Abstract
Key message
Protein content of soybean is critical for utility of soybean meal. A fast-neutron-induced deletion on chromosome 12 was found to be associated with increased protein content.
Abstract
Soybean seed composition affects the utility of soybean, and improving seed composition is an essential breeding goal. Fast neutron radiation introduces genomic mutations resulting in novel variation for traits of interest. Two elite soybean lines were irradiated with fast neutrons and screened for altered seed composition. Twenty-three lines with altered protein, oil, or sucrose content were selected based on near-infrared spectroscopy data from five environments and yield tested at five locations. Mutants with significantly increased protein averaged 19.1–36.8 g kg−1 more protein than the parents across 10 environments. Comparative genomic hybridization (CGH) identified putative mutations in a mutant, G15FN-12, that has 36.8 g kg−1 higher protein than the parent genotype, and whole genome sequencing (WGS) of the mutant has confirmed these mutations. An F2:3 population was developed from G15FN-12 to determine association between genomic changes and increased protein content. Bulked segregant analysis of the population using the SoySNP50K BeadChip identified a CGH- and WGS-confirmed deletion on chromosome 12 to be responsible for elevated protein content. The population was genotyped using a KASP marker designed at the mutation region, and significant association (P < 0.0001) between the deletion on chromosome 12 and elevated protein content was observed and confirmed in the F3:4 generation. The F2 segregants homozygous for the deletion averaged 27 g kg−1 higher seed protein and 8 g kg−1 lower oil than homozygous wild-type segregants. Mutants with altered seed composition are a new resource for gene function studies and provide elite materials for genetic improvement of seed composition.




Similar content being viewed by others
Abbreviations
- BAM:
-
Binary alignment map
- BLAST:
-
Basic local alignment search tool
- BLUP:
-
Best linear unbiased predictor
- BSA:
-
Bulked segregant analysis
- BWA:
-
Burrows-Wheeler aligner
- CGH:
-
Comparative genomic hybridization
- CNV:
-
Copy number variation
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- FN:
-
Fast neutron
- GWAS:
-
Genome-wide association study
- IGV:
-
Integrative Genomics Viewer
- KASP:
-
Kompetitive allele specific PCR
- MG:
-
Maturity Group
- NIL:
-
Near-isogenic line
- NIR:
-
Near-infrared
- PCR:
-
Polymerase chain reaction
- PI:
-
Plant introduction
- QTL:
-
Quantitative trait locus
- RFO:
-
Raffinose family of oligosaccharides
- RIL:
-
Recombinant inbred line
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- SAM:
-
Sequence alignment/map format
- SNP:
-
Single nucleotide polymorphism
- WGS:
-
Whole genome sequencing
References
Anderson JE, Michno J-M, Kono TJY, Stec AO, Campbell BW, Curtin SJ, Stupar RM (2016) Genomic variation and DNA repair associated with soybean transgenesis: a comparison to cultivars and mutagenized plants. BMC Biotechnol 16:41
Andrews S (2014) FastQC a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/
Bandillo N, Jarquin D, Song Q, Nelson R, Cregan P, Specht J, Lorenz A (2015) A population structure and genome-wide association analysis on the usda soybean germplasm collection. Plant Genome 8
Barthelson R (2014a) HTProcess_trimmomatic_0.32. rogerab. CyVerse https://de.cyverse.org/de/?type=apps&app-id=93820714-89b7-4008-a9d1-98cb35ae3b09&system-id=de
Barthelson R (2014b) HTProcess-prepare_directories-and-run_fastqc-0.1. rogerab. CyVerse. https://de.cyverse.org/de/?type=apps&app-id=b4d15ab5-81b6-47e1-8637-26824e1f1679&system-id=de
Barthelson R (2016) SAM-to-sorted-BAM. rogerab. CyVerse. https://pods.iplantcollaborative.org/wiki/display/DEapps/Convert+SAM-to-sorted-BAM
