Abstract
Purpose
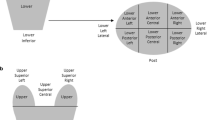
Using high-quality CT-on-rails imaging, the daily motion of the prostate bed clinical target volume (PB-CTV) based on consensus Radiation Therapy Oncology Group (RTOG) definitions (instead of surgical clips/fiducials) was studied. It was assessed whether PB motion in the superior portion of PB-CTV (SUP-CTV) differed from the inferior PB-CTV (INF-CTV).
Patients and methods
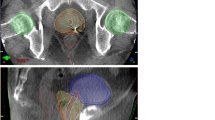
Eight pT2-3bN0-1M0 patients underwent postprostatectomy intensity-modulated radiotherapy, totaling 300 fractions. INF-CTV and SUP-CTV were defined as PB-CTV located inferior and superior to the superior border of the pubic symphysis, respectively. Daily pretreatment CT-on-rails images were compared to the planning CT in the left–right (LR), superoinferior (SI), and anteroposterior (AP) directions. Two parameters were defined: “total PB-CTV motion” represented total shifts from skin tattoos to RTOG-defined anatomic areas; “PB-CTV target motion” (performed for both SUP-CTV and INF-CTV) represented shifts from bone to RTOG-defined anatomic areas (i. e., subtracting shifts from skin tattoos to bone).
Results
Mean (± standard deviation, SD) total PB-CTV motion was −1.5 (± 6.0), 1.3 (± 4.5), and 3.7 (± 5.7) mm in LR, SI, and AP directions, respectively. Mean (± SD) PB-CTV target motion was 0.2 (±1.4), 0.3 (±2.4), and 0 (±3.1) mm in the LR, SI, and AP directions, respectively. Mean (± SD) INF-CTV target motion was 0.1 (± 2.8), 0.5 (± 2.2), and 0.2 (± 2.5) mm, and SUP-CTV target motion was 0.3 (± 1.8), 0.5 (± 2.3), and 0 (± 5.0) mm in LR, SI, and AP directions, respectively. No statistically significant differences between INF-CTV and SUP-CTV motion were present in any direction.
Conclusion
There are no statistically apparent motion differences between SUP-CTV and INF-CTV. Current uniform planning target volume (PTV) margins are adequate to cover both portions of the CTV.
Zusammenfassung
Ziel
Zur Evaluation der interfraktionellen Variabilität des klinischen Zielvolumens der Prostataloge (PB-CTV) anhand der Empfehlungen der Radiation Therapy Oncology Group (RTOG) wurden tägliche CT-„on-Rails“-Bildgebungen (anstelle von Clips/Markern) durchgeführt. Untersucht wurde, ob die Bewegung des oberen PB-CTV- (SUP-CTV) von der des unteren PB-CTV-Abschnitts (INF-CTV) abweicht.
Methoden
Acht Patienten (pT2–3bN0–1M0) erhielten nach Prostatektomie eine intensitätsmodulierte Strahlentherapie mit insgesamt 300 Fraktionen. INF-CTV und SUP-CTV wurden als PB-CTV unterhalb und oberhalb der Symphyse gewertet. Die Abweichungen auf den täglichen Lagekontrollen mittels CT-on-Rails wurden mit den initialen Bestrahlungsplanungs-CTs in folgenden Richtungen verglichen: Links-Rechts (LR), superior-inferior (SI) und anteroposterior (AP). Zwei Parameter wurden definiert: „total PB-CTV motion“, welche die Verschiebung der Hauttätowierungen, und „PB-CTV target motion“ (für SUP-CTV und INF-CTV), welche die Verschiebung der knöchernen Strukturen jeweils zu den durch die RTOG definierten anatomischen Strukturen darstellt.
