Abstract
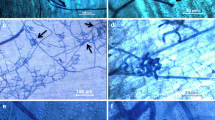
The infection process ofRhizoctonia solani AG-3 was studied on potato sprouts, cv. Bintje, in growth chamber trials at 15 °C. Initially hyphae ofR. solani grew predominantly in the longitudinal direction of the sprouts (runner hyphae). They tended to follow the junctions between epidermis cells as was observed by SEM. The hyphae formed side-branches mainly half-way of the subterranean parts of the sprouts. They branched several times with short swollen cells to form infection cushions. Lesions developed only underneath the infection cushions and were first observed five days after inoculation. The necrotic area was proportional to the area covered with infection cushions on the sprouts. Depth of the lesions could extend up to the vascular bundle. Sprouts were colonized only in healthy tissue in the epidermal layer underneath the infection cushion and in necrotic tissue. A few days after appearance of the lesions,R. solani formed brown, uninfective mycelium on and in the circumference of these lesions.
Aldicarb did not influence any part of the infection process. Ethoprophos delayed the emergence of sprouts, but increased the number of sprouts per tuber. As soon as sprouts had emerged, growth was considerably promoted by ethoprophos. Ethoprophos delayed the appearance of lesions and reduced their size. Oxamyl showed the same effects to a smaller extent.
As the size of lesions appears to be proportional to the size of the infection cushions, any agents that change the size of the infection cushions, such as pesticides or antagonists, may alter the severity of the disease.
Samenvatting
Het infectieproces vanRhizoctonia solani AG-3 werd bestudeerd op aardappelspruiten, cv. Bintje, in een klimaatcel bij 15†C. Aanvankelijk groeide de schimmel met runnerhyfen voornamelijk in de lengterichting van de spruit. Via SEM kon waargenomen worden, dat de hyfen hierbij vooral over de begrenzingen van de epidermiscellen groeiden. Het mycelium vormde veel zijvertakkingen, bestaande uit iets gezwollen korte cellen, welke voornamelijk halverwege op het ondergrondse deel van de spruit gevormd werden. Een dichte massa van deze cellen vormde een infectiekussentje. Lesies, welke vanaf vijf dagen na inoculatie werden waargenomen, bevonden zich slechts onder spruitoppervlak bezet met infectiekussentjes. De lesiegrootte was recht evenredig met het spruitoppervlak dat bezet was met infectiekussentjes. De diepte van de lesies reikte tot aan de vaatbundels. De spruit werd alleen door de schimmel gekoloniseerd in gezond epidermisweefsel onder het infectiekussentje en in necrotisch weefsel. Enkele dagen na verschijning van lesies vormde R.solani bruin, niet infectieus, mycelium op en rondom de lesies.
Aldicarb had geen effect op het infectieproces. Ethoprophos vertraagde de opkomst en verhoogde het aantal tot ontwikkeling gekomen spruiten per knol in gestoomd zand. Direct na opkomst had ethoprophos echter een sterk groeistimulerend effect. Ethoprophos vertraagde de lesievorming en reduceerde de lesiegrootte, vergeleken met onbehandelde planten. Oxamyl vertoonde deze effecten in geringere mate.
Daar de lesiegrootte direct gecorreleerd blijkt met de grootte van het infectiekussentje, mag verwacht worden dat elke beïnvloeding van de ontwikkeling van het mycelium van R.solani, bijvoorbeeld door pesticiden of antagonisten, een verandering van de lesiegrootte ten gevolge heeft.
Similar content being viewed by others
References
Altman, J. & Campbell, C.L., 1977. Effect of herbicides on plant diseases. Annual Review of Phytopathology 15: 361–385.
Bateman, D.F., 1970. Pathogenesis and disease. In: Parmeter, J.R. (Ed.),Rhizoctonia solani, biology and pathology. University of California Press. Berkeley, p. 161–171.