Belfield E, Gan X, Mithani A, Brown C, Jiang C, Franklin K, Alvey E, Wibowo A, Jung M, Bailey K, Kalwani S, Ragoussis J, Mott R, Harberd N (2012) Genome-wide analysis of mutations in mutant lineages selected following fast-neutron irradiation mutagenesis of Arabidopsis thaliana. Genome Res 22:1306–1315
Boerma HR, Hussey RS, Phillips DV, Wood ED, Rowan GB, Finnerty SL (1997) Registration of ‘Benning’ soybean. Crop Sci 37:1982
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinform 30(15):2114–2120
Bolon Y-T, Haun WJ, Xu WW, Grant D, Stacey MG, Nelson RT, Gerhardt DJ, Jeddeloh JA, Stacey G, Muehlbauer GJ, Orf JH, Naeve SL, Stupar RM, Vance CP (2011) Phenotypic and genomic analyses of a fast neutron mutant population resource in soybean. Plant Physiol 156:240–253
Bolon Y-T, Stec AO, Michno J-M, Roessler J, Bhaskar PB, Ries L, Dobbels AA, Campbell BW, Young NP, Anderson JE, Grant DM, Orf JH, Naeve SL, Muehlbauer GJ, Vance CP, Stupar RM (2014) Genome resilience and prevalence of segmental duplications following fast neutron irradiation of soybean. Genetics 198:967–981
Brummer EC, Graef GL, Orf J, Wilcox JR, Shoemaker RC (1997) Mapping QTL for seed protein and oil content in eight soybean populations. Crop Sci 37:370–378
Brzostowski LF, Diers BW (2017) Agronomic evaluation of a high protein allele from PI407788A on chromosome 15 across two soybean backgrounds. Crop Sci 57:2972–2978
Brzostowski LF, Pruski T, Specht JE, Diers BW (2017) Impact of seed protein alleles from three soybean sources on seed composition and agronomic traits. Theor Appl Genet 130:2315–2326
Burton JW (1987) Quantitative genetics: results relevant to soybean breeding. In: Wilcox JR (ed) Soybeans: improvement, production and uses, 2nd edn. ASA, CSSA, and SSSA, Madison
Campbell BW, Hofstad AN, Sreekanta S, Fu F, Kono TJY, O’Rourke JA, Vance CP, Muehlbauer GJ, Stupar RM (2016) Fast neutron-induced structural rearrangements at a soybean NAP1 locus result in gnarled trichomes. Theor Appl Genet 129:1725–1738
Chung J, Babka HL, Graef GL, Staswick PE, Lee DJ (2003) The seed protein, oil, and yield QTL on soybean linkage group I. Crop Sci 43:1053–1067
Cober ER, Voldeng HD (2000) Developing high-protein, high-yield soybean populations and lines. Crop Sci 40:39–42
Cooksey A (2017) BWA mem_longreads-0.7.15. Upendra Devisetty. CyVerse. https://de.cyverse.org/de/?type=apps&app-id=e813971a-7d2d-11e7-9247-008cfa5ae621&system-id=de
DeBolt S (2010) Copy number variation shapes genome diversity in Arabidopsis over immediate family generational scales. Genome Biol Evol 2:441–453
Devisetty UK (2016a) BWA mem 0.7.15. Upendra Devisetty. CyVerse. https://de.cyverse.org/de/?type=apps&app-id=4c7e942e-6408-11e6-a1f5-008cfa5ae621&system-id=de
Devisetty UK (2016b) SAM_to_Sorted_BAM-0.1.19 (app for workflows). Upendra Kumar Devisetty. CyVerse. https://de.cyverse.org/de/?type=apps&app-id=9b837c54-122f-11e6-98c7-dfa2ad9d9ddd&system-id=de
Diers BW, Keim P, Fehr WR, Shoemaker RC (1992) RFLP analysis of soybean seed protein and oil content. Theor Appl Genet 83:608–612
Dobbels AA, Michno J-M, Campbell BW, Virdi KS, Stec AO, Muehlbauer GJ, Naeve SL, Stupar RM (2017) An induced chromosomal translocation in soybean disrupts a KASI ortholog and is associated with a high-sucrose and low-oil seed phenotype. G3 Genes Genomes Genet 7:1215–1223
Eskandari M, Cober E, Rajcan I (2013) Genetic control of soybean seed oil: II QTL and genes that increase oil concentration without decreasing protein or with increased seed yield. Theor Appl Genet 126(6):1677–1687
Fehr WR, Caviness CE (1977) Stages of soybean development. Special Report. 87. https://lib.dr.iastate.edu/specialreports/87/
Glenn TC, Nilsen RA, Kieran TJ, Finger JW Jr, Pierson TW, Bentley KE, Hoffberg SL, Louha S, García-De León FJ, Del Rio Portilla MA, Reed KD, Anderson JL, Meece JK, Aggrey SE, Rekaya R, Alabady M, Bélanger M, Winker K, Faircloth BC (2016) Adapterama I: universal stubs and primers for thousands of dual-indexed Illumina libraries (iTru & iNext). BioRxiv. https://doi.org/10.1101/049114
