Abstract
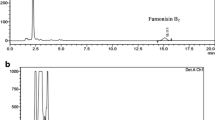
A wide range of moulds representing several genera and species, was recorded in this study from 5 seed samples of each almond, cashew nut, chestnut, hazelnut, pistachio nut and walnut collected from different markets in Ar' Ar, Saudi Arabia. The total counts of fungi were widely fluctuated between 1960–7704 and 1948–7434 colonies/g dry seeds on glucose-Czapek's and glycerol agar media at 28°C, respectively, and represented twenty genera, 53 species and 2 varieties of fungi. The prevalent fungi on the 2 agar media wereAspergillus flavus, A. niger andPenicillium chrysogenum. On glucose-Czapek's agar,Rhizopus stolonifer andAspergillus flavus var.columnaris were isolated from all 6 kinds of nut,A. parasiticus from 5 kinds andA. fumigatus from 4 kinds with high frequencies.Eurotium species were completely absent on glucose-Czapek's agar but they were isolated in high frequency from all kinds of nut on glycerol agar medium. The different nut samples were analyzed by thin layer chromatography for the presence of aflatoxins B1, B2, G1 & G2, citrinin, ochratoxins, patulin, sterigmatocystin, diacetoxyscirpenol, T-2 toxin and zearalenone. Aflatoxins B1 & G1 were detected in 3 out of the 5 samples tested of chestnut at concentrations ranging between 20 to 60 µg/kg. All other samples of almond, cashew nut, hazelnut, pistachio nut, and walnut that were analyzed were mycotoxin free.
Similar content being viewed by others
References
Abdel-Hafez AH, Saber SM. Mycoflora and mycotoxin of hazelnut (Corylus avellana L.) and walnut (Juglans regia L.) seeds in Egypt. Zentralbl Mikrobiol 1993; 148: 137–48.
Burdaspal PA, Gorostidi A, Tejedor MC. A survey of the occurrence of aflatoxins in edible nuts in Spain. Abstracts, International Symposium and Workshop on Food Contamination ‘Mycotoxins and Phycotoxins’, 4–15 November 1990, Cairo, Egypt.
El-Magraby OMO, El-Maraghy SSM. Mycoflora and mycotoxins of peanut (Arachis hypogaea L.) seeds in Egypt, I: Sugar fungi and natural occurrence of mycotoxins. Mycopathologia 1987; 98: 165–70.
El-Magraby OMO, El-Maraghy SSM, Mycoflora and mycotoxins of peanut (Arachis hypogaea L.) seeds in Egypt, III: Cellulose decomposing and mycotoxin producing fungi. Mycopathologia 1988; 104:19–24.
FAO. Food and nutrition paper, No. 13, Perspective on mycotoxin. Rome: Food and Agricultural Organization of the United Nations, 1979.
Jimenez M, Mateo R, Querol A, Huerta T, Hernandez E. Mycotoxins and mycotoxigenic moulds in nuts and sunflower seeds for human consumption. Mycopathologia 1991; 115: 122–28.
Stoloff L. Occurrence of mycotoxins in foods and feeds. In J.V. Rodricks (ed.) Mycotoxins and other fungal related food problems [Advances in Chemistry Series, No. 149]. Washington, DC: American Chemical Society, 1976.
Christensen CM. Influence of small difference in moisture content upon the invasion of hard red winter wheat byAspergillus restrictus andA. repens. Cereal Chem 1963; 40: 385–90.
Moubasher AH, El-Naghy MA, Abdel-Hafez SH. Fungal flora of three grains in Egypt. Mycopath et Mycol Appl 1972; 47: 261–74.
Al-Doory Y. Laboratory medical mycology. London: Lea and Febiger, 1980, 410 pp.
Domsch KH, Gams W, Anderson TH. Compendium of soil fungi. New York: Academic Press, 1980, 859 pp.
Ellis MB. More dematiaceous hyphomycetes. Kew (UK): IMI, 1976, 507 pp.
Kozakiewicz Z.Aspergillus species on stored products. CAB International Mycological Institute. Mycological Papers 1989; 161:1–188.
Onions AHS, Allsopp D, Eggins HOW, Smith's introduction to industrial mycology. London: Edward Arnold, 1981, 398 pp.
Pitt JI. The genusPenicillium and its teleomorphic statesEupenicillium andTalaromyces. London: Academic Press, 1979, 634 pp.
Pitt JI. A laboratory guide to commonPenicillium species. Kew (UK): IMI, 1985, 184 pp.
Raper KB, Fennell DI. The genusAspergillus. Baltimore (USA): Williams and Wilkins, 1965, 686 pp.
Samson RA., Van Reenen-Hoekstra ES. Introduction to food-borne fungi. Baarn, The Netherlands, Centraalbureau voor Schimmelcultures, 1988.
Sivanesan A. Graminicolous species ofBipolaris, Curvularia, Drechslera, Exserohilum and their teleomorphs. CAB International Mycological Institute. Mycological Papers 1987; 158: 1–261.
AOAC. Association of Official Analytical Chemists. Official methods of analysis, 13th ed., Washington, DC 1980, 429 pp.
Gimeno A. Thin layer chromatographic determination of aflatoxins, ochratoxins, sterigmatocystin, zearalenone citrinin, T-2 toxin, diacetoxyscirpenol, penicillic acid patulin and penitrem A. J AOAC 1979; 62: 579–85.
Roberts BA, Patterson DSP. Detection of twelve mycotoxins in mixed animal feedstuffs, using a novel membrane cleanup procedure. J AOAC 1975; 58: 1178–81.
Scott PM, Lawrence JW, Van Walbeak W. Detection of mycotoxins by thin-layer chromatography. Application to screening of fungal extracts. Appl Microbiol 1970; 20:839–42.
Jones BD. Method of aflatoxin analysis G. 70. London: Tropical Products Institute, 1972, 58 pp.
Przybylski W. Formation of derivatives of aflatoxins on TLC plates. J AOAC 1975; 58:163–64.
Hanlin RT. Fungi in developing peanut fruits. Mycopath et Mycol Appl 1969; 38: 99–101.
Moubasher AH, El-Hissy FT, Abdel-Hafez SH, Hassan SKM. The mycoflora of peanuts in Egypt. Mycopathologia 1979; 68: 39–46.
Jackson CR. A field study of fungal associations on peanut fruit. Ca Agr Exp Sta Bull 1968; 26: 29.
Norton DC, Menon SK, Flangas AL. Fungi associated with unblemished spanish peanuts in Texas. Pl Dis Rep 1956; 40: 374–76.
Hocking AD, Pitt JI. Dichloran-Glycerol medium for enumeration of xerophilic fungi from low-moisture foods. Appl Environ Microbiol 1980; 39; 488–92.
Abdel-Hafez SH, Moubasher AH, Barakat A. Keratinophilic fungi and other moulds associated with air dust particles from Egypt. Folia Microbiol 1990; 35: 311–25.
Bullermall LB. Mycotoxins and Food Safety. Food Technology 1986; 40: 59–66.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abdel-Gawad, K.M., Zohri, A.A. Fungal flora and mycotoxins of six kinds of nut seeds for human consumption in Saudi Arabia. Mycopathologia 124, 55–64 (1993). https://doi.org/10.1007/BF01103057
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01103057