Summary
1. Previous studies have concluded that the timing of the locomotor rhythm can be strongly influenced by input from group Ib afferents from leg extensor muscles (Duysens and Pearson 1980; Conway et al. 1987). The main objective of the present study was to obtain additional evidence for this conclusion by examining the characteristics of entrainment of the locomotor rhythm by rhythmic stimulation of group I afferents and by rhythmic force pulses in the ankle extensor muscles. 2. A reduced, non-immobilized preparation was developed in spinal cats that allowed isometric contractions of ankle extensor muscles to be elicited by ventral root stimulation during the expression of locomotor activity. The same preparation was used to examine the influence of electrically stimulating group I afferents from the ankle extensors and the effect of rhythmically stretching these muscles. The locomotor rhythm was initiated by sustained mechanical stimulation of the perineum following the administration of Clonidine and, in some preparations, Naloxone. 3. The timing of the onset of flexor burst activity was examined during entrainment with saw-tooth and ramp-and-hold stretches of the ankle extensor muscles. Flexor bursts were initiated about 200 ms following the release from the stretch, and this latency was independent of the entrainment frequency. 4. The locomotor rhythm was readily entrained by rhythmic contractions of the ankle extensor muscles produced by ventral root stimulation provided the magnitude of the contractions was greater than about 10N. Repetitive stimulation of group I muscle afferents from the ankle extensors also entrained the locomotor rhythm, with the timing of motor activity being similar to that during entrainment with rhythmic muscle contractions. Burst activity in the ipsilateral extensors was coincident with the stimulus trains in both cases. This similarity argues for entrainment being produced mainly by input from group Ib afferents. 5. The functional implication of the results of this and previous studies is that input from group Ib afferents during the stance phase of walking acts to inhibit generation of flexor burst activity and to promote extensor activity. The proposal that a decline in Ib activity near the end of the stance phase is involved in regulating the stance to swing transition is discussed.
Similar content being viewed by others
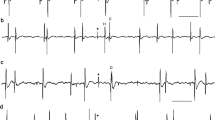
References
Appenteng K, Prochazka A (1984) Tendon organ firing during active muscle lengthening in awake, normally behaving cats. J Physiol 353: 81–91
Barbeau H, Rossignol S (1987) Recovery of locomotion after chronic spinalization in the adult cat. Brain Res 412: 84–95
Conway BA, Hultborn H, Kiehn O (1987) Proprioceptive input resets central locomotor rhythm in the spinal cat. Exp Brain Res 68: 643–656
Duysens JD, Pearson KG (1980) Inhibition of flexor burst generation by loading ankle extensor muscles in walking cats. Brain Res 187: 321–332
Eccles JC, Eccles RM, Lundberg A (1957) Synaptic actions on motoneurones in relation to the two components of the group I muscle afferent volley. J Physiol 136: 527–546
Edgley SA, Jankowska E (1987) An interneuronal relay for group I and II muscle afferents in the midlumbar segments of the cat spinal cord. J Physiol 389: 647–674
Grillner S (1973) Locomotion in the spinal cat. In: Stein RB, Pearson KG, Smith RS, Redford JB (eds). Control of posture and locomotion. Plenum Press, New York, pp 515–536
Grillner S, Dubuc R (1988) Control of locomotion in vertebrates: spinal and supraspinal mechanisms. In: Waxman SG (eds) Functional recovery in neurological disease. Raven Press New York, pp 425–453
Hoffer JA, Caputi AA, Pose IE, Griffiths RI (1989) Roles of muscle activity and load on the relationship between muscle spindle length and whole muscle length in the freely walking cat. Prog Brain Res 80: 75–85
Hoffer JA, Loeb GE, Sugano N, Marks WB, O'Donovan MJ, Pratt CA. (1987) Cat hindlimb motoneurons during locomotion. III. Functional segregation in sartorius. J Neurophysiol 57: 554–562
Lundberg A, Malmgren K, Schomburg ED (1987) Reflex pathways from group II muscle afferents. 1. Distribution and linkage of reflex action to alpha motoneurones. Exp Brain Res 65: 271–281
Matthews PBC (1972) Muscle receptors. Edward Arnold, London
Mense S, Meyer H (1985) Different types of slowly conducting afferent units in cat skeletal muscle and tendon. J Physiol 363: 403–417
Pearson KG, Jiang W, Ramirez JM (1992) The use of naloxone to facilitate the generation of the locomotor rhythm in spinal cats. J Neurosci Methods In Press
Prochazka A, Trend P, Hulliger M, Vincent S (1989) Ensemble proprioceptive activity in the cat step cycle: towards a representative look-up chart. Prog Brain Res 80: 61–74
Rossignol S, Lund JP, Drew T (1988) The role of sensory inputs in regulating patterns of rhythmical movements in higher vertebrates. In: Cohen A, Rossignol S, Grillner S (eds) Neural control of rhythmic movements in vertebrates. John Wiley and Sons, New York, pp 201–283
Sherrington CS (1910) Flexor-reflex of the limb, crossed extension reflex, and reflex stepping and standing (cat and dog). J Physiol 40: 28–121
Shimamura M, Kogure I, Fuwa T (1984) Role of joint afferents in relation to the initiation of forelimb stepping in thalamic cats. Brain Res 297: 225–234
Smith JL (1986) Hindlimb locomotion of the spinal cat synergistic patterns, limb dynamics and novel blends. In: Grillner S, Stein PSG, Stuart DG, Forssberg H, Herman RM (eds) Neurobiology of vertebrate locomotion. Macmillan Press, Hong Kong. pp 185–200
Author information
Authors and Affiliations
Additional information
CRSN, Physiologie, Faculte de Medecine, C.P. 6128, succursal A, Montréal, Quebec, Canada, H3C 3J7
Rights and permissions
About this article
Cite this article
Pearson, K.G., Ramirez, J.M. & Jiang, W. Entrainment of the locomotor rhythm by group Ib afferents from ankle extensor muscles in spinal cats. Exp Brain Res 90, 557–566 (1992). https://doi.org/10.1007/BF00230939
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230939