Goodstein DM, Shu S, Howson R, Neupane R, Hayes RD, Fazo J et al (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40((Database issue)):D1178–D1186. https://doi.org/10.1093/nar/gkr944
Grant D, Nelson RT, Cannon SB, Shoemaker RC (2010) SoyBase, the USDA-ARS soybean genetics and genomics database. Nucleic Acids Res 38(suppl 1):D843–D846. https://doi.org/10.1093/nar/gkp798
Hartwig EE, Hinson K (1972) Association between chemical composition of seed and seed yield of soybeans. Crop Sci 12:829–830
Haun WJ, Hyten DL, Xu WW, Gerhardt DJ, Albert TJ, Richmond T, Jeddeloh JA, Jia G, Springer NM, Vance CP, Stupar RM (2011) The composition and origins of genomic variation among individuals of the soybean reference cultivar Williams 82. Plant Physiol 155:645–655
Herman EM (2014) Soybean seed proteome rebalancing. Front Plant Sci 5(437):1–8
Hwang E-Y, Qijian S, Jia G, Specht JE, Hyten DL, Costa J, Cregan PB (2014) A genome-wide association study of seed protein and oil content in soybean. BMC Genom 15:1–25
Hwang W, Kim M, Kang Y, Shim S, Stacey M, Stacey G, Lee S-H (2015) Genome-wide analysis of mutations in a dwarf soybean mutant induced by fast neutron bombardment. Euphytica 203:399–408
Hymowitz T, Collins FI, Walker WM, Panczner J (1972) Relationship between content of oil, protein, and sugar in soybean seed. Agron J 64:613
Jaureguy LM, Chen P, Scaboo AM (2011) Heritability and correlations among food-grade traits in soybean. Plant Breed 130:647–652
Kabelka EA, Diers BW, Fehr WR, LeRoy AR, Baianu IC, You T, Neece DJ, Nelson RL (2004) Putative alleles for increased yield from soybean plant introductions. Crop Sci 44(3):784–791
Keim P, Olson TC, Shoemaker RC (1988) A rapid protocol for isolating soybean DNA. Soybean Genet Newsl 18:150–152
Kim HK, Kang ST, Oh KW (2006) Mapping of putative quantitative trait loci controlling the total oligosaccharide and sucrose content of Glycine max seeds. J Plant Res 119:533–538
Kim M, Schultz S, Nelson RL, Diers BW (2016) Identification and fine mapping of a soybean seed protein QTL from PI 407788A on chromosome 15. Crop Sci 56(1):219–225. https://doi.org/10.2135/cropsci2015.06.0340
Kuznetsova A, Brockhoff PB, Christensen RHB (2016). lmerTest: tests in linear mixed effects models. R package version 2.0-33. https://CRAN.R-project.org/package=lmerTest
Lee SH, Bailey MA, Mian MAR, Carter TE Jr, Shipe ER, Ashley DA, Parrott WA, Hussey RS, Boerma HR (1996) RFLP loci associated with soybean seed protein and oil content across populations and locations. Theor Appl Genet 93(5–6):649–657
Li H (2011) A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinform 27(21):2987–2993
Li H (2013) Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. arXiv:1303.3997v2 (q-bio.GN)
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinform 25:1754–1760
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R, 1000 Genome Project Data Processing Subgroup (2009) The Sequence alignment/map (SAM) format and SAMtools. Bioinform 25:2078–2079
Liang H, Yu Y, Wang S, Lian Y, Wang T, Wei Y, Gong P, Liu X, Fang X, Zhang M (2010) QTL Mapping of isoflavone, oil and protein contents in soybean (Glycine max L. Merr.). Agric Sci China 9(8):1108–1116
Lu W, Wen Z, Li H, Yuan D, Li J, Zhang H, Huang Z, Cui S, Du W (2013) Identification of the quantitative trait loci (QTL) underlying water soluble protein content in soybean. Theor Appl Genet 126:425–433
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 17(1):10–12. https://doi.org/10.14806/ej.17.1.200
Mian R, McHale L, Li Z, Dorrance AE (2017) Registration of ‘HighPro1’ soybean with high protein and high yield developed from a north x south cross. J Plant Reg 11:51–54
