Abstract
Although the advent of drug-eluting stents has reduced the rates of target vessel revascularization, there are observations of ongoing stent failure occurring very late after stent implantation and presenting as very late restenosis or as very late stent thrombosis. The de novo development of atherosclerosis within the neointimal region, called neoatherosclerosis, has been identified as one of the pathomechanisms of these observed late stent failures. The mechanisms of neoatherosclerosis development and its association with stent failure are currently the subject of intensive research. Optical coherence tomography (OCT) is an invasive imaging modality that allows us to visualize the micromorphology of coronary arteries with near-histological resolution, thus providing detailed assessment of the morphological characteristics of the neointima after stent implantation, including neoatherosclerosis. Several OCT studies have tried to provide in vivo insights in the mechanisms of neoatherosclerosis development and its association with late stent failure. This review summarizes the current insights into neoatherosclerosis obtained with OCT and discusses the association of neoatherosclerosis with late stent failure.
Zusammenfassung
Zwar ist es durch das Aufkommen medikamentenbeschichteter Stents zu einer Senkung der Rate von Zielgefäßrevaskularisierungen gekommen, aber es wurden Fälle fortlaufenden Stentversagens beobachtet, die erst sehr spät nach Stentimplantation auftreten und sich mit großer zeitlicher Verzögerung als Restenosierung oder als sehr späte Stentthrombose manifestieren. Das De-novo-Auftreten einer Atherosklerose innerhalb der neointimalen Region, als Neoatherosklerose bezeichnet, hat sich als einer der Pathomechanismen dieser späten Fälle von Stentversagen herausgestellt. Die Mechanismen der Neoatheroskleroseentstehung und ihr Zusammenhang mit einem Stentversagen werden derzeit noch intensiv untersucht. Die optische Kohärenztomographie (OCT) ist ein invasives bildgebendes Verfahren, mit dem sich die Mikromorphologie der Koronararterien in nahezu histologischer Auflösung darstellen lässt und das somit eine detaillierte Beurteilung der morphologischen Merkmale der Neointima nach Stentimplantation, einschließlich Neoatherosklerose, ermöglicht. Im Rahmen verschiedener OCT-Studien wurde versucht, in vivo einen Einblick in die Mechanismen der Neoatheroskleroseentstehung und ihren Zusammenhang mit spätem Stentversagen zu erhalten. In der vorliegenden Übersichtsarbeit wurde der Schwerpunkt auf die zusammenfassende Darstellung aktueller Erkenntnisse aus der OCT in Bezug auf die Neoatherosklerose gelegt und der Zusammenhang der Neoatherosklerose mit spätem Stentversagen erörtert.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Drug-eluting stents (DES) have reduced the rates of target vessel revascularization and comprise the mainstay of treatment in percutaneous coronary intervention. However, despite the excellent short- and mid-term results with the current DES generation, ongoing stent failure with both bare metal stents (BMS) and DES is a frequent finding very late after stent implantation [1–3]. Very late stent failure clinically manifests as very late restenosis or stent thrombosis. Although the pathogenesis of (very) late stent failure appears to be multifactorial [4, 5], one of the major mechanisms that have been implicated is the de novo development of atherosclerosis within the neointimal region, called neoatherosclerosis [6, 7]. Observations of neoatherosclerosis have been documented both in ex vivo pathological observations and in vivo by intravascular imaging.
Among several imaging modalities that have been used to identify neoatherosclerosis [8–11], optical coherence tomography (OCT) can provide the most comprehensive assessment of the neointimal tissue. OCT allows for the visualization of the micromorphology of coronary arteries with near-histological resolution, differentiating between individual plaque components and providing important quantitative plaque information as the thickness of the fibrous plaque [12, 13]. Thus, by OCT it is possible to assess distinct morphological characteristics of neoatherosclerosis, such as macrophage infiltration, lipid accumulation, in-stent calcification, or neointimal rupture. Consequently, in vivo OCT studies have focused on studying in-stent neoatherosclerosis and its association with late stent failure. This review summarizes the current insights into neoatherosclerosis obtained with OCT and discusses the implications of neoatherosclerosis on long-term outcome after stent implantation.
Definition and OCT imaging evidence of in-stent neoatherosclerosis
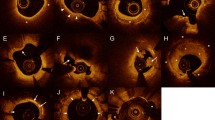
Neoatherosclerosis refers to an atherosclerotic change in neointimal tissue, first described in pathologic specimens of BMS, and more recently in pathologic specimens of DES as well [6, 7]. Although observations of neoatherosclerosis had been sporadically documented from the early application of OCT [14, 15], the tissue properties of the observed neointimal tissue patterns were unknown. Pathological studies were the first to provide more comprehensive insights regarding the histological composition of this entity. These studies defined neoatherosclerosis as the presence of clusters of lipid-laden foamy macrophages with or without necrotic core formation and/or calcification within the neointimal tissue of stented segments [6]. The presence of morphological components of native atherosclerosis within the neointimal tissue implies that these features can be identified by OCT, which has a high accuracy for detection of these characteristics in native atherosclerotic plaques. This hypothesis has been corroborated by a pathologic study of ex vivo stented coronary arteries, imaged by OCT, which showed that the OCT appearance of these characteristics is similar to that in native atherosclerosis [16]. Components of neoatherosclerosis that can be visualized by OCT include macrophage infiltration, necrotic core, in-stent calcifications, and neoatherosclerotic plaque rupture [17]. An in-stent necrotic core visualized by OCT is defined as the presence of signal-poor, highly attenuating regions with poorly delineated borders within the neointima, while in-stent calcifications are defined as well-delineated, signal-poor regions with sharp borders [16]. In accordance with native atherosclerosis, macrophages on OCT appear as a thin signal-bright layer causing high attenuation, while neointimal plaque rupture appears as a discontinuation in the luminal surface with formation of a cavity burrowing into the neointima [16]. Representative OCT images of in-stent neoatherosclerosis are shown in Fig. 1.