Ergebnisse
Die durchschnittliche „total PB-CTV motion“ (± Standardabweichung) betrug jeweils in LR-, SI- und AP-Richtung −1,5 (± 6,0), 1,3 (± 4,5) und 3,7 (± 5,7) mm, die durchschnittliche „PB-CTV target motion“ 0,2 (± 1,4), 0,3 (± 2,4) und 0,0 (± 3,1) mm, die mittlere Bewegung des INF-CTV 0,1 (± 2,8), 0,5 (± 2,2), und 0,2 (± 2,5) mm und die Varianz des SUP-CTV 0,3 (± 1,8), 0,5 (± 2,3) und 0 (± 5,0) mm. Statistisch signifikante Unterschiede zwischen der INF-CTV- und SUP-CTV-Bewegung konnten in keiner Richtung festgestellt werden.
Schlussfolgerung
SUP-CTV und INF-CTV-Bewegung wiesen keinen signifikanten Unterschied auf. Die aktuellen uniformen Säume des Planungszielvolumens sind adäquat und ausreichend, um beide Anteile des CTV abzudecken.




Similar content being viewed by others
References
Underwood W 3rd, Jackson J, Wei JT et al (2005) Racial treatment trends in localized/regional prostate carcinoma: 1992–1999. Cancer 103:538–545
Parker C, Sydes MR, Catton C et al (2007) Radiotherapy and androgen deprivation in combination after local surgery (RADICALS): a new Medical Research Council/National Cancer Institute of Canada phase III trial of adjuvant treatment after radical prostatectomy. BJU Int 99:1376–1379
Thompson IM Jr, Tangen CM, Paradelo J et al (2006) Adjuvant radiotherapy for pathologically advanced prostate cancer: a randomized clinical trial. JAMA 296:2329–2335
Bolla M, van Poppel H, Collette L et al (2005) Postoperative radiotherapy after radical prostatectomy: a randomised controlled trial (EORTC trial 22911). Lancet 366:572–578
Zelefsky MJ, Aschkenasy E, Kelsen S et al (1997) Tolerance and early outcome results of postprostatectomy three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys 39:327–333
Teh BS, Mai WY, Augspurger ME et al (2001) Intensity modulated radiation therapy (IMRT) following prostatectomy: more favorable acute genitourinary toxicity profile compared to primary IMRT for prostate cancer. Int J Radiat Oncol Biol Phys 49:465–472
Wiltshire KL, Brock KK, Haider MA et al (2007) Anatomic boundaries of the clinical target volume (prostate bed) after radical prostatectomy. Int J Radiat Oncol Biol Phys 69:1090–1099
Michalski JM, Lawton C, El Naqa I et al (2010) Development of RTOG consensus guidelines for the definition of the clinical target volume for postoperative conformal radiation therapy for prostate cancer. Int J Radiat Oncol Biol Phys 76:361–368
Ten Haken RK, Forman JD, Heimburger DK et al (1991) Treatment planning issues related to prostate movement in response to differential filling of the rectum and bladder. Int J Radiat Oncol Biol Phys 20:1317–1324
van Herk M, Bruce A, Kroes AP et al (1995) Quantification of organ motion during conformal radiotherapy of the prostate by three dimensional image registration. Int J Radiat Oncol Biol Phys 33:1311–1320
Zelefsky MJ, Happersett L, Leibel SA et al (1997) The effect of treatment positioning on normal tissue dose in patients with prostate cancer treated with three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys 37:13–19
Balter JM, Sandler HM, Lam K et al (1995) Measurement of prostate movement over the course of routine radiotherapy using implanted markers. Int J Radiat Oncol Biol Phys 31:113–118
Crook JM, Raymond Y, Salhani D et al (1995) Prostate motion during standard radiotherapy as assessed by fiducial markers. Radiother Oncol 37:35–42
Litzenberg D, Dawson LA, Sandler H et al (2002) Daily prostate targeting using implanted radiopaque markers. Int J Radiat Oncol Biol Phys 52:699–703
Nederveen AJ, van der Heide UA, Dehnad H et al (2002) Measurements and clinical consequences of prostate motion during a radiotherapy fraction. Int J Radiat Oncol Biol Phys 53:206–214
