Abstract
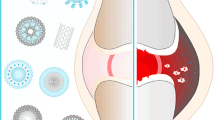
Rheumatoid arthritis (RA) is a chronic, destructive autoimmune disease that can cause disability and have a negative socioeconomic impact. Despite significant advances in therapeutic options, limitations on the routes of administration and the requirement for frequent and long-term dosing often lead to systemic adverse effects and patient non-compliance. Micro and nanoparticle systems that selectively deliver drugs to inflamed synovium have the potential to improve drug efficacy while leaving extrasynovial tissues unaffected. This review summarizes key design parameters of RA-targeted drug carriers and discusses design considerations for improving RA therapies.
Similar content being viewed by others
References
Avnir, Y., Ulmansky, R., Wasserman, V., Even Chen, S., Broyer, M., Barenholz, Y., and Naparstek, Y., Amphipathic weak acid glucocorticoid prodrugs remote loaded into sterically stabilized nanoliposomes evaluated in arthritic rats and in a Beagle dog: A novel approach to treating autoimmune arthritis. Arthritis Rheum., 58, 119–129 (2008).
Butoescu, N., Seemayer, C. A., Foti, M., Jordan, O., and Doelker, E., Dexamethasone-containing PLGA superparamagnetic microparticles as carriers for the local treatment of arthritis. Biomaterials, 30, 1772–1780 (2009).
Champion, J. A. and Mitragotri, S., Role of target geometry in phagocytosis. Proc. Natl. Acad. Sci. U. S. A., 103, 4930–4934 (2006).
Champion, J. A., Katare, Y. K., and Mitragotri, S., Particle shape: a new design parameter for micro- and nanoscale drug delivery carriers. J. Control. Release, 121, 3–9 (2007).
Chandrasekar, D., Sistla, R., Ahmad, F. J., Khar, R. K., and Diwan, P. V., Folate coupled poly(ethyleneglycol) conjugates of anionic poly(amidoamine) dendrimer for inflammatory tissue specific drug delivery. J. Biomed. Mater. Res. A, 82, 92–103 (2007).
Chithrani, B. D., Ghazani, A. A., and Chan, W. C. W., Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano lett., 6, 662–668 (2006).
Council, E. R., Multi-centre controlled trial comparing cortisone acetate and acetyl salicylic acid in the long-term treatment of rheumatoid arthritis. Ann. Rheum. Dis., 16, 277–289 (1957).
Decuzzi, P., Pasqualini, R., Arap, W., and Ferrari, M., Intravascular delivery of particulate systems: does geometry really matter? Pharm. Res., 26, 235–243 (2009).
Doshi, N., Prabhakarpandian, B., Rea-Ramsey, A., Pant, K., Sundaram, S., and Mitragotri, S., Flow and adhesion of drug carriers in blood vessels depend on their shape: a study using model synthetic microvascular networks. J. Control. Release, 146, 196–200 (2010).
Emery, P., Breedveld, F. C., Hall, S., Durez, P., Chang, D. J., Robertson, D., Singh, A., Pedersen, R. D., Koenig, A. S., and Freundlich, B., Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): a randomised, double-blind, parallel treatment trial. Lancet, 372, 375–382 (2008).
Everts, M., Koning, G. A., Kok, R. J., Asgeirsdottir, S. A., Vestweber, D., Meijer, D. K., Storm, G., and Molema, G., In vitro cellular handling and in vivo targeting of E-selectin-directed immunoconjugates and immunoliposomes used for drug delivery to inflamed endothelium. Pharm. Res., 20, 64–72 (2003).
Fang, C., Shi, B., Pei, Y. Y., Hong, M. H., Wu, J., and Chen, H. Z., In vivo tumor targeting of tumor necrosis factoralpha-loaded stealth nanoparticles: effect of MePEG molecular weight and particle size. Eur. J. Pharm. Sci., 27, 27–36 (2006).
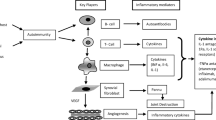
Feldmann, M. and Maini, S. R. N., Role of cytokines in rheumatoid arthritis: an education in pathophysiology and therapeutics. Immunol. Rev., 223, 7–19 (2008).
Firestein, G. S., Evolving concepts of rheumatoid arthritis. Nature, 423, 356–361 (2003).
Furst, D. E., The risk of infections with biologic therapies for rheumatoid arthritis. Semin. Arthritis Rheum., 39, 327–346 (2010).
Garrood, T. and Pitzalis, C., Targeting the inflamed synovium: the quest for specificity. Arthritis Rheum., 54, 1055–1060 (2006).