Morphological characteristics of in-stent neoatherosclerosis on optical coherence tomography images. a In-stent necrotic core (yellow) within the neointima (green). b In-stent calcification (white) and macrophage infiltration (orange). c Neointimal rupture. d In-stent necrotic core (yellow) with thin overlying fibrous cap. Panels on the right are color-coded cartoons of the corresponding OCT images, explaining the composition of the neointimal tissue.
At this point it is important to emphasize the difference of neoatherosclerosis with the previously reported heterogeneous and layered patterns of coverage [18]. These patterns are respectively defined as neointimal tissue with focally changing optical properties and varying backscattering patterns, and as neointimal tissue with concentric layers having different optical properties (an adluminal high scattering layer and an abluminal low scattering layer). Such patterns are often encountered in the context of in-stent restenosis, and although the pathological substrate is not well characterized, histological findings from atherectomy specimens, autopsy, and animal experiments include organized thrombus, fibrin, and myxomatous extracellular matrix with sporadic evidence of inflammation [16, 19–21]. The association of these patterns with neoatherosclerosis is yet unknown, however there is evidence that these patterns similar to the homogeneous pattern may subsequently be replaced by neoatherosclerotic tissue [22, 23]. Representative OCT images of different neointimal coverage patterns are shown in Fig. 2.
Patterns of neointimal coverage as visualized on optical coherence tomography images. a Homogeneous pattern (green); b heterogeneous pattern (light gray); c layered pattern (dark gray). Panels on the bottom right are color-coded cartoons of the corresponding OCT images, explaining the composition of the neointimal tissue.
Although OCT can discern the distinct morphological characteristics of neoatherosclerosis, caution should be applied to image interpretation. This is because macrophage infiltration can sometimes appear as in-stent necrotic core, and vice versa [24]. Also, the distinction of a layered pattern of coverage with in-stent necrotic core can also be challenging sometimes, although the low attenuation observed in the case of the layered pattern can be used to discriminate between these two entities [16]. Finally, with OCT it is not possible to distinguish between true adluminal atherosclerosis from the progression of an abluminal underlying necrotic core into the neointima. Nevertheless, the latter mechanism appears to be less prevalent [25], while it is unclear what the clinical significance of such a discrimination would be.
In vivo prevalence of neoatherosclerosis in BMS and DES with OCT
Several studies have tried to investigate the prevalence of neoatherosclerosis late after BMS or DES implantation using OCT (Table 1). The reported prevalence varies highly between the studies. As these studies differ significantly in stent type, in the interval between stent implantation and follow-up, and in the clinical presentation, this difference in prevalence appears to reflect differences in the composition of the studied population. The first reports of in-stent neoatherosclerosis report a prevalence of 67 % in patients with BMS implantation beyond 5 years [15]. Subsequent studies have shown lower prevalence ranging between 30 and 50 % in long-term follow-up of asymptomatic patients with BMS [26–28]; however, this incidence could be as high as 100 % in BMS restenosis more than 10 years since implantation [29]. With regard to neoatherosclerosis within DES, a prevalence of more than 50 % has been reported in a very late follow-up [27, 28], which can be even higher in stents with failure [14]. These rates tend to generally be in accordance with rates reported by pathological studies, which show a prevalence of 13–65 % depending on stent type and stent age [25].
Major factors associated with in-stent neoatherosclerosis
Stent type and age
Pathological studies were the first to indicate a potential association of stent type and stent age with the prevalence of neoatherosclerosis [6]. DES appear to have a higher prevalence of neoatherosclerosis compared with BMS. Moreover, irrespective of stent type, the interval from implantation appears to be strongly associated with neoatherosclerosis development, an interval which is much lower than the interval required for development of native atherosclerotic plaques [25]. Nakazawa et al. [6] first demonstrated an increased prevalence of neoatherosclerosis in autopsy specimens of first-generation DES compared with BMS despite a shorter interval from implantation. Importantly, in both groups, the prevalence of neoatherosclerosis increased with the interval since implantation [6]. In vivo OCT studies have been in line with pathological studies showing an increased prevalence of neoatherosclerosis in DES at a follow-up of up to 4 years, although at a very long-term follow-up the prevalence is similarly high for both stent types [30]. Furthermore, serial imaging observations in first- and second-generation DES demonstrate that neointimas with a homogeneous or heterogeneous pattern of coverage might develop neoatherosclerosis over time with the prevalence of neoatherosclerosis increasing from 15 % at 9 months to 28 % at 24 months since implantation [22]. This was further corroborated by another serial imaging study in first-generation DES where the incidence of neoatherosclerosis increased from 3 % at mid-phase observations (3–12 months after stent implantation) to 23 % at late-phase observations [23].
