Abstract
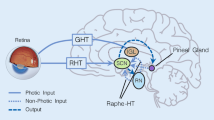
The light/dark cycle to which animals, and possibly humans, are exposed has a major impact on their physiology. The mechanisms whereby specific tissues respond to the light/dark cycle involve the pineal hormone melatonin. The pineal gland, an end organ of the visual system in mammals, produces the hormone melatonin only at night, at which time it is released into the blood. The duration of elevated nightly melatonin provides every tissue with information about the time of day and time of year (in animals that are kept under naturally changing photoperiods). Besides its release in a circadian mode, melatonin is also discharged in a pulsatile manner; the physiological significance, if any, of pulsatile melatonin release remains unknown. The exposure of animals including man to light at night rapidly depresses pineal melatonin synthesis and, therefore, blood melatonin levels drop precipitously. The brightness of light at night required to depress melatonin production is highly species specific. In general, the pineal gland of nocturnally active mammals, which possess rod-dominated retinas, is more sensitive to inhibition by light than is the pineal gland of diurnally active animals (with cone-dominated retinas). Because of the ability of the light/dark cycle to determine melatonin production, the photoperiod is capable of influencing the function of a variety of endocrine and non-endocrine organs. Indeed, melatonin is a ubiquitously acting pineal hormone with its effects on the neuroendocrine system having been most thoroughly investigated. Thus, in nonhuman photoperiodic mammals melatonin regulates seasonal reproduction; in humans also, the indole has been implicated in the control of reproductive physiology.
Similar content being viewed by others
References
Arendt J (1988) Melatonin and the human circadian system. In: Miles A, Philbrick DRS, Thompson C (eds) Melatonin-clinical perspectives. Oxford University Press, Oxford, pp 43–61
Bittman EL (1984) Melatonin and photoperiodic time measurement: Evidence from rodents and ruminants. In: Reiter RJ (ed) The pineal gland. Raven, New York, pp. 155–192
Bittman EL, Karsch FJ (1984) Nightly duration of pineal melatonin secretion determines the reproductive response to inhibitory daylength. Biol Reprod 30:585–592
Brainard GC, Richardson BA, King TS, Matthews SA, Reiter RJ (1983) The suppression of pineal melatonin content and Nacetyltransferase activity by different light irradiances in the Syrian hamster: A dose-response study. Endocrinology 113:293–296
Brainard GC, Richardson BA, King TS, Reiter RJ (1984) The influence of different light spectra on the suppression of pineal melatonin content in the Syrian hamster. Brain Res 294:333–339
Brainard GC, Lewy AJ, Menaker M, Frederickson RH, Miller LS, Weleber RG, Cassone V, Hudson D (1985) Effect of light wavelength on the suppression of nocturnal plasma melatonin in human volunteers. Ann NY Acad Sci 453:376–378
Brainard GC, Podolin PL, Leivy SW, Rollag MD, Cole C, Barker FM (1986) Near-ultraviolet radiation suppresses pineal melatonin content. Endocrinology 119:2201–2205
Cardinali DP, Vacas MI, Lowenstein PR (1985) Mechanisms for controlling pineal activity: An overview. Adv Biosci 53:1–12
Cardinali DP, Vacas MI, Rosenstein RE, Etchegoyen GS, Keller Sarmiento MI, Salveyra CG, Pereyra EN (1987) Multifactorial control of pineal melatonin synthesis: An analysis through binding sites. Adv Pineal Res 2:51–66
Carter DS, Goldman BD (1983) Antigonadal effects of timed melatonin infusion in pinealectomized male Djungarian hamsters (Phodopus sungorus): Duration is the critical factor. Endocrinology 113:1261–1268
Cozzi B, Ravault JP, Ferrandi B, Reiter RJ (1988) Melatonin concentration in cerebral vascular sinuses of sheep and evidence for its episodic release. J Pineal Res 5:535–543
Deveson SL, Arendt J, Forsyth IA (1990) Sensitivity of goats to a light pulse during the night as assessed by suppression of melatonin concentrations in the plasma. J Pineal Res 8:169–177
