Summary
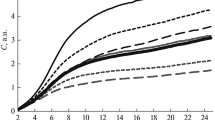
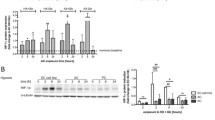
The effects of anoxia and reoxygenation on major antioxidant enzyme activities were investigatedin vitro in immortalized rat brain endothelial cells (RBE4 cells). A sublethal anoxic period of 12 h was assessed for RBE4 cells using the neutral red uptake test. Anoxia markedly influenced the specific activity of catalase and superoxide dismutase, with no major effect on glutathione peroxidase or glutathione reductase. After 24 h postanoxia, the superoxide dismutase activity modulated by the presence or absence of oxygen returned to control value.
Damage and recovery of RBE4 immortalized rat brain endothelial cells in culture after exposure to free radicals and other oxygen-derived species provides a usefulin vitro model to study anoxia-reoxygenation trauma at the cellular level.
Similar content being viewed by others
References
Aebi, H. Catalase. In: Bergmeyer, H. U., ed.. Methods of enzymatic analysis. Vol. 2. New York: Academic Press; 1984:673–684.
Block, E. R.; Patel, J. M.; Sheridan, N. P. Effect of oxygen and endotoxin on lactate dehydrogenase release, 5-hydroxytryptamine uptake, and antioxidant enzyme activities in endothelial cells. J. Cell. Physiol. 122:240–248; 1985.
Buard, A.; Clément, M.; Bourre, J. M. Developmental changes in enzymatic systems involved in protection against peroxidation in isolated rat brain microvessels. Neurosci. Lett. 141:72–74; 1992.
Cazin, M.; Paluszezak, D.; Bianchi, A., et al. Effects of anaerobiosis upon morphology and energy metabolism of alveolar macrophages cultured in gas phase. Eur. Respir. J. 3:1015–1022; 1990.
Chaudière, J.; Gérard, D.; Clément, M., et al. Induction of selenium-glutathione peroxidase by stimulation of metabolic hydrogen peroxide production in vivo. Bioelectrochem. Bioenerg. 18:247–256; 1987.
Dorovini-Zis, K.; Prameya, R.; Bowman, P. Culture and characterization of microvessel endothelial cells derived from human brain. Lab. Invest. 64:425–436; 1991.
Durieu-Trautmann, O.; Bourdoulous, S.; Roux, F., et al. Immortalized rat brain microvessel endothelial cells: II. Pharmacological characterization. In: Drewes, L. R.; Betz, A. L., eds. Frontiers in cerebral vascular biology: transport and its regulation. New York: Plenum Press; 1992:205–210.
Durieu-Trautmann, O.; Federici, C.; Creminon, C., et al. Nitric oxide and endothelin secretion by brain microvessel endothelia cells: regulation by cyclic nucleotides. J. Cell. Physiol. 155:104–111; 1993.
Essman, W. B.; Wollman, S. B. Free radicals, central nervous system processes and brain functions. In: Das, D. K.; Essman, W. B., eds. Oxygen radicals: systemic events and disease processes. Basel, Switzerland: Karger; 1989:172–192.
Flohe, L.; Otting, F. Superoxide dismutase assays. In: Packer, L., ed. Methods in enzymology, oxygen radicals in biological systems. Vol. 105. New York: Academic Press; 1984:93–104.
Halliwell, B. Reactive oxygen species and the central nervous system. J. Neurochem. 59:1609–1623; 1992.
Housset, B.; Junod, A. F. Effects of culture conditions and hyperoxia on antioxidant enzymes in pig pulmonary artery and aortic endothelium. Biochim. Biophys. Acta 716:283–289; 1982.
Invittox protocol. The frame modified neutral red uptake cytotoxicity test. The ERCATT/FRAME data bank of in vitro techniques in toxicology. Vol. 3a. 1990.
Joó, F. The blood-brain barrier in vitro: the second decade. Neurochem. Int. 23:499–521; 1993.
Jornot, L.; Junod, A. F. Response of human endothelial cell antioxidant enzymes to hyperoxia. Am. J. Respir. Cell Mol. Biol. 6:107–115; 1992.
Kong, X. J.; Fanburg, B. L. Regulation of Cu,Zn-superoxide dismutase in bovine pulmonary artery endothelial cells. J. Cell. Physiol. 153:491–497; 1992.
Niki, E. Antioxidant defenses in eukariotic cells: an overview. In: Poli, G.; Albano, E.; Dianzani, M. U., ed. Free radicals: from basic science to medicine. Basel, Switzerland: Birkhaeuser Verlag; 1993:365–373.
Paglia, D. E.; Valentine, W. N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70:158–169; 1967.
Preston, E.; Sutherland, G.; Finsten, A. Three openings of the blood-brain barrier produced by forebrain ischemia in the rat. Neurosci. Lett. 149:75–78; 1993.
Rapoport, S. I. Blood-brain barrier in physiology and medicine. New York: Raven Press; 1976:129–152.
Roux, F.; Durieu-Trautmann, O.; Bourre, J. M., et al. Immortalized rat brain microvesel endothelial cells: I. Expression of blood-brain barrier markers during angiogenesis. In: Drewes, L. R.; Betz, A. L., ed. Frontiers in cerebral vascular biology: transport and its regulation. New York: Plenum Press; 1992:201–204.
Roux, F.; Durieu-Trautmann, O.; Chaverot, N., et al. Regulation of gamma-glutamyl transpeptidase and alkaline phosphatase activities in immortalized rat brain microvessel endothelial cells. J. Cell. Physiol. 159:101–113; 1994.
Shaffer, J. B.; Treanor, C. P.; Del Vecchio, P. J. Expression of bovine and mouse endothelial cell antioxidant enzymes following TNF-α exposure. Free Radical Biol. & Med. 8:497–502; 1990.
Shatos, M. A.; Doherty, J. M.; Stump, D. C., et al. Oxygen radical generated during anoxia followed by reoxygenation reduce the synthesis of tissue-type plasminogen activator and plasminogen activator-inhibitor-1 in human endothelial cell culture. J. Biol. Chem. 33:20443–20448; 1990.
Smith, L. E.; Sweet, E.; Freedman, S., et al. Alterations in endothelial superoxide dismutase levels as a function of growth state in vitro. Invest. Ophthalmol. & Visual Sci. 33:36–41; 1992.
Starke, P. E.; Farber, J. L. Endogenous defenses against cytotoxicity of hydrogen peroxide in cultured rat hepatocytes. J. Biol. Chem. 260:86–92; 1985.
Tayarani, I.; Cloez, I.; Clément, M., et al. Antioxidant enzymes and related trace elements in aging capillaries and choroid plexus. J. Neurochem. 53:817–824; 1989.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rabin, O., Piciotti, M., Drieu, K. et al. Effect of anoxia and reoxygenation on antioxidant enzyme activities in immortalized brain endothelial cells. In Vitro Cell.Dev.Biol.-Animal 32, 221–224 (1996). https://doi.org/10.1007/BF02722949
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02722949