Abstract
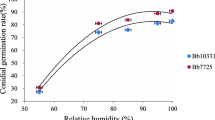
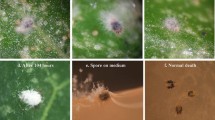
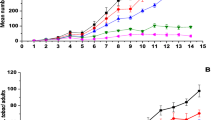
The insecticidal activity of Beauveria bassiana GHA derived from a commercial mycoinsecticide BotaniGard ES against Frankliniella occidentalis was determined in a bioassay by dipping the female adults into a conidial suspension. The 90% lethal concentration of B. bassiana GHA was estimated to be 9.7 × 106 conidia/ml. The lethal times for achieving 90% mortality of thrips inoculated with a 1/500-diluted solution of BotaniGard ES and a 107.5 (3.16 × 107) conidia/ml suspension of B. bassiana GHA were estimated to be five and six days, respectively. When the treated thrips were exposed to a high relative humidity (RH) of over 99% for various periods and then transferred to 60% RH, the requisite lengths of the high-humidity period to achieve 90% mortality of the thrips at six days after inoculation were estimated to be 46 and 47 h in BotaniGard ES and B. bassiana GHA, respectively. Fungal multiplication in the thrips was detected between 48 to 60 h after inoculation by measuring Beauveria-specific DNA in the host following inoculation with a B. bassiana GHA suspension of 107.5 conidia/ml using a real-time quantitative PCR. The mycelial growth in the host hemocoel was not influenced by the low-humidity condition.




Similar content being viewed by others
References
Abe M, Ikegami T (2005) Susceptibility of five species of thrips to different strains of the entomopathogenic fungus, Beauveria bassiana. Appl Entomol Zool 40:667–674. doi:10.1303/aez.2005.667
Bateman R, Carey M, Moore D, Prior C (1993) The enhanced infectivity of Metarhizium flavoviride in oil formulations to desert locusts at low humidities. Ann Appl Biol 122:145–152. doi:10.1111/j.1744-7348.1993.tb04022.x
Butt TM, Brownbridge M (1997) Fungal pathogens of thrips. In: Lewis T (ed) Thrips as crop pests. CAB International, Wallingford, pp 399–433
Castrillo LA, Griggs MH, Vandenberg JD (2008) Quantitative detection of Beauveria bassiana GHA (Ascomycota: Hypocreales), a potential microbial control agent of the emerald ash borer, by use of real-time PCR. Biol Control 45:163–169. doi:10.1016/j.biocontrol.2008.01.005
Cory JS, Ericsson JD (2010) Fungal entomopathogens in a tritrophic context. Biocontrol 55:75–88. doi:10.1007/s10526-009-9247-4
Enkerli J, Widmer F (2010) Molecular ecology of fungal entomopathogens: molecular genetic tools and their applications in population and fate studies. Biocontrol 55:17–37. doi:10.1007/s10526-009-9251-8
Fargues J, Luz C (2000) Effects of fluctuating moisture and temperature regimes on the infection potential of Beauveria bassiana for Rhodnius prolixus. J Invertebr Pathol 75:202–211. doi:10.1006/jipa.1999.4923
Fargues J, Ouedraogo A, Goettel MS, Lomer CJ (1997) Effects of temperature, humidity and inoculation method on susceptibility of Schistocerca gregaria to Metarhizium flavoviride. Biocontrol Sci Technol 7:345–356. doi:10.1080/09583159730758
De Faria MR, Wraight SP (2007) Mycoinsecticides and mycoacaricides: a comprehensive list with worldwide coverage and international classification of formulation types. Biol Control 43:237–256. doi:10.1016/j.biocontrol.2007.08.001
Finney DJ (1978) Statistical methods in biological assay. Charles Griffin and Co., London
Hajek AE, St. Leger RJ (1994) Interactions between fungal pathogens and insect hosts. Annu Rev Entomol 39:293–322
Ibrahim L, Butt TM, Beckett A, Clark SJ (1999) The germination of oil-formulated conidia of the insect pathogen, Metarhizium anisopliae. Mycol Res 103:901–907. doi:10.1017/S0953756298007849
Inglis GD, Goettel MS, Butt TM, Strasser H (2001) Use of hyphomycetous fungi for managing insect pests. In: Butt TM, Jackson CW, Magan N (eds) Fungi as biological agents: progress, problems and potential. CAB International, Wallingford, pp 23–69
Iwase R, Shimizu S (2004) Adhesion and virulence of conidia of Metarhizium anisopliae to Thrips palmi (Thysanoptera: Thripidae). Jpn J Appl Entomol Zool 48:275–280. doi:101303/jjaez2004.275
Jackson MK, Dunlap CA, Jaronski ST (2010) Ecological considerations in producing and formulating fungal entomopathogens for use in insect biocontrol. Biocontrol 55:129–145. doi:10.1007/s10526-009-9240-y
James RR, Croft BA, Shaffer BT, Lighthart B (1998) Impact of temperature and humidity on host–pathogen interactions between Beauveria bassiana and a coccinellid. Environ Entomol 27:1506–1513