Muñoz-Amatriaín M, Eichten SR, Wicker T, Richmond TA, Mascher M, Steuernagel B, Scholz U, Ariyadasa R, Spannagl M, Nussbaumer T, Mayer KF, Taudien S, Platzer M, Jeddeloh JA, Springer JM, Muehlbauer GJ, Stein N (2013) Distribution, functional impact, and origin mechanisms of copy number variation in the barley genome. Genome Biol 14(6):R58. https://doi.org/10.1186/gb-2013-14-6-r58
Nichols DM, Glover KD, Carlson SR, Specht JE, Diers BW (2006) Fine mapping of a seed protein QTL on soybean linkage group I and its correlated effects on agronomic traits. Crop Sci 46:834–839
Patil G, Mian R, Vuong T, Pantalone V, Song Q, Chen P, Shannon G, Carter TE, Nguyen H (2017) Molecular mapping and genomics of soybean seed protein: a review and perspective for the future. Theor Appl Genet 130:1975–1991
Pham AT, McNally K, Abdel-Haleem H, Boerma HR, Li Z (2013) Fine mapping and identification of candidate genes controlling the resistance to southern root-knot nematode in PI 96354. Theor Appl Genet 126(7):1825–1838
Phansak P, Soonsuwon W, Hyten DL, Song Q, Cregan PB, Graef GL, Specht JE (2016) Multi-population selective genotyping to identify soybean [Glycine max (L.) Merr.] seed protein and oil QTLs. G3 Genes Genomes Genet 6:1635–1648
Prenger EM, Mian R, Buckley B, Boerma HR, Li Z (2018). Introgression of a high protein allele into an elite soybean variety results in a high-protein near-isogenic line with yield parity. Chapter 3. MS thesis, University of Georgia
Qi Z, Wu Q, Han X, Sun Y-n, Du X-y, Liu C-y, Jiang H-w, Hu G-h, Chen Q-s (2011a) Soybean oil content QTL mapping and integrating with meta-analysis method for mining genes. Euphytica 179:499–514
Qi Z, Ya-nan S, Qiong W, Chun-yan L, Guo-hua H, Qing-shan C (2011b) A meta-analysis of seed protein concentration QTL in soybean. Can J Plant Sci 91:221–230
Qiu BX, Arelli PR, Sleper DA (1999) RFLP markers associated with soybean cyst nematode resistance and seed composition in a ‘Peking’ x ‘Essex’ population. Theor Appl Genet 98(3–4):356–364
R Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org/
Robinson JT, Thorvaldsdóttir H, Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nat Biotechnol 29:24–26
Schmutz et al (2010) Genome sequence of the palaeopolyploid soybean. Nat 463:178–183
Sebolt AM, Shoemaker RC, Diers BW (2000) Analysis of a quantitative trait locus allele from wild soybean that increases seed protein concentration in soybean. Crop Sci 40:1438–1444
Severin AJ, Woody JL, Bolon Y-T, Joseph B, Diers BW, Farmer AD, Muehlbauer GJ, Nelson RT, Grant D, Specht JE, Graham MA, Cannon SB, May GD, Vance CP, Shoemaker RC (2010) RNA-seq atlas of glycine max: a guide to the soybean transcriptome. BMC Plant Biol 10:160
Song Q, Hyten DL, Jia G, Quigley CV, Fickus EW, Nelson RL, Cregan PB (2013) Development and evaluation of SoySNP50 K, a high-density genotyping array for soybean. PLoS ONE 8:e54985
Song Q, Jenkins J, Jia G, Hyten DL, Pantalone V, Jackson SA, Schmutz J, Cregan PB (2016) Construction of high resolution genetic linkage maps to improve the soybean genome sequence assembly Glyma1.01. BMC Genom 17:33
Springer NM, Ying K, Fu Y, Ji T, Yeh C-T, Jia Y, Wu W, Richmond T, Kitzman J, Rosenbaum H, Iniguez AL, Barbazuk WB, Jeddeloh JA, Nettleton D, Schnable PS (2009) Maize inbreds exhibit high levels of copy number variation (CNV) and presence/absence variation (PAV) in genome content. PLoS Genet 5(11):e1000734. https://doi.org/10.1371/journal.pgen.1000734
Stacey MG, Cahoon RE, Nguyen HT, Cui Y, Sato S, Nguyen CT, Phoka N, Clark KM, Liang Y, Forrester J, Batek J, Do PT, Sleper DA, Clemente TE, Cahoon EB, Stacey G (2016) Identification of homogentisate dioxygenase as a target for vitamin E biofortification in oilseeds. Plant Physiol 172:1506
Teng W, Li W, Zhang Q, Wu D, Zhao X, Li H, Han Y, Li W (2017) Identification of quantitative trait loci underlying seed protein content of soybean including main, epistatic, and QTL × environment effects in different regions of Northeast China. Genome 60(8):649–655. https://doi.org/10.1139/gen-2016-0189