Although DES in general appear to have a higher prevalence of neoatherosclerosis than BMS, it is not clear whether the prevalence of neoatherosclerosis differs between first- and second-generation DES. Second-generation DES have improved biocompatibility due to thinner struts and more biocompatible or bioabsorbable polymers, which translates to lower vascular toxicity and hypersensitivity reactions [31, 32]. However, a human autopsy study did not demonstrate any significant difference in the prevalence of neoatherosclerosis between a second-generation DES, cobalt-chromium everolimus-eluting stent (CoCr-EES), and first-generation paclitaxel-eluting (PES) or sirolimus-eluting stent (SES): CoCr-EES = 29 %; SES = 35 %; PES = 19 % [32]. Conversely, an in vivo OCT study in 212 DES-treated patients with > 50 % stenosis (101 patients had a first-generation and 111 patients had a second-generation DES) demonstrated a significantly lower prevalence of neoatherosclerosis in the second-generation DES group (10.8 vs. 45.5 %, p < 0.001); however, this difference was not significant after adjusting for time since implantation [33]. Similar findings were observed with OCT in a comparison of the coronary arterial response in biodegradable polymer biolimus-eluting stents (BES) versus durable polymer SES and BMS at the 5-year follow-up. The number of cross sections with neoatherosclerosis within BES was similar to BMS (BES = 2.26 % vs. BMS = 2.23 %, p = 0.98) and tended to be lower than SES (BES = 2.26 % vs. SES = 9.90 %, p = 0.07) [34].
The reasons why the temporal course of atherosclerosis within stents is accelerated are not well understood; however, dysfunction of endothelial cells, which are barriers that prevent lipid infiltration and migration of inflammatory cells, has been considered to be the main mechanism. This mechanism can also explain the increased prevalence of neoatherosclerosis observed in DES, where local drug delivery inhibits neointimal hyperplasia, causing delayed coverage and dysfunction of endothelial cells [35].
Clinical factors
In addition to the stent type and age, several patient characteristics have been associated with neoatherosclerosis. Two registries of late stent follow-up have both identified chronic kidney disease as a factor independently associated with neoatherosclerosis [28, 33]. Other identified factors included low-density lipoprotein cholesterol > 70 mg/dl [33], current smoking, and lack of treatment with angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers [28]. In another study, the presence of diabetes mellitus was associated with a higher incidence of neoatherosclerosis at a late DES follow-up (18.3 vs. 5.5 %, p = 0.03) [36]. Further subgroup analysis showed that elevated glycated hemoglobin (HbA1c) levels (> 7 %) in patients with diabetes mellitus were associated with a higher incidence of neoatherosclerosis (28 vs. 7 %, p = 0.048).
Plaque and stent characteristics
Plaque characteristics have also been implicated in the neoatherosclerotic process. Microvessels play a key role in advanced atherosclerosis, which is closely associated with plaque hemorrhage and plaque rupture [37], while previous investigations have demonstrated a higher incidence of microvessels in late in-stent restenotic tissue, suggesting that neovessels might be a trigger for in-stent neoatherosclerosis [38, 39]. Moreover, a spatial correspondence of neoatherosclerosis and microvessels was identified at a follow-up OCT examination of BMS and DES [40], supporting the hypothesis of microvessel involvement in neoatherosclerosis development. In the same study, neoatherosclerosis was more frequently identified in the proximal and distal stent sections, also associated with the morphology of the adjacent vessel segment. Specifically, the presence of lipid plaque in the adjacent stent edges was associated with the presence of neoatherosclerosis at the stent edges, implying that progression of native disease might also contribute to neoatherosclerosis. Apart from plaque characteristics, stent characteristics such as strut thickness seem to play a role even among the same stent type. In a study assessing the prevalence of neoatherosclerosis in BMS at a long-term follow-up (≥ 4 years after implantation), thick-strut stents (≥ 100 μm) had a higher prevalence of neoatherosclerosis compared with thin-strut stents (70 vs. 32 %, p = 0.02) [41].
