Abstract
Background: We analyzed morbidity and mortality, sites of recurrence, and possible prognostic factors in 95 (78 male, 17 female) patients with MPM on phase I–III trials since 1990. A debulking resection to a requisite, residual tumor thickness of ≤ 5 mm was required for inclusion.
Methods: Preoperative tumor volumes were determined by three-dimensional reconstruction of chest computerized tomograms. Pleurectomy (n=39) or extrapleural pneumonectomy (EPP; n=39) was performed. Seventeen patients could not be debulked. Preoperative EPP platelet counts (404,000) and mean tumor volume (491 cm3) were greater than that seen for pleurectomy (344,000, 114 cm3).
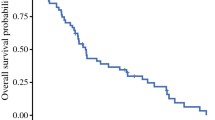
Results: Median survival for all patients was 11.2 months, with that for pleurectomy 14.5 months, that for EPP 9.4 months, and that for unresectable patients 5.0 months. Arrhythmia (n=14; 15%) was the most common complication, and there were two deaths related to surgery (2.0%). Tumor volume of >100 ml, biphasic histology, male sex, and elevated platelet count were associated with decreased survival (p<0.05). Both EPP and pleurectomy had equivalent recurrence rates (27 of 39 [69%] and 31 of 39 [79%], respectively); however, 17 of 27 EPP recurrences as opposed to 28 of 31 pleurectomy recurrences were locoregional (p2=0.013).
Conclusions: Debulking resections for MPM can be performed with low operative mortality. Size and platelet count are important preoperative prognostic parameters for MPM. Patients with poor prognostic indicators should probably enter nonsurgical, innovative trials where toxicity or response to therapy can be evaluated.
Article PDF
Similar content being viewed by others
References
International Mesothelioma Interest Group. A proposed new international TNM staging system for malignant pleural mesothelioma.Chest 1995;108:1122–8.
Pass HW, Temeck BK, Kranda K, Steinberg SM, Pass HI. A phase II trial investigating primary immunochemotherapy for malignant pleural mesothelioma and the feasibility of adjuvant immunochemotherapy after maximal cytoreduction.Ann Surg Oncol 1995;2:214–20.
Kaplan EL, Meier P. Non-parametric estimation from incomplete observation.J Am Stat Assoc 1958;53:457–81.
Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration.Cancer Chem Rep 1966;50:163–70.
Cox D. Regression models and lifetables.J R Stat Soc 1972;34:187–202.
Matthews DE, Farewell VT.Using and understanding medical statistics. Basel: Karger, 1985:148–58.
Jonckheere AR. A distribution-free K-sample test against ordered alternatives.Biometrika 1954;41:133–45.
Temeck BK, Pass HI. Esophagopleural fistula: a complication of photodynamic therapy.South Med J 1995;88:271–3.
Rusch VW, Piantadosi S, Holmes EC. The role of extrapleural pneumonectomy in malignant pleural mesothelioma.J Thorac Cardiovasc Surg 1991;102:1–9.
Pass HI, Pogrebniak HW. Malignant pleural mesothelioma.Curr Probl Surg 1993;30:921–1020.
Rusch VW, Venkatraman E. The importance of surgical staging in the treatment of malignant pleural mesothelioma [Abstract].Am Assoc Thorac Surg 1995;166.
Sugarbaker DJ, Lynch TJ, Strauss GM, et al. Node status has prognostic significance in the multimodality therapy of diffuse, malignant mesothelioma.J Clin Oncol 1993;11:1172–8.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pass, H.I., Kranda, K., Temeck, B.K. et al. Surgically debulked malignant pleural mesothelioma: Results and prognostic factors. Annals of Surgical Oncology 4, 215–222 (1997). https://doi.org/10.1007/BF02306613
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02306613