Abstract
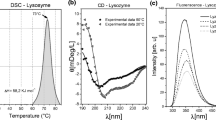
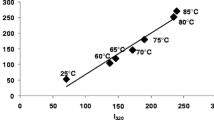
Modifications in the exposure to the solvent of hydrophobic residues, changes in their organization into surface hydrophobic patches, and alterations in the dimerization equilibrium ofβ-lactoglobulin upon thermal treatment at neutralpH were studied. Exposure of tryptophan residues was temperature dependent and was essentially completed on the time scale of seconds. Reorganization of generic hydrophobic protein patches on the protein surface was monitored through binding of 1,8-anilinonaphthalenesulfonate, and was much slower than changes in tryptophan exposure. Different phases in surface hydrophobicity changes were related to the swelling and the subsequent collapse of the protein, which formed a metastable swollen intermediate. Heat treatment ofβ-lactoglobulin also resulted in the formation of soluble oligomeric aggregates. The aggregation process was studied as a function of temperature, demonstrating that (i) dimer dissociation was a necessary step in a sequential polymerization mechanism and (ii) cohesion of hydrophobic patches was the major driving force for aggregation.
Similar content being viewed by others
References
Bonomi, F., and Iametti, S. (1991).Milchwissenschaft 46, 71–74.
Dalgleish, D. G. (1990).Milchwissenschaft 45, 491–494.
Dannenberg, F., and Kessler, H. G. (1988).Milchwissenschaft 43, 139–142.
DeWit, J. N., and Klarenbeek, G. (1984).J. Dairy Sci. 67, 2701–2710.
Elfgman, A. A., and Wheelock, J. V. (1978).J. Dairy Sci. 61, 28–32.
Eynard, L., Iametti, S., Relkin, P., and Bonomi, F. (1992).J. Agric. Food Chem. 40, 1731–1736.
Hambling, S. G., McAlpine, A., and Sawyer, L. (1992). InMilk Proteins (Fox, P. F., ed.), Elsevier, London, pp. 141–190.
Haque, Z., and Kinsella, J. (1988).J. Dairy Res. 55, 67–80.
Hines, M. E., and Foegeding, E. A. (1993).J. Agric. Food Chem. 41, 341–346.
Iametti, S., and Bonomi, F. (1994). IDF Bull.9303.
Kuwajima, K., Yamaya, H., Miwa, S., Sugai, S., and Nagamura, T. (1987).FEBS Lett. 221, 115–118.
Laemmli, U. K. (1970).Nature 227, 680–685.
Mills, O. E. (1976).Biochim. Biophys. Acta 434, 324–332.
Monaco, H. L., Zanotti, G., Spadon, P., Bolognesi, M., Sawyer, L., and Eliopoulos, E. E. (1987).J. Mol. Biol. 197, 695–706.
Pagliarini, E., Iametti, S., Peri, C., and Bonomi, F. (1990).J. Dairy Sci. 73, 41–44.
Papiz, M. Z., Sawyer, L., Eliopoulus, E. E., North, A. C. T., Findlay, J. B. C., Sivaprasadarao, R., Jones, T. A., Newcomer, M. E., and Kraulis, P. J. (1986).Nature 324, 383–385.
Permyakov, Y. A., Yarmolenko, V. V., Kalinichenko, L. P., Morozova, L. A., and Burshtein, E. A. (1982).Biophysics 27, 386–392.
Permyakov, Y. A., Morozova, L. A., and Burshtein, E. A. (1985).Biophys. Chem. 21, 21–31.
Pervaiz, S., and Brew, K. (1985).Science 228, 335–337.
Ptitsyn, O. B. (1992). InProtein Folding (Creighton, T. E., ed.), Freeman, New York, pp. 243–300.
Ptitsyn, O. B., and Semisotnov, G. V. (1991). InConformations and Forces in Protein Folding (Nall, B. A., and Dill, K. A., eds.), AAAS, Washington, D.C., pp. 155–168.
Reddy, I. M., and Kinsella, J. (1990).J. Agric. Food Chem. 38, 366–372.
Relkin, P., Eynard, L., and Launay, B. (1992).Thermochim. Acta 204, 111–121.
Robillard, K. A., Jr., and Wishnia, A. (1972a).Biochemistry 11, 3835–3840.
Robillard, K. A., Jr., and Wishnia, A. (1972b).Biochemistry 11, 3481–3845.
Ruzic, I. (1982).Anal. Chim. Acta 140, 99–113.
Zimmerman, J. K., Barlow, G. H., and Klotz, I. G. (1970).Arch. Biochem. Biophys. 138, 101–109.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cairoli, S., Iametti, S. & Bonomi, F. Reversible and irreversible modifications ofβ-lactoglobulin upon exposure to heat. J Protein Chem 13, 347–354 (1994). https://doi.org/10.1007/BF01901568
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01901568