Summary
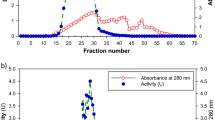
The enzyme laccase has been partially purified from the culture fluid of Ganoderma lucidum by acetone precipitation, ammonium sulphate fractionation and adsorption on alumina Cγ gel. The enzyme has been shown to be specific for ortho and para hydroxyphenolic compounds, having Km values of 5.5×10-5 M and 2.86×10-5 M for catechol and hydroquinone respectively. The optimum pH for the oxidation of catechol and hydroquinone are 5.4 and 5.0 respectively. The enzyme is inactivated above 60°C and is inhibited by enzyme inhibitors and metal chelating agents like azide, cyanide etc.
Similar content being viewed by others
References
Ashraf, M., Frieden, E.: A spectrophotometric method for the determination of catecholase activity of tyrosinase and some of its applications. J. Amer. chem. Soc. 79, 4854–4858 (1957).
Bateman, D. F., Miller, R. L.: Pectic enzymes in tissue degradation. A. Rev. Phytopath. 4, 119–146 (1966).
Blumberg, W. E., Levine, W. G., Margolis, S., Peisach, J.: On the nature of copper in two proteins obtained from Rhus vernicifera latex. Biochem. biophys. Res. Commun. 15, 277–283 (1964).
Fåhraeus, G.: Monophenolase and polyphenolase activity of fungal laccase. Biochim. biophys. Acta (Amst.) 54, 192–194 (1961).
—, Ljunggren, H.: Substrate specificity of a purified fungal laccase. Biochim. biophys. Acta (Amst.) 46, 22–32 (1961).
—, Reinhammer, B.: Large scale production and purification of laccase from cultures of the fungus Polyporus versicolor and some properties of laccase A. Acta chem. scand. 21, 2367–2378 (1967).
Gregg, D. C., Miller, W. H.: A laccase from wild mushroom, Russula foetens. J. Amer. chem. Soc. 62, 1374–1379 (1940).
Husain, A., Kelman, A.: Tissue is disintegrated. In: Plant pathology, Vol. I, pp. 143–188. J. G. Horsefall and A. E. Dimond, eds. New York: Academic Press 1959.
Lalitha Kumari, H.: In: Studies on Areca catechu, Linn. with special reference to Ganoderma lucidum, a fungal pathogen of arecanut palm. Ph.D. Thesis. Submitted to Indian Institute of Science. Bangalore, India (1968).
Law, K.: Laccase and tyrosinase in some wood rotting fungi. Ann. Bot. 19, 561–570 (1955).
Levine, W. G.: Laccase a review. In: The biochemistry of copper. J. Peisach, P. Aisen, and W. E. Blumberg, eds. New York: Academic Press 1966.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the Folin-Phenol reagent. J. biol. Chem. 193, 265–275 (1951).
Malmström, B. G., Reinhammer, B., Vanngard, T.: Two forms of copper (11) in fungal laccase. Biochim. biophys. Acta (Amst.) 156, 67–76 (1968).
Molitoris, H. P., Esser, K.: Die Phenoloxydasen des Ascomyceten Podospora anserina. V. Eigenschaften der Laccase nach weiterer Reinigung. Arch. Mikrobiol. 72, 267–296 (1970).
Nakamura, T.: Purification and physico-chemical properties of laccase. Biochim. biophys. Acta (Amst.) 30, 44–52 (1958).
Neufeld, H. A., Latterell, F. M., Green, L. F., Weintraub, R. L.: Oxidation of meta-polyhydroxyphenols by enzymes from Piricularia oryzae and Polyporus versicolor. Arch. Biochem. 76, 317–327 (1958).
Omura, T.: Studies on laccases of lacquer trees. J. Biochem. (Tokyo) 50, 264–272 (1961).
Rösch, R.: Über die intracellulären Polyphenoloxydasen der Braunfäunfäulepilze. IV. Mitteilung. Untersuchungen über Vorkommen und Substratspezifität von Tyrosinase und intracellulärer Laccase. Arch. Mikrobiol. 54, 80–90 (1966).
Sevčik, V.: Occurrence of phenol oxidase in Streptomyces antibioticus producing actinomycin B. Nature (Lond.) 179, 1191–1192 (1957).
Walch, H.: Bildung und Nachweis von Phenoloxydasen bei einigen Ganodermaarten (Lackporlinge). Arch. Mikrobiol. 60, 314–325 (1968).
Wood, R. K. S.: Pectic and cellulolytic enzymes in plant diseases. A. Rev. Plant Physiol. 11, 299–322 (1960).
Yakushiji, E.: Phenolases. Acta phytochim. (Tokyo) 10, 63–80 (1937).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumari, H.L., Sirsi, M. Purification and properties of laccase from Ganoderma lucidum . Archiv. Mikrobiol. 84, 350–357 (1972). https://doi.org/10.1007/BF00409083
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00409083