Neoatherosclerosis and in-stent restenosis
Although early in-stent restenosis mainly results from aggressive neointimal proliferation [42], recent data also suggest that neoatherosclerosis may play an important pathophysiological role, especially in late restenosis. Pathological studies have demonstrated that neoatherosclerosis represents a common substrate in patients with late stent failure [43], while in vivo OCT studies have further elucidated the role of neoatherosclerosis in the development of late stent failure (Fig. 3). The presence of neoatherosclerosis has been associated with a higher degree of neointimal hyperplasia, independent from stent type and time since implantation [44]. Although during the early phases after DES implantation, homogeneous or heterogeneous patterns of coverage are the predominant pathology in in-stent restenosis, in later follow-up intervals restenosis is often associated with the presence of neoatherosclerosis [38, 45]. A study focusing on 50 patients with DES restenosis at a median follow-up of 32 months since implantation showed a prevalence of neoatherosclerosis of 52 % [9]. Also, in lesions with very late BMS restenosis beyond 10 years since implantation, neoatherosclerosis was detected in 100 % of the stents with a high frequency of neointimal rupture and neointimal thrombi [29]. However, a comparison of restenotic lesions of DES and BMS showed that neoatherosclerosis occurs earlier in DES compared with BMS and develops more diffusely along stented vessels with thinner cap and greater total lipid core [8]. Whether the contribution of neoatherosclerosis in restenosis of second-generation DES is similar to that of first-generation DES is not well established; however, observations of restenosis due to neoatherosclerosis have also been reported for second-generation DES [33].
Late restenosis due to neoatherosclerosis. a Angiogram of patient presenting with stable angina 15 years after sirolimus-eluting stent implantation reveals a possible restenotic lesion in the site of the previously implanted stent. Optical coherence tomography study reveals a homogeneous pattern of coverage (green) proximal to the stent (b), but with neoatherosclerosis development (yellow) at the distal segment (c and d) causing lumen compromise. Panels on the right are color-coded cartoons of the corresponding OCT images, explaining the composition of the neointimal tissue.
Neoatherosclerosis and stent thrombosis
Neoatherosclerosis also appears to contribute to the development of (very) late stent thrombosis (Fig. 4). Initial pathological studies demonstrated a role for the underlying plaque in BMS thrombosis, while subsequent studies in DES incriminated an impaired healing response with high rates of uncovered struts and vascular toxicity-associated malapposition as the main pathogenetic factor [46, 47]. Nevertheless, more recent pathological and intracoronary imaging data support the hypothesis that neoatherosclerosis is an active causal mechanism in a high percentage of cases with late stent thrombosis [32, 48, 49]. Autopsy studies have shown that thrombosis might develop on the grounds of neointimal plaque rupture [32], while thrombus aspirate samples from patients with very late BMS thrombosis demonstrate the presence of atherosclerotic plaque components such as foamy macrophages and cholesterol crystals within the aspirates [48]. Simultaneously, a number of studies have examined the OCT findings in patients with late and very late stent thrombosis, in an attempt to identify the pathomechanisms [50–54]. Despite minor discrepancies in the exact prevalence of neoatherosclerosis and the relative contribution of neointimal plaque rupture in each study, these studies collectively demonstrate a very high prevalence of plaque rupture as a pathomechanism for very late BMS thrombosis [55], and an almost equal contribution for neointimal plaque rupture and impaired healing in (very) late DES thrombosis [50, 52, 53]. Compared to an impaired healing response, neointimal rupture is a mechanism occurring mainly at longer intervals since stent implantation [50, 56], in neointimal regions with high necrotic core content and thin fibrous cap, similarly to what is observed in native atherosclerosis, but also with a very high incidence of macrophage infiltration [49].
Very late thrombosis due to neoatherosclerosis. a Angiogram of patient presenting with ST-elevation myocardial infarction 7 years after everolimus-eluting stent implantation in the right coronary artery showing total occlusion. After thrombus aspiration an in-stent lesion is obvious (b), while optical coherence tomography study shows neoatherosclerosis development (yellow) with neointimal rupture leading to cavity formation (Cav) and thrombus (red) (c–f). Homogeneous neointima is indicated in green. Panels on the bottom are color-coded cartoons of the corresponding OCT images, explaining the composition of the neointimal tissue.
Evidence also suggests that the clinical presentation of late stent failure is not exclusively affected by the presence of neoatherosclerosis but also by the morphological characteristics. In patients with in-stent restenosis, a higher incidence of OCT-defined thin-cap fibroatheroma, intimal rupture, and thrombi was identified in patients presenting with unstable angina compared with patients with stable symptoms [9]. Our group investigated the differences in the prevalence of neoatherosclerosis with regard to the clinical presentation [49]. Both stent restenosis and stent thrombosis had a higher prevalence of neoatherosclerosis compared with the asymptomatic group (stent thrombosis: 67 % vs. restenosis: 85 % vs. asymptomatic: 32 %; p < 0.001). However, the prevalence of neointimal rupture was higher in the stent thrombosis group (stent thrombosis: 41 % vs. restenosis: 15 % vs. asymptomatic: 5 %; p < 0.001) (Fig. 5a). This finding underscores the contribution of neoatherosclerosis development in the pathogenesis of late stent failure, while it indicates that neointimal rupture is mainly associated with acute presentation, although not infrequently encountered in stable or asymptomatic in-stent lesions.