Jaronski ST (2010) Ecological factors in the inundative use of fungal entomopathogens. Biocontrol 55:159–185. doi:10.1007/s10526-009-9248-3
Jensen SE (2000) Insecticide resistance in the western flower thrips, Frankliniella occidentalis. Integr Pest Manag Rev 5:131–146. doi:10.1023/A:1009600426262
Luz C, Fargues J (1999) Dependence of the entomopathogenic fungus, Beauveria bassiana, on high humidity for infection of Rhodnius prolixus. Mycopathologia 146:33–41. doi:10.1023/A:1007019402490
Masuda T (2000) Microbial control of the diamondback moth, Plutella xylostella, by an entomopathogenic fungus, Beauveria bassiana. II. Effects of temperature on mycoses and conidial invasion time. Jpn J Appl Entomol Zool 44:177–182. doi:101303/jjaez2000.177 (In Japanese with English summary)
Moino A Jr, Alves SB, Lopes RB, Oliveira PM, Neves J, Pereira RM, Vieira SA (2002) External development of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae in the subterranean termite Heterotermes tenuis. Sci Agric 59:267–273. doi:10.1590/S0103-90162002000200010
Mukawa S, Goto C (2006) In vivo characterization of a group II nucleopolyhedrovirus isolated from Mamestra brassicae (Lepidoptera: Noctuidae) in Japan. J Gen Virol 87:1491–1500. doi:10.1099/vir.0.81853-0
Murai T, Ishii T (1982) Simple rearing method for flower thrips (Thysanoptera: Thripidae) on pollen. Jpn J Appl Entomol Zool 26:149–154 (In Japanese with English summary)
Murai T, Loomans AJM (2001) Evaluation of an improved method for mass-rearing of thrips and a thrips parasitoid. Entomol Exp Appl 101:281–289. doi:10.1023/A:1019252708938
Osborne LS, Landa Z (1992) Biological control of whiteflies with entomopathogenic fungi. Florida Entomol 75:456–471
Sakakibara M, Kawakami K (1990) The effect of DDVP against Cordyceps brongniartii, natural enemy of yellow-spotted longicorn beetle (Psacothea hilaris). J Seric Sci Jpn 59:210–217 (In Japanese with English summary)
Sakuma M (1998) Probit analysis of preference data. Appl Entomol Zool 33:339–347
SAS Institute (2002) JMP statistics and graphics guide, version 5. SAS Institute, Cary
Shipp JL, Zhang Y, Hunt DWA, Ferguson G (2003) Influence of humidity and greenhouse microclimate on the efficacy of Beauveria bassiana (Balsamo) for control of greenhouse arthropod pests. Environ Entomol 32:1154–1163. doi:10.1603/0046-225X-32.5.1154
Sokal RR, Rohlf FJ (1995) Biometry, 3rd ed. WH Freeman and Co., New York
Ugine TA, Wraight SP, Sanderson JP (2005) Acquisition of lethal doses of Beauveria bassiana conidia by western flower thrips, Frankliniella occidentalis, exposed to foliar spray residues of formulated and unformulated conidia. J Invertebr Pathol 90:10–23. doi:101016/jjip2005.07.003
Ugine TA, Wraight SP, Sanderson JP (2007) A tritrophic effect of host plant on susceptibility of western flower thrips to the entomopathogenic fungus Beauveria bassiana. J Invertebr Pathol 96:162–172. doi:101016/jjip2007.05.004
Ullman DE, Sherwood JL, German TL (1997) Thrips as vectors of plant pathogens. In: Lewis T (ed) Thrips and crop pests. CAB International, Wallingford, pp 539–565
Vestergaard S, Butt TM, Bresciani J, Gillespie AT, Eilenberg J (1999) Light and electron microscopy studies of the infection of the western flower thrips Frankliniella occidentalis (Thysanoptera: Thripidae) by the entomopathogenic fungus Metarhizium anisopliae. J Invertebr Pathol 73:25–33. doi:10.1006/jipa.1998.4802
Wraight SP, Jackson MA, de Kock SL (2001) Production, stabilization and formulation of fungal biocontrol agents. In: Butt TM, Jackson CW, Magan N (eds) Fungi as biological agents: progress, problems and potential. CAB International, Wallingford, pp 253–287
Acknowledgments
We are grateful to Hitoshi Kawamata and Shinichi Numata for valuable suggestions about this study. We thank Yoshito Suzuki, Masanori Koike, Satoshi Yamanaka, and Shohei Okuno for critically reading the manuscript. This work was supported by a research fellowship to S. Mukawa from the Ibaraki Agricultural Center.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mukawa, S., Tooyama, H. & Ikegami, T. Influence of humidity on the infection of western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae), by Beauveria bassiana . Appl Entomol Zool 46, 255–264 (2011). https://doi.org/10.1007/s13355-011-0033-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-011-0033-2