Summary
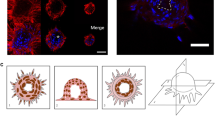
Specific interactions between cells and the extracellular matrix (ECM) in which they are embedded play a vital role in tissue organization. In recent years, many of the individual components of the extracellular matrix have been isolated and their molecular structures elucidated, but the detailed topography of most extracellular matrices, as they are deposited by cells, is still largely unknown. In this study, the insoluble extracellular matrix produced by cultured rat vascular smooth muscle cells has been characterized morphologically using high-resolution electron microscopy of rotary platinum replicas. These cells grew as flat sheets in culture, secreting their matrix laterally and basally. The matrix was composed of a cross-linked fibrillar meshwork. Some fine fibers (10 to 15 nm in diameter) were naked, but most of the filamentous mesh was covered with coarse granular material. Limited digestion with trypsin or pancreatic elastase removed most of this coating, indicating that the granules were glycoproteins and proteoglycans. Another subset of matrix fibrils (20 to 40 nm in diameter) was identified as type I collagen by direct comparison with purified bovine skin collagen. In addition to exposing the underlying filamentous substructure of the matrix, protease treatment also revealed large, straight fiber bundles and globules of amorphous material suspended in the filamentous web. This novel view of a complex matrix promises to provide spatial information that will be useful in future studies of cell interactions with the ECM.
Similar content being viewed by others
References
Adamson, E. D. The effect of collagen on cell division, cellular differentiation, and embryonic development. In: Weiss, J. B.; Jayson, M. I. V., eds. Collagen in health and disease. Edinburgh: Churchill Livingstone; 1982: 218–243.
Aggeler, J.; Kapp, L. N.; Tseng, S. C. G., et al. Regulation of protein secretion in Chinese hamster ovary cells by cell cycle position and cell density. Plasminogen activator, procollagen and fibronectin. Exp. Cell Res. 139: 275–283; 1982.
Aggeler, J.; Takemura, R.; Werb, Z. High-resolution three-dimensional views of membrane-associated clathrin and cytoskeleton in critical-point-dried macrophages. J. Cell Biol. 97: 1452–1458; 1983.
Birk, D. E.; Trelstad, R. L. Extracellular compartments in matrix morphogenesis: collagen fibril, bundle, and lamellar formation by corneal fibroblasts. J. Cell Biol. 99: 2024–2033; 1984.
Bissell, M. J.; Hall, H. G.; Parry, G. How does the extracellular matrix direct gene expression? J. Theor. Biol. 99: 31–68; 1981.
Charonis, A. S.; Tsilibary, E. C.; Yurchenco, P. D., et al. Binding of type IV collagen to laminin. A morphologic study. J. Cell Biol. 100: 1848–1852; 1985.
Chung, E.; Miller, E. J. Collagen polymorphism: characterization of molecules with the chain composition α1 (III)3 in human tissues. Science 183: 1200–1201; 1974.
De Clerck, Y. A.; Jones, P. A. The effect of ascorbic acid on the nature and production of collagen and elastin by rat smoothmuscle cells. Biochem. J. 186: 217–225; 1980.
Dorfman, A. Proteoglycan biosynthesis. In: Hay, E. D., ed. Cell biology of extracellular matrix. New York: Plenum Press; 1981; 115–138.
Erickson, H. P.; Carrell, N.; McDonagh, J. Fibronectin molecule visualized in electron microscopy: a long, thin, flexible strand J. Cell Biol. 91: 673–678; 1981.
Furcht, L. T. Structure and function of the adhesive glycoprotein fibronectin. Mod. Cell Biol. 1: 53–177; 1983.
Furthmayr, H.; Madri, J. A. Rotary shadowing of connective tissue macromolecules. Collagen Relat. Res. 2: 349–363; 1982.
Hassell, J. R.; Robey, P. G.; Barrach, H.-J., et al. Isolation of a heparan sulfate-containing proteoglycan from basement membrane. Proc. Natl. Acad. Sci. USA 77: 4494–4498;1980.