a Prevalence of neoatherosclerosis and neointimal rupture according to clinical presentation [49]. b Change in the understanding of stent failure by intravascular imaging insights. Although stent restenosis had traditionally been considered a benign entity associated with stable symptoms and stent thrombosis had been considered a life-threatening entity associated with acute presentation, intravascular imaging has disclosed the participation of neoatherosclerosis in both entities, with several imaging findings such as in-stent necrotic core, neointimal rupture, and thrombus being common between them
Overall, intravascular imaging has changed the understanding of the pathogenesis of late restenosis and thrombosis by disclosing that neoatherosclerosis is a common pathomechanism in both entities (Fig. 5b). Stent restenosis had traditionally been considered a rather benign entity associated with stable symptoms, while stent thrombosis was seen as a life-threatening entity associated with acute presentation, with these two entities considered to be associated with different pathogenetic substrates. However, the presence—to a different extent—of common imaging findings such as in-stent necrotic core, neointimal rupture, and thrombus in both entities implies that these two presentations can occur as different manifestations of the same unfavorable healing process.
Neoatherosclerosis and bioresorbable scaffolds
Bioresorbable vascular scaffolds (BVS) are a new technology for percutaneous revascularization that are resorbed after an interval of 2–4 years since implantation. Conceptually, after this interval, the permanent vessel caging with the associated endothelial dysfunction disappears and this can lead to a reduction of stent-induced complications such as hypersensitivity reactions or neoatherosclerosis. First-in-man studies of BRS have demonstrated a favorable healing response at a very long-term follow-up with complete strut reabsorption, late luminal enlargement, and a potentially favorable plaque modification, while no cases of necrotic core accumulation of adluminal origin were observed [57]. As after bioresorption, the area corresponding to struts and neointima is consolidated with the underlying plaque, it resembles a native atherosclerotic plaque that is now well separated by the lumen by a signal-rich layer, a process modulated by hemodynamic factors [58] (Fig. 6). Nevertheless, as these first-in-man studies have focused on simple lesions, data on more complex lesions including thrombotic lesions are scarce [59]. Studies on more complex populations are ongoing, without, however, any worrying signs regarding neoatherosclerosis development in human studies of BVS thus far. Moreover, studies reporting on (very) late BVS thrombosis have not shown any evidence of scaffold thrombosis due to neointimal plaque rupture [60]. Therefore, the further development and implementation in the clinical practice of bioresorbable technologies might allow these complications to be overcome, although more long-term data are needed to confirm a potential benefit of this technology.
Paradigm of healing response in metal stents and bioresorbable scaffolds. After metal stent implantation, struts are preserved and the neointimal area is clearly delineated between the stent and lumen contour even at long-term follow-up, with possible development of neoatherosclerosis within the neointima. Conversely, in long-term follow-up of bioresorbable scaffolds, neointimal boundaries are unclear after bioresorption (dotted line), and the intima resembles a native plaque, defined as neoplaque. The signal-rich layer is the layer that separates the underlying plaque components from the lumen. BVS bioresorbable vascular scaffold. (Adapted from Karanasos et al. [57])
Newer OCT techniques for assessment of neoatherosclerosis
As previously mentioned, OCT is associated with some limitations in assessment of neoatherosclerosis. Nevertheless, new developments in OCT could improve in-stent tissue characterization and provide an enhanced and more objective neointimal tissue characterization. One of these developments is quantitative attenuation imaging. Tissue properties such as attenuation have been shown in ex vivo studies to be associated with the presence of macrophages and/or necrotic core [61, 62], while in vivo studies have demonstrated the potential of this technique for the detection of lipid plaques and fibroatheromas [63]. Therefore, this technique could be used for the assessment of neoatherosclerosis, since the optical properties of tissue components such as macrophages and a necrotic core are similar between native atherosclerosis and neoatherosclerosis. OCT assessment of neoatherosclerosis can also be improved through software for automated fibrous cap thickness measurement that can reduce human variability in the assessment of in-stent fibroatheromas [64]. Furthermore, the application of technologies such as polarization-sensitive OCT in humans can provide further structural information such as collagen and smooth muscle content of neointimal tissue [65], while high-speed OCT catheters will be able to acquire images from an entire coronary artery in less than 1 s [66].
Conclusion and perspectives
Recent evidence has highlighted the significance of in-stent neoatherosclerosis as a disease entity that comprises an important substrate for late adverse cardiac events after stent implantation and is one of the major pitfalls of the current generation of metallic stents. Pathological studies have demonstrated that infiltration of lipid-laden foamy macrophages with or without necrotic core and/or calcification formation constitutes the pathological substrate. A number of in vivo studies have used OCT—an imaging modality that can accurately identify morphological characteristics such as in-stent necrotic core, in-stent calcification, macrophage infiltration, and neointimal plaque rupture—in order to assess the in vivo characteristics and implications of this disease entity. These have demonstrated that neoatherosclerosis appears earlier and more frequently in drug-eluting stents compared with bare metal stents, is associated with several clinical and morphological factors, while its prevalence increases over time. Importantly, an association of neoatherosclerosis with late stent failure has been demonstrated, manifesting either as late restenosis, or in cases with neointimal plaque rupture as late stent thrombosis. Bioresorbable technologies hold promise for the reduction of this entity; however, their long-term healing response needs to be better documented. Further studies are needed so as to demonstrate what the prognostic implications of neoatherosclerosis detection are, and whether the natural history of this disease entity can be modified. Meanwhile, new devices for percutaneous revascularization need to be developed that can alleviate this entity, in order to be able to reduce the incidence of very late events after percutaneous coronary intervention.
