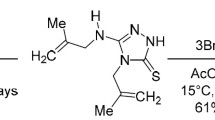
Abstract
2-Allylthiopyridine reacts with iodine to form 3-iodomethyl-2,3-dihydrothiazolo[3,2-a]pyridinium triiodide, but in reaction with bromine a mixture of 3- and 2-bromomethyl-2,3-dihydrothiazolo[3,2-a]pyridinium bromides is obtained.
Similar content being viewed by others
References
K. Undheim and K. R. Reistad,Acta Chem. Scand.,24, 2949 (1970).
A. M. Shestopalov, L. A. Rodinovskaya, Yu. A. Sharanin, and V. P. Litvinov,Khim. Geterotsikl. Soedin., No. 2, 256 (1990).
L. A. Rodinovskaya, Yu. A. Sharanin, A. M. Shestopalov, and V. P. Litvinov,Khim. Geterotsikl. Soedin., No. 6, 805 (1988).
V. P. Litvinov, Yu. A. Sharanin E. E. Apenova, A. M. Shestopalov, V. Yu. Mortikov, V. N. Nesterov, V. E. Shklover, and Yu. T. Struchkov,Khim. Geterotsikl. Soedin., No. 5, 690 (1987).
D. G. Kim, Yu. V. Troitskova, and N. A. Goncharova, in:Abstracts of Papers of the IVth All-Union Scientific Conference on the Chemistry of N-Containing Heterocyclic Compounds [in Russian], Novosibirsk (1987), p. 107.
D. S. Tarbell and M. A. McCall,J. Am. Chem. Soc.,74, 48 (1952).
Additional information
Chelyabinsk State University, Chelyabinsk 454021; Russia
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 334–336, March, 1999.
Rights and permissions
About this article
Cite this article
Kim, D.G. Halocyclization of 2-allylthiopyridine. Chem Heterocycl Compd 35, 290–292 (1999). https://doi.org/10.1007/BF02259357
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02259357