Ebadi M, Hexum TD, Pfeiffer RF, Govitrapong P (1989) Pineal and retinal peptides and their receptors. Pineal Res Rev 7:1–156
English J, Arendt J, Paulton A, Symons AM (1987) Short-term variations of circulating melatonin in the ewe. J Pineal Res 4:359–366
Gonzalez-Brito A, Troiani ME, Menendez-Pelaez A, Delgado MJ, Reiter RJ (1990) mRNA transcription determined the lag period for the induction of pineal melatonin synthesis in the Syrian hamster pineal gland. J Cell Biochem 44:55–60
Hoffmann K, Illnerova H, Vanecek J (1985) Change in the duration of night-time melatonin peak may be a signal driving photoperiod response in the Djungarian hamster (Phodopus sungorus). Neurosci Lett 56:39–42
Kappers JA (1960) The development, topographical relations and innervation of the epiphysis cerebri in the albino rat. Z Zellforsch 52:163–215
Klein DC (1985) Photoperiod regulation of the mammalian pineal gland. In: Photoperiodism, melatonin and the pineal. Pitman, London, pp 38–51
Lang U, Sizonenko P (1988) Melatonin and human adrenocortical function. In: Miles A, Philbrick DRS, Thompson C (eds) Melatonin — clinical perspective. Oxford University Press, Oxford, pp 79–91
Lerchl A, Nonaka KO, Stokkan K-A, Reiter RJ (1990) Marked rapid alterations in nocturnal pineal serotonin metabolism in mice and rats exposed to weak intermittent magnetic fields. Biochem Biophys Res Commun 169:102–108
Lerchl A, Nonaka KO, Reiter RJ (1991) Pineal gland “magnetosensitivity” to static magnetic fields is a consequence of induced electric currents (eddy currents). J Pineal Res 10:109–116
Lewy AJ (1983) Biochemistry and regulation of mammalian melatonin production. In: Relkin R (ed) The pineal gland. Elsevier, New York, pp 77–128
Lewy AJ, Wehr TA, Goodwin FK, Newsome DA, Markey SP (1980) Light suppresses melatonin secretion in humans. Science 210:1267–1269
Lincoln GA, Short RV (1980) Seasonal breeding: Nature's contraceptive. Rec Prog Horm Res 36:1–52
Maestroni GJM, Conti A, Pierpaoli W (1989) Melatonin, stress, and the immune system. Pineal Res Rev 7:203–226
McIntyre IM, Norman TR, Burrows GD, Armstrong SM (1989) Human melatonin suppression by light is intensity dependent. J Pineal Res 6:149–156
Menendez-Pelaez A, Buzzell GR, Nonaka KO, Reiter RJ (1990) In vivo administration of isoproterenol or forskolin during the light phase induces increases in the melatonin content of the Syrian hamster pineal gland without a rise in N-acetyltransferase activity. Neurosci Lett 110:314–318
Pang SF, Yip PCY (1988) Secretory pattern of pineal melatonin in rats. J Pineal Res 5:279–292
Pangerl B, Pangerl A, Reiter RJ (1990) Circadian variations of adrenergic receptors in the mammalian pineal gland. A review. J Neural Transm 81:17–30
Pickard GE (1982) The afferent connections of the suprachiasmatic nucleus of the golden hamster with emphasis on the retinohypothalamic projection. J Comp Neurol 211:65–83
Reiter RJ (1973) Comparative physiology: Pineal gland. Annu Rev Physiol 35:305–328
Reiter RJ (1975) The pineal gland and seasonal reproductive adjustments. Int J Biometeorol 19:282–288
Reiter RJ (1981) The mammalian pineal gland: Structure and function. Am J Anat 162:287–313
Reiter PJ (1983) Pineal gland: An intermediary between the environment and the endocrine system. Psychoneuroendocrinology 8:31–40
Reiter RJ (1985) Action spectra, dose-response relationships and temporal aspects of light's effects on the pineal gland. Ann NY Acad Sci 453:215–230
Reiter RJ (1986a) The pineal gland: An important link to the environment. News Physiol Sci 1:202–205
Reiter RJ (1986b) Normal patterns of melatonin levels in the pineal gland and body fluids of humans and experimental animals. J Neural Transm (Suppl) 21:35–54
Reiter RJ (1987) The melatonin message: Duration versus coincidence hypotheses. Life Sci 46:2119–2131
Reiter RJ (1988) Comparative aspects of pineal melatonin rhythms in mammals. In: Atkins H (ed) ISI atlas of science: animal and plant sciences, vol 1, no 2. ISI Press, Philadelphia, pp 111–116