Gerlag, D. M., Borges, E., Tak, P. P., Ellerby, H. M., Bredesen, D. E., Pasqualini, R., Ruoslahti, E., and Firestein, G. S., Suppression of murine collagen-induced arthritis by targeted apoptosis of synovial neovasculature. Arthritis Res., 3, 357–361 (2001).
Gref, R., Minamitake, Y., Peracchia, M. T., Trubetskoy, V., Torchilin, V., and Langer, R., Biodegradable long-circulating polymeric nanospheres. Science, 263, 1600–1603 (1994).
Gregoriadis, G., Engineering liposomes for drug delivery: progress and problems. Trends Biotechnol., 13, 527–537 (1995).
Hafstrom, I., Albertsson, K., Boonen, A., Van Der Heijde, D., Landewe, R., and Svensson, B., Remission achieved after 2 years treatment with low-dose prednisolone in addition to disease-modifying anti-rheumatic drugs in early rheumatoid arthritis is associated with reduced joint destruction still present after 4 years: an open 2-year continuation study. Ann. Rheum. Dis., 68, 508–513 (2009).
Hattori, Y., Sakaguchi, M., and Maitani, Y., Folate-linked lipid-based nanoparticles deliver a NFkappaB decoy into activated murine macrophage-like RAW264.7 cells. Biol. Pharm. Bull., 29, 1516–1520 (2006).
Hollander, J., Brown, E., Jessar, R., and Brown, C., Comparative effects of use of hydrocortisone as a local antiarthritic agent. JAMA, 147, 1629–1635 (1951).
Hood, J. D., Bednarski, M., Frausto, R., Guccione, S., Reisfeld, R. A., Xiang, R., and Cheresh, D. A., Tumor regression by targeted gene delivery to the neovasculature. Science, 296, 2404–2407 (2002).
Horisawa, E., Hirota, T., Kawazoe, S., Yamada, J., Yamamoto, H., Takeuchi, H., and Kawashima, Y., Prolonged antiinflammatory action of DL-lactide/glycolide copolymer nanospheres containing betamethasone sodium phosphate for an intra-articular delivery system in antigen-induced arthritic rabbit. Pharm. Res., 19, 403–410 (2002a).
Horisawa, E., Kubota, K., Tuboi, I., Sato, K., Yamamoto, H., Takeuchi, H., and Kawashima, Y., Size-dependency of DLlactide/glycolide copolymer particulates for intra-articular delivery system on phagocytosis in rat synovium. Pharm. Res., 19, 132–139 (2002b).
Ishihara, T., Kubota, T., Choi, T., and Higaki, M., Treatment of experimental arthritis with stealth-type polymeric nanoparticles encapsulating betamethasone phosphate. J. Pharmacol. Exp. Ther., 329, 412–417 (2009).
Jamar, F., Chapman, P., Manicourt, D., Glass, D., Haskard, D., and Peters, A., A comparison between 111In-anti-Eselectin mAb and 99Tcm-labelled human non-specific immunoglobulin in radionuclide imaging of rheumatoid arthritis. Br. J. Radiol., 70, 473 (1997).
Khoury, M., Louis-Plence, P., Escriou, V., Noel, D., Largeau, C., Cantos, C., Scherman, D., Jorgensen, C., and Apparailly, F., Efficient new cationic liposome formulation for systemic delivery of small interfering RNA silencing tumor necrosis factor alpha in experimental arthritis. Arthritis Rheum., 54, 1867–1877 (2006).
Koch, A., Angiogenesis as a target in rheumatoid arthritis. Ann. Rheum. Dis., 62, ii60-ii67 (2003).
Koning, G. A., Schiffelers, R. M., Wauben, M. H., Kok, R. J., Mastrobattista, E., Molema, G., Ten Hagen, T. L., and Storm, G., Targeting of angiogenic endothelial cells at sites of inflammation by dexamethasone phosphate-containing RGD peptide liposomes inhibits experimental arthritis. Arthritis Rheum., 54, 1198–1208 (2006).
Lawrence, R. C., Helmick, C. G., Arnett, F. C., Deyo, R. A., Felson, D. T., Giannini, E. H., Heyse, S. P., Hirsch, R., Hochberg, M. C., Hunder, G. G., Liang, M. H., Pillemer, S. R., Steen, V. D., and Wolfe, F., Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis Rheum., 41, 778–799 (1998).
Levick, J. R., Hypoxia and acidosis in chronic inflammatory arthritis; relation to vascular supply and dynamic effusion pressure. J. Rheumatol., 17, 579–582 (1990).
Liang, L. S., Jackson, J., Min, W., Risovic, V., Wasan, K. M., and Burt, H. M., Methotrexate loaded poly(L-lactic acid) microspheres for intra-articular delivery of methotrexate to the joint. J. Pharm. Sci., 93, 943–956 (2004).
