Abstract
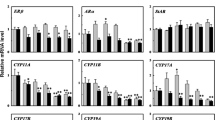
β-Agonists, which are used as human pharmaceuticals or feed additives, have been detected in aquatic environments. β-Agonists have also been proposed for use in aquaculture. However, there are limited data available regarding the adverse effects of β-agonists in aquatic organisms. In this study, ractopamine was selected as the representative β-agonist, and medaka embryos were exposed at concentrations ranging from 5 to 625 μg/L for 44 days. In contrast to what has been found in mammals, ractopamine caused no growth response in medaka. However, the transcriptional changes of genes related to the hypothalamic–pituitary–gonadal (HPG) axis, especially in females, suggested that β-agonists may have the potential to disrupt the endocrine system. Moreover, genes involved in anti-oxidative activity or detoxification were affected in a gender-specific manner. These findings, particularly the effects on the endocrine system of fish, will advance our understanding of the ecotoxicity of β-agonists.


Similar content being viewed by others
References
Bartelt-Hunt S, Snow DD, Damon-Powell T et al (2011) Occurrence of steroid hormones and antibiotics in shallow groundwater impacted by livestock waste control facilities. J Contam Hydrol 123:94–103
Beggel S, Werner I, Connon RE et al (2012) Impacts of the phenylpyrazole insecticide fipronil on larval fish: time-series gene transcription responses in fathead minnow (Pimephales promelas) following short-term exposure. Sci Total Environ 426:160–165
Elanco Animal Health (1998) Environmental assessement for the use of ractopamine hydrochloride premix in the feed of cattle. http://www.fda.gov/downloads/AnimalVeterinary/DevelopmentApprovalProcess/EnvironmentalAssessments/UCM303565.pdf. Accessed 29 Jan 2015
General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China (2006) Determination of ractopamine in animal feeds – high performance liquid chromatographic method (GBIT 20189-2006), Beijing
Haji-Abadi SMAJ, Soofiani NM, Sadeghi AA et al (2010) Effects of supplemental dietary l-carnitine and ractopamine on the performance of juvenile rainbow trout, Oncorhynchus mykiss. Aquac Res 41:1582–1591
Li C, Wu YL, Yang T et al (2010) Simultaneous determination of clenbuterol, salbutamol and ractopamine in milk by reversed-phase liquid chromatography tandem mass spectrometry with isotope dilution. J Chromatogr A 1217:7873–7877
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 25:402–408
Lortie MB, Arnason T, Dugan SG et al (2004) The impact of feeding β-adrenergic agonists on rainbow trout muscle β-adrenoceptors and protein synthesis. J Fish Biol 65:769–787
Massarsky A, Trudeau VL, Moon TW (2011) β-Blockers as endocrine disruptors: the potential effects of human β-blockers on aquatic organisms. J Exp Zool A 315:251–265
Moccia RD, Gurure RM, Atkinson JL et al (1998) Effects of the repartitioning agent ractopamine on the growth and body composition of rainbow trout, Oncorhynchus mykiss (Walbaum), fed three levels of dietary protein. Aquac Res 29:687–694
Mustin WT, Lovell RT (1992) Feeding the repartitioning agent, ractopamine, to channel catfish (Ictalurus punctatus) increases weight-gain and reduces fat deposition. Aquaculture 109:145–152
Owen SF, Giltrow E, Huggett DB et al (2007) Comparative physiology, pharmacology and toxicology of β-blockers: mammals versus fish. Aquat Toxicol 82:145–162
Patil JG, Hinze SJ (2008) Simplex PCR assay for positive identification of genetic sex in the Japanese medaka, Oryzias latipes. Mar Biotechnol 10:641–644
Ricks CA, Dalrymple RH, Baker PK et al (1984) Use of a β-agonist to alter fat and muscle deposition in steers. J Animal Sci 59:1247–1255
Sanderson JT (2006) The steroid hormone biosynthesis pathway as a target for endocrine-disrupting chemicals. Toxicol Sci 94:3–21
Satpathy BB, Mukherjee D, Ray AK (2001) Effects of dietary inclusion of the β-adrenergic agonist, salbutamol, on growth performance and whole-body composition of rohu Labeo rohita (Hamilton) fingerlings fed diets containing two protein levels. Aquac Res 32:739–747
Shao B, Chen D, Zhang J et al (2009) Determination of 76 pharmaceutical drugs by liquid chromatography–tandem mass spectrometry in slaughterhouse wastewater. J Chromatogr A 1216:8312–8318
Sun LW, Shao XL, Chi J et al (2011) Transcriptional responses in the brain, liver and gonad of Japanese ricefish (Oryzias latipes) exposed to two anti-estrogens. Comp Biochem Physiol C 153:392–401
Valavanidis A, Vlahogianni T, Dassenakis M et al (2006) Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol Environ Saf 64:178–189
Van de Harr MJ (2005) Regulation of nutrient partitioning and overview of the hormones that are affected by nutrition or that directly alter metabolism. In: Pond WG, Church DC, Pond KR, Schoknecht PA (eds) Basic animal nutrition and feeding. Wiley, Hoboken, pp 275–289
Vandenberg GW, Moccia RD (1998) Growth performance and carcass composition of rainbow trout, Oncorhynchus mykiss (Walbaum), fed the β-agonist ractopamine. Aquac Res 29:469–479
Vandenberg GW, Leatherland JF, Moccia RD (1998) The effects of the β-agonist ractopamine on growth hormone and intermediary metabolite concentrations in rainbow trout, Oncorhynchus mykiss (Walbaum). Aquac Res 29:79–87
Wu MH, Shariat-Madar B, Haron MH et al (2011) Ethanol-induced attenuation of oxidative stress is unable to alter mRNA expression pattern of catalase, glutathione reductase, glutathione-S-transferase (GST1A), and superoxide dismutase (SOD3) enzymes in Japanese rice fish (Oryzias latipes) embryogenesis. Comp Biochem Physiol C 153:159–167
Yamamoto T (1953) Artificially induced sex-reversal in genotypic males of the medaka (Oryzias latipes). J Exp Zool 123:571–594
Yu TH, Lin AYC, Wang XH et al (2011) Occurrence of β-blockers and β-agonists in hospital effluents and their receiving rivers in southern Taiwan. Desalin Water Treat 32:49–56
Zhang X, Hecker M, Park JW et al (2008) Real-time PCR array to study effects of chemicals on the hypothalamic–pituitary–gonadal axis of the Japanese medaka. Aquat Toxicol 88:173–182
Acknowledgments
We gratefully acknowledge the National Natural Science Foundation of China (No. 21377118) and Program for Changjiang Scholars and Innovative Research Team in University (IRT13096) for supporting this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, L., Wang, S., Lin, X. et al. Early Life Exposure to Ractopamine Causes Endocrine-Disrupting Effects in Japanese Medaka (Oryzias latipes). Bull Environ Contam Toxicol 96, 150–155 (2016). https://doi.org/10.1007/s00128-015-1659-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1659-5