Summary
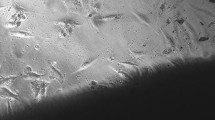
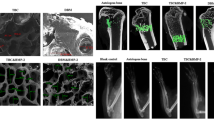
The biocompatibility and osteogenic activity of allogenic decalcified bone matrix (DBM) used as a carrier for bone tissue engineering were studied. Following the method described by Urist, allogenic DBM was made.In vitro, DBM and bone marrow stromal cell (BMSC) from rabbits were co-cultured for 3–7 days and subjected to HE staining, and a series of histomorphological observations were performed under phase-contrast microscopy and scanning electron microscopy (SEM).In vivo the mixture of DBM/BMSC co-cultured for 3 days was planted into one side of muscules sacrospinalis of rabbits, and the DBM without BMSC was planted into other side as control. Specimens were collected at postoperative week 1, 2 and 4, and subjected to HE staining, and observed under SEM. The results showed during culturein vitro, the BMSCs adherent to the wall of DBM grew, proliferated and had secretive activity. Thein vivo experiment revealed that BMSCs and undifferentiated mesenchymal cells in the perivascular region invaded gradually and proliferated together in DBM/BMSC group, and colony-forming units of chondrocytes were found. Osteoblasts, trabecular bone and medullary cavity appeared. The inflammatory reaction around muscles almost disappeared at the second weeks. In pure DBM group, the similar changes appeared from the surface of the DBM to center, and the volume of total regenerate bones was less than the DBM/BMSC group at the same time. The results indicated that the mixture of DBM and BMSC had good biocompatibility and ectopic induced osteogenic activity.
Similar content being viewed by others
References
Urist M R. Bone formation by autoinduction. Science, 1965, 150:893
Kawamuna M, Teixido J, Laiho Net al. Chondroosteogenetic response to crude bone matrix proteins bound to hydroxyapatite. Clin Orthop, 1990, 217:281
Oakes D A, Lee C C, Lieberman J R. An evaluation of human demineralized bone matrices in a rat femoral defect model. Clin Orthop, 2003, 281:290
Edwards J T, Diegmann M H, Scarborough N L. Osteoinduction of human demineralized bone: Characterization in a rat model. Clin Orthop, 1998, 357:219
Coutts R D, Healey R M, Ostrander R. Tissue engineering, cells, scaffolds, and growth factors: Matrices for cartilage repair. Clin Orthop, 2001, (391 Suppl): S271
Wang J C, Kanim L E, Nagakawa I Set al. Dose-dependent toxicity of a commercially available demineralized bone matrix material. Spine, 2001, 26:1429.
Langstaff S, Sayer M, Smith T Jet al. Resorbable bioceramics based on stabilized calcium phosphates. Part I: rational design, sample preparation and material characterization. J Biomaterial, 1999, 20:1727
Eckart H, Bundgaard K G, Christensen K Set al. Effects of locally applied vascular endothelial growth factor (VEGF) and VEGF-inhibitor to the rabbit tibia during distraction osteogenesis. J Orthop Res, 2003, 21:335
Author information
Authors and Affiliations
Additional information
ZIENG Dong male, born in 1978, Postgraduate Student
Rights and permissions
About this article
Cite this article
Dong, Z., Shuhua, Y., Jin, L. et al. Experimental study on allogenic decalcified bone matrix as carrier for bone tissue engineering. Current Medical Science 24, 147–150 (2004). https://doi.org/10.1007/BF02885415
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02885415