Liang, L. S., Wong, W., and Burt, H. M., Pharmacokinetic study of methotrexate following intra-articular injection of methotrexate loaded poly(L-lactic acid) microspheres in rabbits. J. Pharm. Sci., 94, 1204–1215 (2005).
Liggins, R. T., Cruz, T., Min, W., Liang, L., Hunter, W. L., and Burt, H. M., Intra-articular treatment of arthritis with microsphere formulations of paclitaxel: biocompatibility and efficacy determinations in rabbits. Inflamm. Res., 53, 363–372 (2004).
Listing, J., Strangfeld, A., Kary, S., Rau, R., Von Hinueber, U., Stoyanova-Scholz, M., Gromnica-Ihle, E., Antoni, C., Herzer, P., Kekow, J., Schneider, M., and Zink, A., Infections in patients with rheumatoid arthritis treated with biologic agents. Arthritis Rheum., 52, 3403–3412 (2005).
Mansouri, S., Cuie, Y., Winnik, F., Shi, Q., Lavigne, P., Benderdour, M., Beaumont, E., and Fernandes, J. C., Characterization of folate-chitosan-DNA nanoparticles for gene therapy. Biomaterials, 27, 2060–2065 (2006).
Mcneil, J. and Binette, J., Prevalence of disabilities and associated health conditions among adults—United States, 1999. MMWR Morb. Mortal. Wkly. Rep., 50, 120–125 (2001).
Metselaar, J. M., Wauben, M. H., Wagenaar-Hilbers, J. P., Boerman, O. C., and Storm, G., Complete remission of experimental arthritis by joint targeting of glucocorticoids with long-circulating liposomes. Arthritis Rheum., 48, 2059–2066 (2003).
Metselaar, J. M., Van Den Berg, W. B., Holthuysen, A. E., Wauben, M. H., Storm, G., and Van Lent, P. L., Liposomal targeting of glucocorticoids to synovial lining cells strongly increases therapeutic benefit in collagen type II arthritis. Ann. Rheum. Dis., 63, 348–353 (2004).
Montesinos, M. C., Takedachi, M., Thompson, L. F., Wilder, T. F., Fernandez, P., and Cronstein, B. N., The antiinflammatory mechanism of methotrexate depends on extracellular conversion of adenine nucleotides to adenosine by ecto-5’-nucleotidase: findings in a study of ecto-5’-nucleotidase gene-deficient mice. Arthritis Rheum., 56, 1440–1445 (2007).
Muller, R. H., Mader, K., and Gohla, S., Solid lipid nanoparticles (SLN) for controlled drug delivery — a review of the state of the art. Eur. J. Pharm. Biopharm., 50, 161–177 (2000).
Naor, D. and Nedvetzki, S., CD44 in rheumatoid arthritis. Arthritis Res. Ther., 5, 105–115 (2003).
Natarajan, V., Krithica, N., Madhan, B., and Sehgal, P. K., Formulation and evaluation of quercetin polycaprolactone microspheres for the treatment of rheumatoid arthritis. J. Pharm. Sci., 100, 195–205 (2011).
O’dell, J. R., Haire, C. E., Erikson, N., Drymalski, W., Palmer, W., Eckhoff, P. J., Garwood, V., Maloley, P., Klassen, L. W., Wees, S., Klein, H., and Moore, G. F., Treatment of rheumatoid arthritis with methotrexate alone, sulfasalazine and hydroxychloroquine, or a combination of all three medications. N. Engl. J. Med., 334, 1287–1291 (1996).
Owens, D. E. 3rd and Peppas, N. A., Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm., 307, 93–102 (2006).
Paulos, C. M., Turk, M. J., Breur, G. J., and Low, P. S., Folate receptor-mediated targeting of therapeutic and imaging agents to activated macrophages in rheumatoid arthritis. Adv. Drug Deliv. Rev., 56, 1205–1217 (2004).
Paulos, C. M., Varghese, B., Widmer, W. R., Breur, G. J., Vlashi, E., and Low, P. S., Folate-targeted immunotherapy effectively treats established adjuvant and collagen-induced arthritis. Arthritis Res. Ther., 8, R77 (2006).
Pincus, T., Marcum, S. B., and Callahan, L. F., Longterm drug therapy for rheumatoid arthritis in seven rheumatology private practices: II. Second line drugs and prednisone. J. Rheumatol., 19, 1885–1894 (1992).
Piscaer, T. M., Müller, C., Mindt, T. L., Lubberts, E., Verhaar, J. A., Krenning, E. P., Schibli, R., De Jong, M., and Weinans, H., Imaging of activated macrophages in experimental osteoarthritis using folate targeted animal singlephoton-emission computed tomography/computed tomography. Arthritis Rheum., 63, 1898–1907 (2011).
