Abstract
Objective
To test the hypothesis that the rate of degradation of exogenously administered albumin is fater with bolus administration that with continuous infusion and thus that a bolus administration is less efficacious in restoring blood albumin concentration (BAC) in the hypoalbuminemic critically ill pediatric patient.
Design
A prospective, controlled study of two groups of patients.
Setting
Pediatric intensive care unit (PICU) of a children's hospital.
Patients
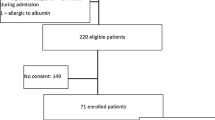
37 cirtically ill hypoalbuminemic patients. (BAC≤2.8 g/dl), in whom no overt protein-losing disease was identified, were divided into two treatment groups and included in a 60-h study.
Interventions
18 patients were given an i.v. bolus of 1 g/kg of 25% albumin over 4 h. This treatment was repeated after 24 and 48 h. Nineteen other patients were given the same dose of 1 g/kg of 25% albumin as a continuous 24-h infusion throughout the 60-h study period. BAC along with sodium, potassium, and total and ionized calcium were measured in the serum of blood samples obtained at predetermined intervals.
Measurement and main results
A 4 h bolus of albumin resulted in an acute rise in BAC, which declined to baseline within 24 h. A continuous infusion resulted in a steady rise in BAC with 24-h levels significantly higher than baseline. The percent change in mean BAC from baseline, calculated at 12-h intervals during the 60-h study period, showed a steady increase in the continuous infusion group with a 34% increase after the first 24 h. In contrast, the 4-h bolus method resulted in major fluctuations in the BAC values with only a 14% increase (p<0.05) after 24 h. Albumin's volume of distribution, half-life and elimination constant, calculated based on blood albumin values during the first 24 h after the bolus administration, were 0.12±0.03 1/kg, 4.6±1.8 h and 0.17±0.06 h−1, respectively. This half-life did not apply to the continuous infusion group as a steady state was not achieved after 30 h (6 half-lives), and BAC continued to rise throughout the 60-h study period. No significant changes in blood electrolytes were observed with either method.
Conclusions
The half-life of exogenous albumin in the critically ill hypoalbuminemic pediatric patient is short if given as a bolus. Continuous infusion therapy appears to be more efficacious in increasing BAC over time, as the half-life with this method appears to be longer.
Similar content being viewed by others
References
Sugerman HJ, Tatum JL, Burke TS, et al. (1984) Gamma scintigraphic analysis of albumin flux in patients with acute respiratory distress syndrome. Surgery 95:674–682
Mobarhan S (1988) The role of albumin in nutritional support. J Am Coll Nutr 6:445–452
Byrne K, Tatum JL, et al. (1992) Increased morbidity with increased pulmonary albumin flux in sepsis-related adult respiratory distress syndrome. Crit Care Med 1: 28–34
Rothschild MA, Ortaz M, Schreiber SS (1988) Serum albumin. Hepatology 8: 385–401
Brinson RR, Kolts BE (1988) Diarrhea associated with severe hypoalbuminemia: a comparison of a peptidebased chemically defined diet and standard enteral alimentation. Cirt Care Med 16: 130–136
Moss G (1982) Malabsorption associated with extreme malnutrition: importance of replacing plasma albumin. J Am Coll Nutr 1: 89–92
Moss G (1967) Postoperative metabolism. The role of the plasma albumin in the enteral absorption of water and electrolytes. Pac Med Surg 75: 355–358
Powanda MC, Moyer E (1981) Plasma proteins and wound healing. Surg Gynecol Obstet 153: 149–755
Mullen JL (1981) Consequences of malnutrition in the surgical patient. Surg Clin North Am 61: 465–498
Howland WS, Schweizer O, Ragasa J, et al. (1976) Colloid oncotic pressure and levels of albumin and total protein during major surgical procedures. Surg Gynecol Obstet 143: 592–596
Emerson T (1989) Unique features of albumin: a brief review. Crit Care Med 7: 690–694
Tullis J (1977) Albumin 1. Backround and use.JAMA 4: 355–358
Tullis J (1977) Albumin 2. Guidelines for clinical use. JAMA 5: 460–462
Guthrie RD, Hines C (1991) Use of intravenous albumin in the critically ill patient. Am J Gastroenterol 86: 255–263
Kaminski MV Jr, Williams SD (1990) Review of the rapid normalization of serum albumin with modified total parenteral solutions. Crit Care Med 3: 327–335
Reinhardt GF, Myscofski JW, Willkens DB, et al. (1980) Incidence and mortality of hypoalbuminemic patients in hospitalized veterans. JPEN 4: 357–359
Mullen JL, Gertner MH, Buzby GP, et al. (1979) Implications of malnutrition in the surgical patient. Arch Surg 114: 121–125
Mullen JL, Buzby GP, Mathews DC, et al. (1980) Reducton of operative morbidity and mortality by combined preoperative and postoperative nutritional support. Ann Surg 192: 604–613
Wojtysiak SL, Brown RO, Roberson D, et al. (1992) Effect of hypoalbuminemia and parenteral nutrition on free water excretion and electrolyte-free water resorption. Crit. Care Med 2: 164–169
Rothschild MA, Ortaz M, Schreiber SS (1972) Albumin Synthesis. N Engl J Med 286: 748–757
Chien S, Sinclair DE, Dellenback RJ, et al. (1964) Effect of endotoxin on capillary permeability to macromolecules. Am J Physiol 207: 518–522
Waldmann TA (1970) Albumin catabolism. In: Roesneoer VM, Ortaz M, Rothschild MA (eds) Albumin structure, function, and uses. Pergamon Press, New York, pp 255–273
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Greissman, A., Silver, P., Nimkoff, L. et al. Albumin bolus administration versus continuous infusion in critically ill hypoalbuminemic pediatric patients. Intensive Care Med 22, 495–499 (1996). https://doi.org/10.1007/BF01712175
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01712175