Abstract
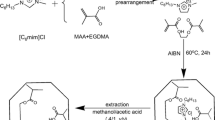
A molecularly imprinted polymer (MIP) having both amidine and imidazole functional groups in the active site has been prepared usingp-nitrophenyl phosphate as a transition state analogue (TSA). The imprinted polymer MIP with amidine and imidazole found to have the highest hydrolysis activity compared with other MIPs with either amidine or imidazole groups only. It is postulated a cooperative effect between amidine and imidazole in the hydrolysis ofp-nitrophenyl methyl carbonate (NPMC) as a substrate when both groups were arranged in proximity by molecular imprinting. The rate enhancement of the hydrolysis by MIP was 60 folds over the uncatalyzed solution reaction and two folds compared with the control non-imprinted polymer CP1 having both functional groups. The enzyme-mimetic catalytic hydrolysis ofp-nitrophenyl acetate by MIP was evaluated in buffer at pH 7.0 withK m of 1.06 mM andk cat of 0.137 h−1.
Similar content being viewed by others
References
G. Wulff,Chem. Rev.,102, 1 (2002);Angew. Chem. Int. Ed. Engl.,34, 1812 (1995);Chemtech.,19, 19 (November 1998).
K.-D. Ahn and J.-M. Kim,Polymer Sci. & Technol. (Korean),9, 137 (1998) and11, 332 (2000).
F. L. Dicker and O. Hayden,Adv. Mater.,12, 311 (2000); S. Piletsky, S. Alcock, and A. R. F. Turner,Trend Biotech.,19, 9 (2001).
B. Sellergren,Angew. Chem. Int. Ed. Engl.,39, 1031 (2000).
M. Whitcomb and E. N. Vulfson,Adv. Mater.,13, 467 (2000).
H. Asansuma, T. Hishiya, and M. Komiyama,Adv. Mater.,12, 1019 (2000).
A. Kugimiya, Y. Kuwada, and T. Takeuchi,J. Chromatogr. A,938, 131 (2001).
O. Kriz, O. Ramstrom, and K. Mosbach,Anal. Chem.,69, 345A (1997).
R. A. Lerner, S. J. Benkvoic, and P. G. Schultz,Science,252, 659 (1991).
A. Leonhardt and K. Mosbach,React. Polym.,6, 285 (1987); D. K. Robinson and K. Mosbach,J. Chem. Soc., Chem. Commun., 969 (1989).
K. Ohkubo, Y. Urata, S. Hirota, Y. Honda, Y. Fujishita, and T. Sagawa,J. Mol. Catal.,87, L21 (1994).
J. V. Beach and K. J. Shea,J. Am. Chem. Soc.,116, 379 (1994).
B. Sellergren, R. N. Karmalkar, and K. J. Shea,J. Org. Chem.,65, 4009 (2000).
J.-M. Kim, K.-D. Ahn, and G. Wulff,Macromol. Chem. Phys.,202, 1105 (2001).
A. G. Strikovsky, D. Kasper, M. Grün, B. S. Green, J. Hradil, and G. Wulff,J. Am. Chem. Soc.,122, 6295 (2000).
G. Wulff, T. Gross, and R. Schönfeld,Angew. Chem. Int. Ed. Engl.,36, 1962 (1997).
J.-M. Kim, K.-D. Ahn, G. Strikovsky, and G. Wulff,Bull. Korean Chem. Soc.,22, 689 (2001).
G. Wulff and R. Schönfeld,Adv. Mater.,10, 957 (1998)
C. G. Overberger, T. St. Pierre, N. Vorchheimer, J. Lee, and S. Yaroslavsky,J. Am. Chem. Soc.,87, 296 (1969).
B. S. Lee, M. G. Kulkarni, and R. A. Mashelkar,React. & Func. Polym.,39, 37 (1999).
H. Dugas,Bioorganic Chemistry, Third Ed., Springer-Verlag, New York, USA, 1996, pp 192.
Author information
Authors and Affiliations
Additional information
Dedicated to Dr. Un Young Kim on the occasion of his retirement.
Rights and permissions
About this article
Cite this article
Chen, W., Han, DK., Ahn, KD. et al. Molecularly imprinted polymers having amidine and imidazole functional groups as an enzyme-mimetic catalyst for ester hydrolysis. Macromol. Res. 10, 122–126 (2002). https://doi.org/10.1007/BF03218301
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03218301