Abstract
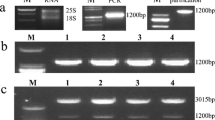
In plants ascorbic acid (AsA) is a strong antioxidant or reductant that can be converted to dehydroascorbate (DHA) by oxidation. DHA, a very short-lived chemical, can either be hydrolyzed irreversibly to 2,3-diketogulonic acid or recycled to AsA by dehydroascorbate reductase (DHAR).DHAR cDNA, isolated from sesame hairy roots, was inserted into two plant expression vector syrtems with theCaMV35S promoter (CaMV35S : :DHAR) and a potato tuber-specific promoter,Patatin (Patatin : :DHAR). Southern and northern blot hybridization analyses indicated thatDHAR cDNA was successfully integrated into the potato genome and actively transcribed. High levels of sesameDHAR transcript and DHAR enzyme activity were determined, by thePatatin promoter, in regenerated potato tubers, but their levels in leaves were very low. In contrast, much higher amounts of transcript were accumulated in the leaves of CaMV35S : :DHAR regenerants than in the tubers while the activity of DHAR enzyme was higher in the latter. AsA content in the tubers of Patatin : :DHAR transgenic lines was also increased (1.1- to 1.3-fold) compared with that of non-transgenic plants. However, this was not true for the transgenic leaves. In contrast, theCaMV35S promoter was associated with AsA accumulations in both the tubers (up to 1.6-fold) and the leaves (up to 1.5-fold). However, more detailed analyses indicated that this increased enzyme activity was not always accompanied by an elevation in AsA content from transgenic plants. This suggests that other factors may limit the accumulation of vitamin C via ascorbate-recycling in transgenic potato plants.
Similar content being viewed by others
Literature Cited
Agius F, Gonzales-Lamothe R, Caballero JL, Munoz-Blanco J, Botella MA, Valpuesta V (2003) Engineering increased vitamin C levels in plants by overexpression of a D-galactuionic acid reduotase. Nat Biotechnol21: 177–181
Asada K (1999) The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Physiol Plant Mol Biol50: 601–639
Chen Z, Young TE, Ling J, Chang SC, Gallie DR (2003) Increasing vitamin C content of plants through enhanced ascorbate recycling. Proc Natl Acad Sci USA100: 3525–3530
Chun JA, Seo JY, Han MO, Lee JW, Yi YB, Park GY, Lee SW, Bae SC, Cho KJ, Chung CH (2006) Comparative expression and characterization of dehydroascorbate reductase cDNA from transformed sesame hairy roots using real-time RT-PCR. J Plant Biol49: 507–512
Chun JA, Lee WH, Han MO, Lee JW, Yi YB, Goo YM, Lee SVV, Bae SC, Cho KJ, Chung CH (2007) Molecular and biochemical characterizations of dehydroascorbate reductase from sesame (Sesamum indicum L.) hairy root cultures. J Agric Food Chem55: 6067–6073
Crawford TC, Crawford SA (1980) Synthesis of L-ascorbic acid. Adv Carbohydr Chem Biochem37: 79–155
Davey MW, van Montagu M, Inzé D, Sanmartin M, Kanellis A, Smirnoff N, Benzie I JJ, Strain JJ, Favell D, Fletcher J (2000) Plant L-ascorbic acid: Chemistry, function, metabolism, bio-availability and effects of processing. J Sci Food Agric80: 825–860
Doyle JJ Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh tissue. Phytochem Bull19: 11–15
Farran I, Sánchez-Serrano JJ, Medina JF, Prieto J, Mingo-Castel AM (2002) Targeted expression of human serum to potato tubers. Transgen Res11: 337–346
Foyer CH (2000) Ascorbic acid,In RG Aischer, JL Hess, eds, Antioxidants in Higher Plants. CRC Press, Boca Raton, Ann Arbor, London, Tokyo, pp 31–58
Han JS, Kozukue N, Young KS, Lee KR, Friedman M (2004) Distribution of ascorbic acid in potato tubers and home-processed and commercial potato foods. J Agric Food Chem52: 6516–6521
Hossain AM, Asada K (1984) Purification of dehydroascorbate reductase from spinach and its characterization as a thiol enzyme. Plant Cell Physiol25: 85–92
Hossain AM, Nakano Y, Asada K (1984) Monodehydroascorbate reductase in spinach chloroplasts and its participation in regeneration of ascorbate for scavenging hydrogen peroxide. Plan Cell Physiol25: 385–395