References
Daemen J, Wenaweser P, Tsuchida K et al (2007) Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study. Lancet 369:667–678
Räber L, Magro M, Stefanini GG et al (2012) Very late coronary stent thrombosis of a newer-generation everolimus-eluting stent compared with early-generation drug-eluting stents a prospective cohort study. Circulation 125:1110–1121
Yamaji K, Kimura T, Morimoto T et al (2010) Very long-term (15–20 years) clinical and angiographic outcome after coronary bare metal stent implantation. Circ Cardiovasc Interv 3:468–475
Holmes DR Jr, Kereiakes DJ, Garg S et al (2010) Stent thrombosis. J Am Coll Cardiol 56:1357–1365
Windecker S, Meier B (2007) Late coronary stent thrombosis. Circulation 116:1952–1965
Nakazawa G, Otsuka F, Nakano M et al (2011) The pathology of neoatherosclerosis in human coronary implants bare-metal and drug-eluting stents. J Am Coll Cardiol 57:1314–1322
Inoue K, Abe K, Ando K et al (2004) Pathological analyses of long-term intracoronary Palmaz-Schatz stenting; is its efficacy permanent? Cardiovasc Pathol 13:109–115
Ali ZA, Roleder T, Narula J et al (2013) Increased thin-cap neoatheroma and periprocedural myocardial infarction in drug-eluting stent restenosis: multimodality intravascular imaging of drug-eluting and bare-metal stents. Circ Cardiovasc Interv 6:507–517
Kang SJ, Mintz GS, Akasaka T et al (2011) Optical coherence tomographic analysis of in-stent neoatherosclerosis after drug-eluting stent implantation. Circulation 123:2954–2963
Muraoka Y, Sonoda S, Kashiyama K et al (2012) Evaluation of in-stent neointimal tissue components using integrated backscatter intravascular ultrasound: comparison of drug-eluting stents and bare-metal stents. Int J Cardiovasc Imaging 28:1635–1641
Yokoyama S, Takano M, Yamamoto M et al (2009) Extended follow-up by serial angioscopic observation for bare-metal stents in native coronary arteries: from healing response to atherosclerotic transformation of neointima. Circ Cardiovasc Interv 2:205–212
Tearney GJ, Regar E, Akasaka T et al (2012) Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J Am Coll Cardiol 59:1058–1072
Karanasos A, Ligthart J, Witberg K et al (2012) Optical coherence tomography: potential clinical applications. Curr Cardiovasc Imaging Rep 5:206–220
Tanimoto S, Aoki J, Serruys PW et al (2006) Paclitaxel-eluting stent restenosis shows three-layer appearance by optical coherence tomography. EuroIntervention 1:484
Takano M, Yamamoto M, Inami S et al (2009) Appearance of lipid-laden intima and neovascularization after implantation of bare-metal stents extended late-phase observation by intracoronary optical coherence tomography. J Am Coll Cardiol 55:26–32
Nakano M, Vorpahl M, Otsuka F et al (2012) Ex vivo assessment of vascular response to coronary stents by optical frequency domain imaging. JACC Cardiovasc Imaging 5:71–82
Karanasos A, Ligthart JM, Regar E (2012) In-stent neoatherosclerosis: a cause of late stent thrombosis in a patient with “full metal jacket” 15 years after implantation: insights from optical coherence tomography. JACC Cardiovasc Interv 5:799–800
Gonzalo N, Serruys PW, Okamura T et al (2009) Optical coherence tomography patterns of stent restenosis. Am Heart J 158:284–293
Nagai H, Ishibashi-Ueda H, Fujii K (2010) Histology of highly echolucent regions in optical coherence tomography images from two patients with sirolimus-eluting stent restenosis. Catheter Cardiovasc Interv 75:961–963
Tellez A, Afari ME, Buszman PP et al (2014) Peri-strut low-intensity areas in optical coherence tomography correlate with peri-strut inflammation and neointimal proliferation: an in-vivo correlation study in the familial hypercholesterolemic coronary swine model of in-stent restenosis. Coron Artery Dis 25:595–601
Kim JS, Afari ME, Ha J et al (2014) Neointimal patterns obtained by optical coherence tomography correlate with specific histological components and neointimal proliferation in a swine model of restenosis. Eur Heart J Cardiovasc Imaging 15:292–298
Kim JS, Hong MK, Shin DH et al (2012) Quantitative and qualitative changes in DES-related neointimal tissue based on serial OCT. JACC Cardiovasc Imaging 5:1147–1155
Kozuki A, Shinke T, Otake H et al (2013) Temporal course of vessel healing and neoatherosclerosis after DES implantation. JACC Cardiovasc Imaging 6:1121–1123
Van Soest G, Regar E, Goderie TP et al (2011) Pitfalls in plaque characterization by OCT: image artifacts in native coronary arteries. JACC Cardiovasc Imaging 4:810–813
Otsuka F, Byrne RA, Yahagi K et al (2015) Neoatherosclerosis: overview of histopathologic findings and implications for intravascular imaging assessment. Eur Heart J. pii: ehv205. 2015 May 20. [EPub ahead of print]