Garsa AA, Verma V, Michalski JM, Gay HA (2014) Transperineal ultrasound-guided implantation of electromagnetic transponders in the prostatic fossa for localization and tracking during external beam radiation therapy. Pract Radiat Oncol 4:415–421
Schiffner DC, Gottschalk AR, Lometti M et al (2007) Daily electronic portal imaging of implanted gold seed fiducials in patients undergoing radiotherapy after radical prostatectomy. Int J Radiat Oncol Biol Phys 67:610–619
Canter D, Greenberg RE, Horwitz EM et al (2010) Implantation of electromagnetic transponders following radical prostatectomy for delivery of IMRT. Can J Urol 17:5375–5369
Fortin I, Carrier JF, Beauchemin MC, Beliveau-Nadeau D, Delouya G, Taussky D (2014) Using fiducial markers in the prostate bed in postprostatectomy external beam radiation therapy improves accuracy over surgical clips. Strahlenther Onkol 190:467–471
Verma V, Chen L, Michalski JM, et al (2015) Evaluation of 3 T pelvic MRI imaging in prostate cancer patients receiving post-prostatectomy IMRT. World J Urol 33:69–75
Klayton T, Price R, Buyyounouski MK et al (2012) Prostate bed motion during intensity-modulated radiotherapy treatment. Int J Radiat Oncol Biol Phys 84(1):130–136
Kupelian PA, Langen KM, Willoughby TR et al (2006) Daily variations in the position of the prostate bed in patients with prostate cancer receiving postoperative external beam radiation therapy. Int J Radiat Oncol Biol Phys 66:593–596
Kruse JJ, Herman MG, Hagness BJ, Davis TM, Pisansky TM (2001) Daily monitoring of patient setup variation and prostatic fossa tissue motion with an electronic portal imaging device. Int J Radiat Oncol Biol Phys 31:S387
van Herk M, Bruce A, Kroes AP, Shouman T, Touw A, Lebesque JV (1995) Quantification of organ motion during conformal radiotherapy of the prostate by three-dimensional image registration. Int J Radiat Oncol Biol Phys 33:1311–1320
Wang KK, Vapiwala N, Deville C (2012) A study to quantify the effectiveness of daily endorectal balloon for prostate intrafraction motion management. Int J Radiat Oncol Biol Phys 83(3):1055–1063
Huang K, Palma DA, Scott D et al (2012) Inter- and intra-fractional uncertainty in prostate bed image-guided radiotherapy. Int J Radiat Oncol Biol Phys 84(2):402–407
Bell LJ, Cox J, Eade T, Rinks M, Herschtal A, Kneebone A (2015) Determining optimal planning target volume and image guidance policy for post-prostatectomy intensity modulated radiotherapy. Radiat Oncol 10:151
Rudat V, Nour A, Hammoud M, Alaradi A, Mohammed A (2016) Image-guided intensity-modulated radiotherapy of prostate cancer: analysis of interfractional errors and acute toxicity. Strahlenther Onkol 192:109–117
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
V. Verma, S. Chen, S. Zhou, C.A. Enke, and A.O. Wahl declare that they have no competing interests.
This study was approved by the University of Nebraska Medical Center Institutional Review Board and Ethics Committee, and complies with the Helsinki Declaration. All patients’ rights and identifying information were kept confidential. There are no identifiable persons in the manuscript.
Additional information
This study was presented at the 2010 meeting of the American Society for Radiation Oncology.
Rights and permissions
About this article
Cite this article
Verma, V., Chen, S., Zhou, S. et al. Prostate bed target interfractional motion using RTOG consensus definitions and daily CT on rails. Strahlenther Onkol 193, 38–45 (2017). https://doi.org/10.1007/s00066-016-1077-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-016-1077-6