Boosalis, M.G. & Scharen, A.L., 1959. Methods for microscopic detection ofAphanomyces euteiches andRhizoctonia solani and for isolation ofRhizoctonia solani associated with plant debris. Phytopathology 49: 192–198.
Dodman, R.L. & Flentje, N.T., 1970. The mechanism and physiology of plant penetration byRhizoctonia solani. In: Parmeter, J.R. (Ed.),Rhizoctonia solani, biology and pathology. University of California Press. Berkeley, p. 149–160.
Emden, J.H. van, 1965.Rhizoctonia solani; results of recent experiments. European Potato Journal 8: 188–189.
Flentje, N.T., Dodman, R.L. & Kerr, A., 1963. The mechanism of host penetration byThanatephorus cucumeris. Australian Journal of Biological Sciences 16: 784–799.
Fukutomi, M. & Takada, H., 1979. Ultrastructure of infection process ofRhizoctonia solani Kühn in cucumber hypocotyls. Annals of the Phytopathological Society of Japan 45: 453–462.
Heitefuss, R., 1973. Der Einfluss von Herbiziden auf bodenbürtige Pflanzenkrankheiten. Proceedings of the European Weed Research Council, Symposium Herbicides-Soil. p. 98–128.
Hide, G.A. & Corbett, D.C.M., 1974. Field experiments in the control ofVerticillium dahliae andHeterodera rostochiensis on potatoes. Annals of Applied Biology 78: 295–307.
Hofman, T.W. & Bollen, G.J., 1987. Effects of granular nematicides on growth and microbial antagonism toRhizoctonia solani. Netherlands Journal of Plant Pathology 93: 201–214.
Kenning, L.A. & Hanchey, P., 1980. Ultrastructure of lesion formation inRhizoctonia-infected bean hypocotyls. Phytopathology 70: 998–1004.
Leach, L.D. & Garber, R.H., 1970. Control ofRhizoctonia. In: Parmeter, J.R. (Ed.),Rhizoctonia solani, biology and pathology. University of California Press, Berkeley, p. 189–198.
Leach, S.S. & Frank, J.A., 1982. Influence of three systemic insecticides onVerticillium wilt andRhizoctonia disease complex of potato. Plant Disease 66: 1180–1182.
Marshall, D.S. & Rush, M.C., 1980a. Relation between infection byRhizoctonia solani andR. oryzae and disease severity in rice. Phytopathology 70: 941–946.
Marshall, D.S. & Rush, M.C., 1980b. Infection cushion formation on rice sheaths byRhizoctonia solani. Phytopathology 70: 947–950.
Matsuura, K., 1986. Scanning electron microscopy of the infection process ofRhizoctonia solani in leaf sheaths of rice plants. Phytopathology 76: 811–814.
Parmeter, J.R., 1970.Rhizoctonia solani, biology and pathology. University of California Press. Berkeley.
Ruppel, E.G. & Hecker, R.J., 1982. Increased severity ofRhizoctonia root rot in sugar beet treated with systemic insecticides. Crop Protection 1: 75–81.
Scholte, K., 1987. The effect of crop rotation and granular nematicides on the incidence ofRhizoctonia solani in potato. Potato Research 30: 187–199.
Sumner, D.R., 1974. Interactions of herbicides and nematicides with root diseases of snapbean and southern peam. Phytopathology 64: 1353–1358.
Sumner, D.R., 1978. Interaction of herbicides and ethoprop with root diseases of cucumber. Plant Disease Reporter 62: 1093–1097.
Tisserat, N., Altman, J. & Campbell, C.L., 1977. Pesticide-plant disease interactions: the influence of aldicarb on growth ofRhizoctonia solani and damping-off of sugar beet seedlings. Phytopathology 67: 791–793.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hofman, T.W., Jongebloed, P.H.J. Infection process of Rhizoctonia solani on Solanum tuberosum and effects of granular nematicides. Netherlands Journal of Plant Pathology 94, 243–252 (1988). https://doi.org/10.1007/BF01977314
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01977314