Hay, E. D. Cell and extracellular matrix: their organization and mutual dependence. Mod. Cell Biol. 2: 509–548; 1983.
Heuser, J. E. Three-dimensional visualization of coated vesicle formation in fibroblasts. J. Cell Biol. 84: 560–583; 1980.
Heuser, J. E.; Kirschner, M. W. Filament organization revealed in platinum replicas of freeze-dried cytoskeletons. J. Cell Biol. 86: 212–234; 1980.
Hynes, R. O. Fibronectin and its relation to cellular structure and behavior. In: Hay, E. D., ed. Cell biology of extracellular matrix. New York: Plenum Press; 1981: 295–334.
Jones, P. A. construction of an artificial blood vessel wall from cultured endothelial and smooth muscle cells. Proc. Natl. Acad. Sci. USA 76: 1882–1886; 1979.
Jones, P. A.; Scott-Burden, T.; Gevers, W. Glycoprotein, elastin, and collagen secretion by rat smooth muscle cells. Proc. Natl. Acad. Sci. USA 76: 353–357; 1979.
Jones, P. A.; Werb, Z. Degradation of connective tissue matrices by macrophages. II. Influence of matrix composition on proteolysis of glycoproteins, elastin, and collagen by macrophages in culture. J. Exp. Med. 152: 1527–1536; 1980.
Klein, L.; Itoh, T.; Geil, P. H. Relationship between collagen fibril and subfibril diameters as revealed by transverse freezefracture and −etching techniques. In: Veis, A., ed. The chemistry and biology of mineralized connective tissues. New York: Elsevier/North-Holland; 1981: 129–132.
Kleinman, H. K.; McGarvey, M. L.; Hassell J. R., et al. Basement membrane complexes with biological activity. Biochemistry 25: 312–318; 1986.
Linsenmayer, T. F., Collagen. In: Hay, E. D., ed. Cell biology of extracellular matrix. New York: Plenum Press; 1981: 5–38.
Rapraeger, A.; Jalkanen, M.; Bernfield, M. Cell surface proteoglycan associates with the cytoskeleton at the basolateral cell surface of mouse mammary epithelial cells. J. Cell Biol. 103:2683–2696; 1986.
Scott-Burden, T.; Murray, E.; Diehl, T., et al. Glycosaminoglycan synthesis by smooth muscle cells cultured in the absence and presence of ascorbic acid. Hoppe-Seyler's Z. Physiol. Chem. 364:61–70; 1983.
Sear, C. H. J.; Grant, M. E.; Jackson, D. S.. The nature of the microfibrillar glycoproteins of elastic fibers. A biosynthetic study. Biochem. J. 194:587–598; 1981.
Timpl, R.; Wiedemann, H.; van, Delden, V., et al. A network model for the organization of type IV collagen molecules in basement membranes. Eur. J. Biochem. 120: 203–211; 1981.
Tsilibary, E. C.; Charonis, A. S. The role of the main noncollagenous domain (NCl) in type IV collagen self-assembly. J. Cell Biol. 103:2467–2474; 1986.
Turley, E. A.; Erickson, C. A.; Tucker, R. P. The retention and ultrastructural appearances of various extracellular matrix molecules incorporated into three-dimensional hydrated collagen lattices. Dev. Biol. 109: 347–369; 1985.
Werb, Z.; Banda, M. J.; Jones, P. A. Degradation of connective tissue matrices by macrophages. I. Proteolysis of elastin, glycoproteins. and collagen by proteinases isolated from macrophages. J. Exp. Med. 152: 1340–1357; 1980.
Yamada, K. M. Cell surface interactions. Ann. Rev. Biochem. 52: 761–799; 1983.
Author information
Authors and Affiliations
Additional information
These studies were supported in part by NIH Biomedical Research Support grant S07-RR-05684.
Rights and permissions
About this article
Cite this article
Aggeler, J. Three-dimensional organization of the extracellular matrix secreted by cultured rat smooth muscle cells. In Vitro Cell Dev Biol 24, 633–638 (1988). https://doi.org/10.1007/BF02623600
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02623600