Thorne JC, Fehr WR (1970) Incorporation of high-protein, exotic germplasm into soybean populations by 2- and 3-way crosses. Crop Sci 10:652–655
Thorvaldsdóttir H, Robinson JT, Mesirov JP (2013) Integrative genomics viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 14:178–192
Valliyodan B, Shi H, Nguyen HT (2015) A simple analytical method for high-throughput screening of major sugars from soybean by normal-phase HPLC with evaporative light scattering detection. Chromatogr Res Int 15:8
van de Wiel CCM, Schaart JG, Lotz LAP, Smulders MJM (2017) New traits in crops produced by genome editing techniques based on deletions. Plant Biotechnol Rep 11:1–8. https://doi.org/10.1007/s11816-017-0425-z
Vaughn M (2012) Uncompress files with gunzip. CyVerse http://www.gnu.org/software/gzip/
Vaughn JN, Nelson RL, Song Q, Cregan PB, Li Z (2014) The genetic architecture of seed composition in soybean is refined by genome-wide association scans across multiple populations. G3 Genes Genomes Genet 4:2283–2294
Walls R (2018) Concatenate multiple files. BusyBox. CyVerse. https://de.cyverse.org/de/?type=apps&app-id=77830f32-084a-11e8-a871-008cfa5ae621&system-id=de
Warrington CV, Abdel-Haleem H, Hyten DL, Cregan PB, Orf JH, Killam AS, Bajjalieh N, Li Z, Boerma HR (2015) QTL for seed protein and amino acids in the Benning × Danbaekkong soybean population. Theor Appl Genet 128:839–850
Wilcox JR, Cavins JF (1995) Backcrossing high seed protein to a soybean cultivar. Crop Sci 35:1036–1041
Wilcox JR, Zhang GD (1997) Relationships between seed yield and seed protein in determinate and indeterminate soybean populations. Crop Sci 37:361–364
Wilson RF (2004) Seed composition. In: Boerma HR, Specht JE (eds) Soybean: improvement, production and uses, 3rd edn. ASA and CSSA, Madison, pp 671–677
Yin XH, Vyn TJ (2005) Relationships of isoflavone, oil, and protein in seed with yield of soybean. Agron J 97:1314–1321
Yu P, Wang C, Xu Q, Feng Y, Yuan X, Yu H, Wang Y, Tang S, Wei X (2011) Detection of copy number variations in rice using array-based comparative genomic hybridization. BMC Genom 12:372. https://doi.org/10.1186/1471-2164-12-372
Acknowledgements
We thank the United Soybean Board and the John Ingle Innovation in Plant Breeding Award for funding this research. Thanks to Dale Wood, Earl Baxter, Brice Wilson, Jeremy Nation, Greg Gokalp, Ricky Zoller, Tatyana Nienow, Troy Kieran, and Swarnali Louha from University of Georgia, and Adrian Stec and Jean-Michel Michno from University of Minnesota for their technical support. Thanks to Blair Buckley for growing the yield trial at Bossier, LA. Thanks to Drs. Henry Nguyen and Tri Vuong from University of Missouri for analyzing the sugar content.
Author information
Authors and Affiliations
Contributions
EMP conducted the experiment, analyzed the data, and drafted the manuscript; RMS and TG conducted comparative genomic hybridization and whole genome sequencing, respectively; MARM performed field tests; AO conducted seed composition analysis; and ZL designed and organized the experiment, interpreted the results, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Communicated by Albrecht E. Melchinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
122_2019_3399_MOESM2_ESM.tif
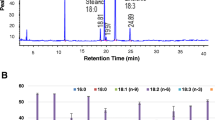
Fig. S1. Protein differences among genotypes in the F3 Benning × G15FN-12 populationProtein content is reported in g kg-1 on a dry matter basis. P<.0001. An “n” indicates the number of F3 families measured for each corresponding genotype. (TIFF 60353 kb)
Rights and permissions
About this article
Cite this article
Prenger, E.M., Ostezan, A., Mian, M.A.R. et al. Identification and characterization of a fast-neutron-induced mutant with elevated seed protein content in soybean. Theor Appl Genet 132, 2965–2983 (2019). https://doi.org/10.1007/s00122-019-03399-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-019-03399-w