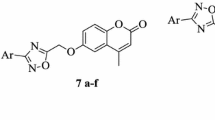
Abstract
A new series of 7-O-substituted pyridyl-4-methyl coumarin derivatives were synthesized and characterized based on their spectral data. All the target compounds were evaluated for their in vitro antifungal activity against eight important fungal pathogens. This study showed that the introduction of the substituted pyridyl moiety at the 7-hydroxy position of coumarin could enhance the antifungal activities. It also indicated that a bulky substituent was not beneficial to the antifungal activity of those coumarin derivatives.

Similar content being viewed by others
References
Alvey L, Prado S, Saint-Joanis B, Michel S, Koch M, Cole ST, Tillequin F, Janin YL (2009) Diversity-oriented synthesis of furo [3,2-f] chromanes with antimycobacterial activity. Eur J Med Chem 44(6):2497–2505
Borges F, Roleira F, Milhazes N, Santana L, Uriarte E (2005) Simple coumarins and analogues in medicinal chemistry: occurrence, synthesis and biological activity. Curr Med Chem 12(8):887–916
Chopra I, Schofield C, Everett M, O’Neill A, Miller K, Wilcox M, Frere JM, Dawson M, Czapiewski L, Urleb U, Courvalin P (2008) Lancet infectious diseases treatment of healthcare associated infections caused by gram negative bacteria a consensus statement. Lancet Infect Dis 8(2):133–139
Emmanuel-Giota AA, Fylaktakidou KC, Hadjipavlou-Litina DJ, Litinas KE, Nicolaides DN (2001) Synthesis and biological evaluation of several 3-(coumarin-4-yl) tetrahydroisoxazole and 3-(coumarin-4-yl) dihydropyrazole derivatives. J Heterocycl Chem 38(3):717–722
Fridkin SK, Jarvis WR (1996) Epidemiology of nosocomial fungal infections. Clin Microbiol Rev 9(4):499–511
Ghate M, Manohar D, Kulkarni V, Shobha R, Kattimani SY (2003) Synthesis of vanillin ethers from 4-(bromomethyl) coumarins as anti-inflammatory agents. Eur J Med Chem 38(3):297–302
Jeso V, Nicolaou KC (2009) Total synthesis of tovophyllin B. Tetrahedron Lett 50(11):1161–1163
Jeu L, Piacenti FJ, Lyakhovetskiy AG, Fung HB (2003) Voriconazole. Clin Ther 25(5):1321–1381
Keri RS, Hosamani KM, Shingalapur RV, Reddy HR (2009) 2-Azetidinone derivatives: design, synthesis, in vitro anti-microbial, cytotoxic activities and DNA cleavage study. Eur J Med Chem 44(12):5123–5130
Kulkarni MV, Kulkarni GM, Lin CH, Sun CM (2006) Recent advances in coumarins and 1-azacoumarins as versatile biodynamic agents. Curr Med Chem 13(23):2795–2818
Kumar A, Sharma S, Gupta LP, Ahmad P, Srivastava SP, Rahuja N, Tamrakar AK, Srivastava AK (2012) Synthesis of propiophenone derivatives as new class of antidiabetic agents reducing body weight in db/db mice. Bioorg Med Chem 20(6):2172–2179
Lepesheva GI, Zaitseva NG, Nes WD, Zhou W, Arase M, Liu J, Hill GC, Waterman MR (2006) CYP51 from Trypanosoma cruzi: a phyla-specific residue in the B′ helix defines substrate preferences of sterol 14α-demethylase. J Biol Chem 281(6):3577–3585
National Committee for Clinical Laboratory Standards (2002) Reference method for broth dilution antifungal susceptibility testing of yeasts approved standard document M27–A2. National Committee for Clinical Laboratory Standards, Wayne
Okaniwa M, Hirose M, Imada T, Ohashi T, Hayashi Y, Miyazaki T, Arita T, Yabuki M, Kakoi K, Kato J, Takagi T, Kawamoto T, Yao S, Sumita A, Tsutsumi S, Tottori T, Oki H, Sang BC, Yano J, Aertgeerts K, Yoshida S, Ishikawa T (2012) Design and synthesis of novel DFG-out RAF/vascular endothelial growth factor receptor 2 (VEGFR2) inhibitors. 1. Exploration of [5,6]-fused bicyclic scaffolds. J Med Chem 55(7):3452–3578
Padmaja A, Payani T, Dinneswara RG, Padmavathi V (2009) Synthesis, antimicrobial and antioxidant activities of substituted pyrazoles, isoxazoles, pyrimidine and thioxopyrimidine derivatives. Eur J Med Chem 44(11):4557–4566
Padmaja A, Rajasekhar C, Muralikrishna A, Padmavathi V (2011) Synthesis and antioxidant activity of oxazolyl/thiazolylsulfonylmethyl pyrazoles and isoxazoles. Eur J Med Chem 46(10):5034–5038
Raghu M, Nagaraj A, Reddy CS (2009) ChemInform abstract: synthesis and in vitro study of novel bis-[3-(2-arylmethylidenimino-1,3-thiazol-4-yl)-4-hydroxy-2H-chromen-2-one-6-yl]methane and bis-[3-(2-arylidenhydrazo-1,3-thiazol-4-yl)-4-hydroxy-2H- chromen-2-one-6-yl]methane as potential antimicrobial agents. J Heterocycl Chem 40(36):261–267
Riveiro ME, Kimpe ND, Moglioni A, Vazquez R, Monczor F, Shayo C, Davio C (2010) Coumarins: old compounds with novel promising therapeutic perspectives. Curr Med Chem 17(13):1325–1338
Ronad PM, Noolvi MN, Sapkal S, Dharbhamulla S, Maddi VS (2010) Synthesis and antimicrobial activity of 7-(2-substituted phenylthiazolidinyl) -benzopyran-2-one derivatives. Eur J Med Chem 45(1):85–89
Shi Y, Zhou CH (2011) Synthesis and evaluation of a class of new coumarin triazole derivatives as potential antimicrobial agents. Bioorg Med Chem Lett 21(3):956–960
Wingard JR, Leather H (2004) A new era of antifungal therapy. Biol Blood Marrow Transplant 10(2):73–90
Wu L, Wang X, Xu W, Farzaneh F, Xu R (2009) The structure and pharmacological functions of coumarins and their derivatives. Curr Med Chem 16(32):4236–4260
Xie L, Takeuchi Y, Cosentino LM, Lee KH (1999) Anti-AIDS agents. 37. Synthesis and structure–activity relationships of (3′R,4′R)-(+)-cis-khellactone derivatives as novel potent anti-HIV agents. J Med Chem 42(14):2662–2672
Xu ZQ, Pupek K, Suling WJ, Enache L, Flavin MT (2006) Pyranocoumarin, a novel anti-TB pharmacophore: synthesis and biological evaluation against mycobacterium tuberculosis. Bioorg Med Chem 14(13):4610–4626
Acknowledgments
This work was supported by the Pharmaceutical Education research project of Chinese Association of Higher Medical Education Professional Committee (Grant No. ZD201220), the Creativity and Innovation Training Program of Second Military Medical University (Grant No. ZD2008008), and the Shanghai Leading Academic Discipline Project (No. B906).
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiaoyun Chai and Shichong Yu contributed equally to this study.
Rights and permissions
About this article
Cite this article
Chai, X., Yu, S., Wang, X. et al. Synthesis and antifungal activity of novel 7-O-substituted pyridyl-4-methyl coumarin derivatives. Med Chem Res 22, 4654–4662 (2013). https://doi.org/10.1007/s00044-013-0470-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-013-0470-2