Hou J, Qi H, Zhang M et al (2010) Development of lipid-rich plaque inside bare metal stent: possible mechanism of late stent thrombosis? An optical coherence tomography study. Heart 96:1187–1190
Kitabata H, Loh JP, Pendyala LK et al (2014) Intra-stent tissue evaluation within bare metal and drug-eluting stents > 3 years since implantation in patients with mild to moderate neointimal proliferation using optical coherence tomography and virtual histology intravascular ultrasound. Cardiovasc Revasc Med 15:149–155
Yonetsu T, Kato K, Kim SJ et al (2012) Predictors for neoatherosclerosis: a retrospective observational study from the optical coherence tomography registry. Circ Cardiovasc Imaging 5:660–666
Kang SJ, Song HG, Ahn JM et al (2012) OCT-verified neoatherosclerosis in BMS restenosis at 10 years. JACC Cardiovasc Imaging 5:1267–1268
Yonetsu T, Kim JS, Kato K et al (2012) Comparison of incidence and time course of neoatherosclerosis between bare metal stents and drug-eluting stents using optical coherence tomography. Am J Cardiol 110:933–939
Kim JS, Kim JS, Shin DH et al (2012) Optical coherence tomographic comparison of neointimal coverage between sirolimus- and resolute zotarolimus-eluting stents at 9 months after stent implantation. Int J Cardiovasc Imaging 28:1281–1287
Otsuka F, Vorpahl M, Nakano M et al (2014) Pathology of second-generation everolimus-eluting stents versus first-generation sirolimus- and paclitaxel-eluting stents in humans. Circulation 129:211–223
Lee SY, Hur SH, Lee SG et al (2015) Optical coherence tomographic observation of in-stent neoatherosclerosis in lesions with more than 50 % neointimal area stenosis after second-generation drug-eluting stent implantation. Circ Cardiovasc Interv 8:e001878
Kuramitsu S, Sonoda S, Yokoi H et al (2014) Long-term coronary arterial response to biodegradable polymer biolimus-eluting stents in comparison with durable polymer sirolimus-eluting stents and bare-metal stents: five-year follow-up optical coherence tomography study. Atherosclerosis 237:23–29
Joner M, Nakazawa G, Finn AV et al (2008) Endothelial cell recovery between comparator polymer-based drug-eluting stents. J Am Coll Cardiol 52:333–342
Tian F, Chen Y, Liu H et al (2014) Assessment of characteristics of neointimal hyperplasia after drug-eluting stent implantation in patients with diabetes mellitus: an optical coherence tomography analysis. Cardiology 128:34–40
Virmani R, Kolodgie FD, Burke AP et al (2005) Atherosclerotic plaque progression and vulnerability to rupture: angiogenesis as a source of intraplaque hemorrhage. Arterioscler Thromb Vasc Biol 25:2054–2061
Ino Y, Kubo T, Kitabata H et al (2013) Difference in neointimal appearance between early and late restenosis after sirolimus-eluting stent implantation assessed by optical coherence tomography. Coron Artery Dis 24:95–101
Kim BK, Kim JS, Shin DH et al (2012) Optical coherence tomography evaluation of in-stent restenotic lesions with visible microvessels. J Invasive Cardiol 24:116–120
Tian J, Ren X, Uemura S et al (2014) Spatial heterogeneity of neoatherosclerosis and its relationship with neovascularization and adjacent plaque characteristics: optical coherence tomography study. Am Heart J 167:884–892.e882
Kitabata H, Kubo T, Komukai K et al (2012) Effect of strut thickness on neointimal atherosclerotic change over an extended follow-up period (≥ 4 years) after bare-metal stent implantation: intracoronary optical coherence tomography examination. Am Heart J 163:608–616
Toutouzas K, Karanasos A, Stefanadis C (2013) Inflammatory mechanisms of adverse reactions to BMS. Curr Vasc Pharmacol 11:379–391
Park SJ, Kang SJ, Virmani R et al (2012) In-stent neoatherosclerosis: a final common pathway of late stent failure. J Am Coll Cardiol 59:2051–2057
Vergallo R, Yonetsu T, Uemura S et al (2013) Correlation between degree of neointimal hyperplasia and incidence and characteristics of neoatherosclerosis as assessed by optical coherence tomography. Am J Cardiol 112:1315–1321
Habara M, Terashima M, Nasu K et al (2013) Morphological differences of tissue characteristics between early, late, and very late restenosis lesions after first generation drug-eluting stent implantation: an optical coherence tomography study. Eur Heart J Cardiovasc Imaging 14:276–284
Farb A, Burke AP, Kolodgie FD et al (2003) Pathological mechanisms of fatal late coronary stent thrombosis in humans. Circulation 108:1701–1706
Finn AV, Joner M, Nakazawa G et al (2007) Pathological correlates of late drug-eluting stent thrombosis: strut coverage as a marker of endothelialization. Circulation 115:2435–2441
Yamaji K, Inoue K, Nakahashi T et al (2012) Bare metal stent thrombosis and in-stent neoatherosclerosis. Circ Cardiovasc Interv 5:47–54