Ishikawa T, Dowdle J, Smirnoff N (2006) Progress in manipulating ascorbic acid biosynthesis and accumulation in plants. Physiol Plant126: 343–355
Jain AK, Nessler CK (2000) Metabolic engineering of an alternative pathway for ascorbic acid biosynthesis in plants. Mol Breed6: 73–78
Kato Y, Urano J, Maki Y, Ushimaru T (1997) Purification and characterization of dehydroacorbate reductase from rice. Plant Cell Physiol38: 173–178
Kwon SY, Ahn YO, Lee HS, Kwak SS (2001) Biochemical characterization of transgenic tobacco plants expressing a human dehydroascorbate reductase gene. J Biochem Mol Biol34: 316–321
Kwon SY, Choi SM, Ahn YO, Lee HS, Lee HB, Park YM, Kwak SS (2003) Enhanced stress-tolerance of transgenic tobacco plants expressing a human dehydroascorbate reductase gene. J Plant Physiol160: 347–353
Lee JY, Seo HW, Yang MS, Robb EJ, Nazar RN, Lee SW (2004) Plant defense gene promoter enhances the reliability ofshiva-1 gene-induced resistance to soft rot disease in potato. Planta220: 165–171
Levine M, Conry-Cantilenat C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, Kings J, Cantilena LR (1996) Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc Natl Acad Sci USA93: 3704–3709
Liu XJ, Prat S, Willmitzer L, Frommer WB (1990) Cis regulatory elements directing tuber-specific and sucrose-inducible expression of a chimeric class I patatin promoter/GUS-gene fusion. Mol Gen Genet223: 401–406
Loewus FA (1999) Biosynthesis and metabolism of ascorbic acid in plants and of analogs of ascorbic acid in fungi. Phytochemistry52: 193–210
Lorence A, Chevone BI, Mendes P, Nestler CL (2004) Myo-inositol oxygenase offers a possible entry point into plant ascorbate biosynthesis. Plant Physiol134: 1200–1205
Mignery GA, Pikaard CS, Park WD (1988) Molecular characterization of the patatin multigene family of potato. Gene62: 27–44
Niyogi KK (1999) Photoprotection revisited: Genetic and molecular approaches. Annu Rev Plant Physiol Plant Mol Biol50: 333–359
Rocha-Sosa M, Sonnewald U, Frommer W, Stratmann M, Schell J, Willmitzer L (1989) Both developmental and metabolic signals activate the promoter of a class I patatin gene. EMBO J8: 23–29
Roig MG, Rivera ZS, Kennedy JF (1993) L-Ascorbic acid: An overview. Intl J Biochem Biophys145: 685–692
Savini I, Catani MV, Rossi A, Duranti G, Ranalli M, Melino G, Sabatini S, Avigliano L (2003) Vitamin C recycling is enhanced in the adaptive response to leptin-incluced oxidative stress in Keratinoctes. J Invest Dermatol121: 786–793
Shimaoka T, Yokota A, Miyake C (2000) Purification and characterization of chloroplast dehydroascorbate reductase from spinach leaves. Plant Cell Physiol41: 1110–1118
Tedone L, Hancock RD, Alberino S, Haupt S, Viola R (2004) Longdistance transport of L-ascorbic acid in potato. BMC Plant Biol4: 16–22
Topuz A, Ozdemir F (2007) Assessment of carotenoids, capsaicinoids and ascorbic acid composition of some selected pepper cultivars (Capsicum annum L.) grown in Turkey. J Food Compost Anal20: 596–602
Urano J, Nakagawa T, Maki Y, Masumura T, Tanaka K, Murata N, Ushimaru T (2000) Molecular cloning and characterization of a rice dehydroascorbate reductase. FEBS Lett466: 107–111
Vethanayagam JG, Green EH, Rose RC, Bode AM (1999) Glutathione-dependent ascorbate recycling activity of rat serum albumin. Free Radical Biol Med26: 1591–1598
Visser RGF, Jacobsen EHM, Schans MJ, Witholt B, Feenstra WJ (1989) Transformation of homozygous diploid potato with anAgrobacterium tumefaciens binary vector system by adventitious shoot regeneration on leaf and stem segments. Plant Mol Biol12: 329–337
Wheeler G, Jones MA, Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature393: 365–369
Zou L, Li H, Ouyang B, Zhang J, Ye Z (2006) Cloning and mapping of genes involved in tomato ascorbic acid biosynthesis and metabolism. Plant Sci170: 120–127
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goo, YM., Chun, H.J., Kim, TW. et al. Expressional characterization of dehydroascorbate reductase cDNA in transgenic potato plants. J. Plant Biol. 51, 35–41 (2008). https://doi.org/10.1007/BF03030738
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03030738