Abstract
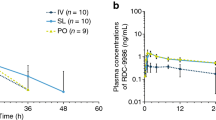
Imipramine hydrochloride (IMI) was administered to 12 healthy volunteers on three occasions in random sequence: 12.5 mg IV, 50 mg orally after overnight fast, and 50 mg orally 30 min after eating a standardized breakfast. IMI concentrations were measured by gas-liquid chromatography using nitrogen-phosphorous detection and pharmacokinetic and bioavailability parameters determined by iterative nonlinear least-squares regression analysis. After IV administration, mean kinetic variables were: volume of distribution, 21.0 l/kg; total clearance, 12.8 ml/min per kg, and elimination half-life, 21.2 h. Mean absolute bioavailability of IMI in the fasting state was 43.6%. When IMI was administered immediately after the standardized meal, absolute bioavailability was 44.1%. After oral administration, the time to peak IMI level was not changed by concurrent food ingestion (2.8 vs 3.2 h after dosage), and the peak IMI concentration was no different (35 vs 30 ng/ml). Thus concurrent food ingestion has no effect on IMI absolute bioavailability, peak concentration attained after oral dosing, or the time to peak concentration.
Similar content being viewed by others
References
Abernethy DR, Greenblatt DJ, Shader RI (1981) Tricyclic antidepressant determination in human plasma by gas-liquid chromatography using nitrogen-phosphorous detection. Pharmacology 23:57–63
Amsterdam J, Brunswick D, Mendels J (1980) The clinical application of tricyclic antidepressant pharmacokinetics and plasma levels. Am J Psychiatry 137:653–662
Anonymous (1959) Weights of insured persons in the United States associated with lowest mortality. Stat Bull Metrop Life Insur Co 40:Nov–Dec
Brinkshulte M, Breyer-Pfaff U (1979) Binding of tricyclic antidepressants and perazine to human plasma. Naunyn-Schmiedeberg's Arch Pharmacol 308:1–7
Brunswick DJ, Mendels J (1977) Reduced levels of tricyclic antidepressants in plasma from vacutainers. Comm Psychopharmacol 1:131–134
Evans GH, Nies AS, Shand DG (1973) The disposition of propranolol III: Decreased half-life and volume of distribution as a result of plasma binding in man, monkey, dog, and rat. J Pharmacol Exp Ther 186:114–122
Gram LF, Christiansen J (1975) First-pass metabolism of imipramine in man. Clin Pharmacol Ther 17:555–563
Greenblatt DJ, Koch-Weser J (1975) Clinical pharmacokinetics. N Engl J Med 293:702–705, 964–970
Jorgenson OS, Lober M, Christiansen J, Gram LF (1980) Plasma concentration and clinical effect of imipramine treatment of childhood enuresis. Clin Pharmacokinet 5:386–393
Kristensen CB, Gram LF (1982) Equilibrium dialysis for determination of protein binding in imipramine: evaluation of a method. Acta Pharmacol Toxicol 50:130–136
Kuhn R (1958) The treatment of depressive states with G22355 (imipramine hydrochloride). Am J Psychiatry 115:459–464
Loo JCK, Riegelman S (1970) Assessment of pharmacokinetic constants from post-infusion blood curves obtained after i.v. infusion. J Pharm Sci 59:53–55
Marquardt DW (1963) An algorithm for least-squares estimation of non-linear parameters. J Soc Ind Appl Math 11:431–441
McAllister RG (1982) Clinical pharmacology of slow channel blocking agents. Prog Cardiovasc Dis 25:83–102
Melander A, Danielson K, Schersten B, Wahlin E (1977a) Enhancement of the bioavailability of propranolol and metoprolol by food. Clin Pharmacol Ther 22:108–112
Melander A, Berlin-Wahlen A, Bodin NO, Danielson K, Gustafsson B, Lindgren S, Westerlund D (1977b) Bioavailability of d-propoxyphene, acetylsalicyclic acid, and phenazone in a combination tablet (Doleron): interindividual variation and influence of food intake. Acta Med Scand 202:119–124
Melander A, Danielson K, Hanson A, Rudell B, Schersten B, Thulin T, Wahlin E (1977c) Enhancement of hydralazine bioavailability by food. Clin Pharmacol Ther 22:104–107
Newton R (1981) The side effect profile of trazodone in comparison to an active control and placebo. J Clin Psychopharmacol 1 (6 Supp 1):895–935
Rawlins MD, Henderson DB, Hijab AR (1977) Pharmacokinetics of paracetamol (acetaminophen) after intravenous and oral administration. Eur J Clin Pharmacol 11:283–286
Schomerus M, Spiegelhalder B, Stieren B, Eichelbaum M (1976) Physiological disposition of verapamil. Cardiovase Res 10: 605–612
Shand DG, Kornhauser DM, Wilkinson GR (1975) Effects of route of administration and blood flow on hepatic drug elimination. J Pharmacol Exp Ther 195:424–432
Sjoqvist F, Bertilsson L, Asberg M (1980) Monitoring tricyclic antidepressants. Ther Drug Monit 2:85–93
Svensson CK, Edwards DJ, Mauriello PM, Barde SH, Foster AC, Middleton E, Lalka D (1983) Effect of food on hepatic blood flow: implications in the food effect phenomenon. Clin Pharmacol Ther 34:316–323
Usanis RA (1972) NLIN-Nonlinear least-squares estimate of parameters (Library Services Document No. LSR-089-1: Research Triangle Park, N.C., Triangle Universities Computation Center
Wagner JG (1975) Fundamentals of clinical pharmacokinetics. Drug Intelligence Publications Hamilton, Ill
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abernethyl, D.R., Divoll, M., Greenblatt, D.J. et al. Absolute bioavailability of imipramine: Influence of food. Psychopharmacology 83, 104–106 (1984). https://doi.org/10.1007/BF00427432
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00427432