Karanasos A, Ligthart J, Witberg K et al (2013) Association of neointimal morphology by optical coherence tomography with rupture of neoatherosclerotic plaque very late after coronary stent implantation. In: Progress in Biomedical Optics and Imaging – Proceedings of SPIE. p 856542
Amabile N, Souteyrand G, Ghostine S et al (2014) Very late stent thrombosis related to incomplete neointimal coverage or neoatherosclerotic plaque rupture identified by optical coherence tomography imaging. Eur Heart J Cardiovasc Imaging 15:24–31
Amioka M, Shiode N, Kawase T et al (2014) Causes of very late stent thrombosis investigated using optical coherence tomography. Intern Med 53:2031–2039
Kang S-J, Lee CW, Song H et al (2013) OCT analysis in patients with very late stent thrombosis. JACC Cardiovasc Interv 6:695–703
Karanasos A, Witberg K, Van Geuns RJ et al (2012) Morphological characteristics by optical coherence tomography of ruptured neoatherosclerotic plaques in patients with very late stent thrombosis (abstract). Eur Heart J 33:176–176
Ko YG, Kim DM, Cho JM et al (2012) Optical coherence tomography findings of very late stent thrombosis after drug-eluting stent implantation. Int J Cardiovasc Imaging 28:715–723
Karanasos A, Witberg K, Ligthart J et al (2013) In-stent neoatherosclerosis: Are first generation drug eluting stents different than bare metal stents? An optical coherence tomography study. In: Progress in Biomedical Optics and Imaging – Proceedings of SPIE. p 856543
Karanasos A, Ligthart J, Schultz C et al (2012) Clinical and angiographic predictors of neointimal rupture as a culprit for very late stent thrombosis. Insights from optical coherence tomography (abstract). Eur Heart J 33:399–399
Karanasos A, Simsek C, Gnanadesigan M et al (2014) OCT assessment of the long-term vascular healing response 5 years after everolimus-eluting bioresorbable vascular scaffold. J Am Coll Cardiol 64:2343–2356
Karanasos A, Schuurbiers JC, Garcia-Garcia HM et al (2015) Association of wall shear stress with long-term vascular healing response following bioresorbable vascular scaffold implantation. Int J Cardiol 191:279–283
Karanasos A, Muramatsu T, Diletti R et al (2015) Early and late optical coherence tomography findings following everolimus-eluting bioresorbable vascular scaffold implantation in myocardial infarction: a preliminary report. Hellenic J Cardiol 56:125–135
Karanasos A, Van Mieghem N, Van Ditzhuijzen N et al (2015) Angiographic and optical coherence tomography insights into bioresorbable scaffold thrombosis: single-center experience. Circ Cardiovasc Interv 8:e002369
Van Soest G, Goderie T, Regar E et al (2010) Atherosclerotic tissue characterization in vivo by optical coherence tomography attenuation imaging. J Biomed Opt 15:011105
Gnanadesigan M, Van Soest G, White S et al (2014) Effect of temperature and fixation on the optical properties of atherosclerotic tissue: a validation study of an ex-vivo whole heart cadaveric model. Biomed Opt Express 5:1038–1049
Regar E, Gnanadesigan M, Van Der Steen AF et al (2013) Quantitative optical coherence tomography tissue-type imaging for lipid-core plaque detection. JACC Cardiovasc Interv 6:891–892
Zahnd G, Karanasos A, Van Soest G et al (2015) Quantification of fibrous cap thickness in intracoronary optical coherence tomography with a contour segmentation method based on dynamic programming. Int J Comput Assist Radiol Surg. doi:10.1007/s11548-015-1164-7
Nadkarni SK, Pierce MC, Park BH et al (2007) Measurement of collagen and smooth muscle cell content in atherosclerotic plaques using polarization-sensitive optical coherence tomography. J Am Coll Cardiol 49:1474–1481
Wang T, Wieser W, Springeling G et al (2013) Intravascular optical coherence tomography imaging at 3200 frames per second. Opt Lett 38:1715–1717
Alfonso F, Dutary J, Paulo M et al (2012) Combined use of optical coherence tomography and intravascular ultrasound imaging in patients undergoing coronary interventions for stent thrombosis. Heart 98:1213–1220
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B.C. Zhang, A. Karanasos, and E. Regar state that there are no conflicts of interest. The accompanying manuscript does not include studies on humans or animals.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, BC., Karanasos, A. & Regar, E. OCT demonstrating neoatherosclerosis as part of the continuous process of coronary artery disease. Herz 40, 845–854 (2015). https://doi.org/10.1007/s00059-015-4343-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-015-4343-y
Keywords
- Atherosclerosis
- Optical coherence tomography
- Percutaneous coronary intervention
- Drug-eluting stents
- Bare metal stents