::0:0. Accessed 5 May 2011
Norwegian Institute of Public Health (2006) [Watersupply ABC: chapter E, water distribution system]. Drinking water quality and hygiene [in Norwegian]. Available at http://www.fhi.no/eway/default.aspx?pid=233&trg=MainArea_5661&MainArea_5661=5631:0:15,3030:1:0:0:::0:0. Accessed 13 May 2011
Norwegian Institute of Public Health (2008) [Watersupply ABC: chapter D, water treatment]. Drinking water quality and hygiene [in Norwegian]. Available at http://www.fhi.no/eway/default.aspx?pid=233&trg=MainArea_5661&MainArea_5661=5631:0:15,3030:1:0:0:::0:0. Accessed 13 May 2011
Tomten SE, Hoestmark AT (2003) Acid and bone resorption. In: Bjertness E (ed) Osteoporosis: from mechanisms and risk factors to prevention, geographical differences in a country with the highest incidence rates of hip fractures in the world. The Norwegian Academy of Science and Letters, Oslo, pp 35–42
Rylander R (2008) Drinking water constituents and disease. J Nutr 138:423S–425S
Krapf R, Seldin DW, Alpern RJ (2000) Clinical syndromes of metabolic acidosis. In: Seldin DW, Giebisch G (eds) The kidney, physiology and pathophysiology, 3rd edn. Lippincott Williams and Wilkins, Philadelphia, pp 2073–2130
Frings-Meuthen P, Baecker N, Heer M (2008) Low-grade metabolic acidosis may be the cause of sodium chloride-induced exaggerated bone resorption. J Bone Miner Res 23:517–524
World Health Organization (2004) Guidelines for drinking-water quality, vol 1, 3rd edn. World Health Organization, Geneva
Frengstad B, Banks D, Siewers U (2001) The chemistry of Norwegian groundwaters: IV. The pH-dependence of element concentrations in crystalline bedrock groundwaters. Sci Total Environ 277:101–117
Nordberg GF, Goyer RA, Clarkson TW (1985) Impact of effects of acid precipitation on toxicity of metals. Environ Health Perspect 63:169–180
Prescott LM, Harley JP, Klein DA (2002) Microbial growth. In: Microbiology, 5th edn. McGraw-Hill, New York, pp 123–125
Ministry of health and care services (2011) [Regulations on water supply and drinking water], Norwegian Law Gazette [in Norwegian]. Available at http://www.lovdata.no/cgi-wift/ldles?doc=/sf/sf/sf-20011204-1372.html, Accessed 11 April 2011
Norwegian Institute of Public Health (2004) [Watersupply ABC: chapter B, water quality]. Drinking water quality and hygiene [in Norwegian]. Available at http://www.fhi.no/eway/default.aspx?pid=233&trg=MainArea_5661&MainArea_5661=5631:0:15,3030:1:0:0:::0:0. Accessed 29 April 2011
Naess O, Sogaard AJ, Arnesen E, Beckstrom AC, Bjertness E, Engeland A, Hjort PF, Holmen J, Magnus P, Njolstad I, Tell GS, Vatten L, Vollset SE, Aamodt G (2008) Cohort profile: cohort of Norway (CONOR). Int J Epidemiol 37:481–485
Norwegian Institute of Public Health (2011) [Waterworks register]. Drinking water quality and hygiene [in Norwegian]. Available at http://www.fhi.no/eway/default.aspx?pid=233&trg=MainArea_5661&MainArea_5661=5631:0:15,2873:1:0:0:::0:0. Accessed 24 March 2011
Cressie NAC (1993) Statistics for spatial data, revth edn. John Wiley & Sons INC, New York
Krieger NS, Frick KK, Bushinsky DA (2004) Mechanism of acid-induced bone resorption. Curr Opin Nephrol Hypertens 13:423–436
Wynn E, Krieg MA, Aeschlimann JM, Burckhardt P (2009) Alkaline mineral water lowers bone resorption even in calcium sufficiency: alkaline mineral water and bone metabolism. Bone 44:120–124
Rylander R, Remer T, Berkemeyer S, Vormann J (2006) Acid–base status affects renal magnesium losses in healthy, elderly persons. J Nutr 136:2374–2377
Heaney RP, Rafferty K (2001) Carbonated beverages and urinary calcium excretion. Am J Clin Nutr 74:343–347