Reiter RJ (1990) Signal transduction mechanisms related to melatonin synthesis in the mammalian pineal gland. Adv Pineal Res 4:27–38
Reiter RJ (1991a) Pineal gland: Interface between the photoperiodic environment and the endocrine system. Trends Endocrinol Metab 2:13–19
Reiter RJ (1991b) Pineal melatonin: Cell biology of its synthesis and of its physiologic interactions. Endocrine Rev 12:151–180
Reiter RJ (1991c) Dynamic organization of endocrine networks: The neuroendocrine-reproductive axis and the pineal gland as examples. Ann NY Acad Sci 618:11–27
Reiter RJ, Hurlbut EC, Brainard GC, Steinlechner S, Richardson BA (1983) Influence of light irradiance on hydroxyindole-O-methyltransferase activity, serotonin N-acetyltransferase activity, and radioimmunoassayable melatonin levels in the pineal gland of the diurnally active Richardson's ground squirrel. Brain Res 188:151–157
Reiter RJ, Joshi BN, Heinzeller T, Nürnberger F (1986) A single 1 or 5 second light pulse at night inhibits hamster pineal melatonin. Endocrinology 118:1906–1909
Robinson JE (1987) Photoperiodic and steroidal regulation of the luteinizing hormone pulse generator in ewes. In: Crowley WF Jr, Hofler JG (eds) The episodic secretion of hormones. Wiley, New York, pp 159–166
Rollag MD, Panke ES, Trakulrungsi W, Trakulrungsi C, Reiter RJ (1980) Quantification of daily melatonin synthesis in the hamster pineal gland. Endocrinology 106:232–236
Romero JA, Zatz M, Axelrod J (1975) Beta-adrenergic stimulation of pineal N-acetyltransferase: Adenosine 3′, 5′-cyclic monophosphate stimulates both RNA and protein synthesis. Proc Natl Acad Sci USA 72:2107–2111
Rusak B, Robertson HA, Wisden W, Hunt SP (1990) Light pulses that shift rhythms induce gene expression in the suprachiasmatic nucleus. Science 248:1237–1240
Sack RL, Lewy AJ (1988) Melatonin and major affective disorder. In: Miles A, Philbrick DRS, Thompson C (eds) Melatoninclinical perspectives. Oxford University Press, Oxford, pp 205–227
Santana C, Menendez-Pelaez A, Reiter RJ, Guerrero JM (1990) Treatment with forskolin for 8 h during the day increases melatonin synthesis in the Syrian hamster pineal gland in organ culture: The long lag period is required for RNA synthesis. J Neurosci Res 25:545–548
Stetson MH, Watson-Whitmyre M (1984) Physiology of the pineal and its hormone melatonin in annual reproduction in rodents. In: Reiter RJ (ed) The pineal gland. Raven, New York, pp 109–132
Stumpf WE (1988a) The endocrinology of sunlight and darkness. Naturwissenschaften 75:247–251
Stumpf WE (1988b) Vitamin D — Soltriol, the heliogenic steroid hormone: Somatrophic activator and modulator. Histochemistry 89:209–219
Sugden DS (1989) Melatonin biosynthesis in the mammalian pineal gland. Experientia 45:922–928
Vivien-Roels B, Pevet P (1983) The pineal gland and the synchronization of reproductive cycles with variations of the environmental climatic conditions, with special reference to temperature. Pineal Res Rev 1:92–144
Vriend J, Steiner M (1988) Melatonin and thyroid function. In: Miles A, Philbrick DRS, Thompson C (eds) Melatonin — clinical perspectives. Oxford University Press, Oxford, pp 92–117
Waldhauser F, Waldhauser M (1988) Melatonin and ageing. In: Miles A, Philbrick DRS, Thompson C (eds) Melatonin-clinical perspectives. Oxford University Press, Oxford, pp 174–189
Wilson BW, Leung F, Buschbom R, Stevens RG, Anderson LE, Reiter RJ (1988) Electric fields, the pineal gland and cancer. In: Gupta D, Attanasio A, Reiter RJ (eds The pineal gland and cancer. Brain Research Promotion, Tübingen, pp 245–259
Wurtman RJ (1975) The effects of light on man and other mammals. Annu Rev Physiol 37:467–483
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reiter, R.J. Neuroendocrine effects of light. Int J Biometeorol 35, 169–175 (1991). https://doi.org/10.1007/BF01049063
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01049063