Abstract
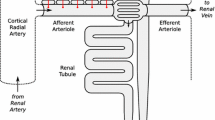
To test whether a mathematical model combining dynamic models of the tubuloglomerular feedback (TGF) mechanism and the myogenic mechanism was sufficient to explain dynamic autoregulation of renal blood flow, we compared model simulations with experimental data. To assess the dynamic characteristics of renal autoregulation, a broad band perturbation of the arterial pressure was employed in both the simulations and the experiments. Renal blood flow and tubular pressure were used as response variables in the comparison. To better approximate the situationin vivo where as large number of individual nephrons act in parallel, each simulation was performed with 125 parallel versions of the model. The key parameters of the 125 versions of the model were chosen randomly within the physiological range. None of the constituent models, i.e., the TGF and the myogenic, could alone reproduce the experimental observations. However, in combination they reproduced most of the features of the various transfer functions calculated from the experimental data. The major discrepancy was the presence of a bimodal distribution of the admittance phase in the simulations. This is not consistent with most of the experimental data, which shows a unimodal curve for the admittance phase. The ability of the model to reproduce the experimental data supports the hypothesis that dynamic autoregulation of renal blood flow is due to the combined action of TGF and the myogenic response.
Similar content being viewed by others
Literature
Aukland, K. and A. H. Øien. 1987. Renal autoregulation: models combining tubuloglomerular feedback and myogenic response.Am. J. Physiol. 252, F768-F784.
Başar, E. and C. Weiss. 1968. Analyse des Frequenzganges druckinduzierter Änderungen des Strömungswiderstandes isolierter Rattennieren.Pflügers Arch. ges. Physiol. 304, 121–135.
Bendat, J. A. and A. G. Piersol. 1971.Random Data: Analysis and Measurement Procedures. New York: John Wiley.
Daniels, F. H., W. J. Arendshorst and R. G. Roberds. 1990. Tubuloglomerular feedback and autoregulation in spontaneously hypertensive rats.Am. J. Physiol. 258, F1479-F1489.
Davis, M. J. and P. J. Sikes. 1990. Myogenic responses of isolated arterioles: test for a ratesensitive mechanism.Am. J. Physiol. 259, H1890-H1990.
Edwards, R. M. 1983. Segmental effects of norepinephrine and angiotensin II on isolated renal microvessels.Am. J. Physiol. 244, F526-F534.
Eggert, P., V. Theiman and C. Weiss. 1979. Periodic changes of blood flow in thein vivo rat kidney.Pflügers Arch. ges. Physiol. 382, 63–66.
Grände, P.-O., J. Lundvall and S. Mellander. 1977. Evidence for a rate-sensitive regulatory mechanism in myogenic microvascular control.Acta physiol. Scand. 99, 432–447.
Grände, P.-O. and S. Mellander. 1978. Characteristics of static and dynamic regulatory mechanisms in myogenic microvascular control.Acta physiol. Scand. 102, 231–245.
Hander, D. R., R. Gilbert and J. H. Lombard. 1987. Vascular smooth muscle cell depolarization and activation in renal arteries on elevation of transmural pressure.Am. J. Physiol. 253, F778-F781.
Holstein-Rathlou, N.-H. 1992. Dynamic aspects of the tubuloglomerular feedback mechanism.Dan. Med. Bull. 39, 134–154.
Holstein-Rathlou, N.-H. and P. P. Leyssac. 1987. Oscillations in proximal intratubular pressure: a mathematical model.Am. J. Physiol. 252, F560-F572.
Holstein-Rathlou, N.-H. and D. J. Marsh 1990. A dynamic model of the tubuloglomerular feedback mechanism.Am. J. Physiol. 258, F1448-F1459.
Holstein-Rathlou, N.-H., A. W. Wagner and D. J. Marsh. 1991. Tubuloglomerular feedback dynamics and renal blood flow autoregulation in rats.Am. J. Physiol. 260, F53-F68.
Kenner, T. and K. Ono. 1971. The low frequency input impedance of the renal artery.Pflügers Arch. ges. Physiol. 324, 155–164.
Kuo, L., M. J. Davis and W. M. Chilian. 1992. Endothelial modulation of arterial tone.NIPS,7, 5–9.
Layton, H. E., E. B. Pitman and L. C. Moore. 1991. Bifurcation analysis of TGF-mediated oscillations in SNGFR.Am. J. Physiol. 261, F904-F919.
Moore, L. C., J. Schnermann and S. Yarimizu. 1979. Feedback mediation of SNGFR autoregulation in hydropenic and DOCA- and salt-loaded rats.Am. J. Physiol. 237, F63-F74.
Press, W. H., B. P. Flannery, S. A. Taukolsky and W. T. Vettering. 1986.Numerical Recipes. New York: Cambridge University Press.
Sakai, T., E. Hallman and D. J. Marsh. 1986. Frequency domain analysis of renal autoregulation in the rat.Am. J. Physiol. 250, F364-F373.
Sakai, T., D. A. Craig, A. S. Wexler and D. J. Marsh. 1986. Fluid waves in renal tubules.Biophys. J. 50, 805–813.
Ursino, M. and G. Fabbri. 1992. Role of the myogenic mechanism in the genesis of microvascular oscillations (vasomotion): analysis with a mathematical model.Microvasc. Res. 43, 156–177.
Young, D. K. and D. J. Marsh. 1981. Pulse wave propagation in rat renal tubules: implications for GFR autoregulation.Am. J. Physiol. 240, F446-F458.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holstein-Rathlou, N.H., Marsh, D.J. A dynamic model of renal blood flow autoregulation. Bltn Mathcal Biology 56, 411–429 (1994). https://doi.org/10.1007/BF02460465
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02460465