Tucker KL, Morita K, Qiao N, Hannan MT, Cupples LA, Kiel DP (2006) Colas, but not other carbonated beverages, are associated with low bone mineral density in older women: the Framingham Osteoporosis Study. Am J Clin Nutr 84:936–942
Bernstein CN, Leslie WD (2004) Osteoporosis and inflammatory bowel disease. Aliment Pharmacol Ther 19:941–952
Henderson B, Nair SP (2003) Hard labour: bacterial infection of the skeleton. Trends Microbiol 11:570–577
Amcheslavsky A, Hemmi H, Akira S, Bar-Shavit Z (2005) Differential contribution of osteoclast- and osteoblast-lineage cells to CpG-oligodeoxynucleotide (CpG-ODN) modulation of osteoclastogenesis. J Bone Miner Res 20:1692–1699
Ciampolini J, Harding KG (2000) Pathophysiology of chronic bacterial osteomyelitis. Why do antibiotics fail so often? Postgrad Med J 76:479–483
Mjoberg B, Hellquist E, Mallmin H, Lindh U (1997) Aluminum, Alzheimer's disease and bone fragility. Acta Orthop Scand 68:511–514
Chen Z, Kooperberg C, Pettinger MB, Bassford T, Cauley JA, LaCroix AZ, Lewis CE, Kipersztok S, Borne C, Jackson RD (2004) Validity of self-report for fractures among a multiethnic cohort of postmenopausal women: results from the Women’s Health Initiative observational study and clinical trials. Menopause 11:264–274
Ismail AA, O’Neill TW, Cockerill W et al (2000) Validity of self-report of fractures: results from a prospective study in men and women across Europe. EPOS Study Group. European Prospective Osteoporosis Study Group. Osteoporos Int 11:248–254
Nevitt MC, Cummings SR, Browner WS et al (1992) The accuracy of self-report of fractures in elderly women: evidence from a prospective study. Am J Epidemiol 135:490–499
Joakimsen RM, Fonnebo V, Sogaard AJ, Tollan A, Stormer J, Magnus JH (2001) The Tromso study: registration of fractures, how good are self-reports, a computerized radiographic register and a discharge register? Osteoporos Int 12:1001–1005
Acknowledgments
This study was supported by grants from the Norwegian Research Council. The authors wish to acknowledge the services of Cohort of Norway (CONOR), the contributing research centers delivering data to CONOR, and all the study participants. Carl Fredrik Nordheim and Liliane Myrstad (Norwegian Waterworks Register) have contributed greatly in the collection and validation of data on water quality.
Conflicts of interest
Professor Tell did not report receiving fees, honoraria, grants, or consultancies. The Department of Public Health and Primary Health Care, University of Bergen, Bergen, Norway is, however, involved in studies with funding from a pharmaceutical company as a research grant to (and administered by) the university. This study has no relation to the present study. All other authors declare that they do not have any financial conflicts of interest. In addition, all authors report that they did not have any limitations on access to data or other materials critical to the work being reported.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
The members of the NOREPOS Core Research Group are Nina Emaus, Guri Grimnes (Tromsø), Haakon E. Meyer, Anne Johanne Søgaard (Oslo), Berit Schei, Siri Forsmo (Nord-Trøndelag), Grethe S. Tell, and Clara Gram Gjesdal (Bergen).
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 228 kb)
Rights and permissions
About this article
Cite this article
Dahl, C., Søgaard, A.J., Tell, G.S. et al. Is the quality of drinking water a risk factor for self-reported forearm fractures? Cohort of Norway. Osteoporos Int 24, 541–551 (2013). https://doi.org/10.1007/s00198-012-1989-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-1989-7