Ratcliffe, J. H., Hunneyball, I. M., Wilson, C. G., Smith, A., and Davis, S. S., Albumin microspheres for intra-articular drug delivery: investigation of their retention in normal and arthritic knee joints of rabbits. J. Pharm. Pharmacol., 39, 290–295 (1987).
Roh, K. H., Martin, D. C., and Lahann, J., Biphasic Janus particles with nanoscale anisotropy. Nat. Mater., 4, 759–763 (2005).
Romberg, B., Hennink, W. E., and Storm, G., Sheddable coatings for long-circulating nanoparticles. Pharm. Res., 25, 55–71 (2008).
Saag, K. G., Glucocorticoid use in rheumatoid arthritis. Curr. Rheumatol. Rep., 4, 218–225 (2002).
Schiffelers, R. M., Koning, G. A., Ten Hagen, T. L., Fens, M. H., Schraa, A. J., Janssen, A. P., Kok, R. J., Molema, G., and Storm, G., Anti-tumor efficacy of tumor vasculaturetargeted liposomal doxorubicin. J. Control. Release, 91, 115–122 (2003).
Schmitt, F., Lagopoulos, L., Kauper, P., Rossi, N., Busso, N., Barge, J., Wagnieres, G., Laue, C., Wandrey, C., and Juillerat-Jeanneret, L., Chitosan-based nanogels for selective delivery of photosensitizers to macrophages and improved retention in and therapy of articular joints. J. Control. Release, 144, 242–250 (2010).
Senolt, L., Vencovsky, J., Pavelka, K., Ospelt, C., and Gay, S., Prospective new biological therapies for rheumatoid arthritis. Autoimmun. Rev., 9, 102–107 (2009).
Sharma, G., Valenta, D. T., Altman, Y., Harvey, S., Xie, H., Mitragotri, S., and Smith, J. W., Polymer particle shape independently influences binding and internalization by macrophages. J. Control. Release, 147, 408–412 (2010).
Shehata, T., Ogawara, K., Higaki, K., and Kimura, T., Prolongation of residence time of liposome by surface-modification with mixture of hydrophilic polymers. Int. J. Pharm., 359, 272–279 (2008).
Shi, Q., Wang, H., Tran, C., Qiu, X., Winnik, F. M., Zhang, X., Dai, K., Benderdour, M., and Fernandes, J. C., Hydrodynamic delivery of chitosan-folate-DNA nanoparticles in rats with adjuvant-induced arthritis. J. Biomed. Biotechnol., 2011, 148763 (2011).
Shive, M. S. and Anderson, J. M., Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev., 28, 5–24 (1997).
Simon, L. S., DMARDs in the treatment of rheumatoid arthritis: current agents and future developments. Int. J. Clin. Pract., 54, 243–249 (2000).
Smolen, J. S. and Steiner, G., Therapeutic strategies for rheumatoid arthritis. Nat. Rev. Drug Discov., 2, 473–488 (2003).
Solomon, D. H., Katz, J. N., Jacobs, J. P., La Tourette, A. M., and Coblyn, J., Management of glucocorticoid induced osteoporosis in patients with rheumatoid arthritis: Rates and predictors of care in an academic rheumatology practice. Arthritis Rheum., 46, 3136–3142 (2002).
Strand, V., Kimberly, R., and Isaacs, J. D., Biologic therapies in rheumatology: lessons learned, future directions. Nat. Rev. Drug Discov., 6, 75–92 (2007).
Svensson, B., Boonen, A., Albertsson, K., Van Der Heijde, D., Keller, C., and Hafstrom, I., Low-dose prednisolone in addition to the initial disease-modifying antirheumatic drug in patients with early active rheumatoid arthritis reduces joint destruction and increases the remission rate: a twoyear randomized trial. Arthritis Rheum., 52, 3360–3370 (2005).
Tarner, I. H., Harle, P., Muller-Ladner, U., Gay, R. E., and Gay, S., The different stages of synovitis: acute vs chronic, early vs late and non-erosive vs erosive. Best Pract. Res. Clin. Rheumatol., 19, 19–35 (2005).
Tarner, I. H. and Muller-Ladner, U., Drug delivery systems for the treatment of rheumatoid arthritis. Expert Opin. Drug Deliv., 5, 1027–1037 (2008).
Tomita, T., Takeuchi, E., Tomita, N., Morishita, R., Kaneko, M., Yamamoto, K., Nakase, T., Seki, H., Kato, K., Kaneda, Y., and Ochi, T., Suppressed severity of collagen-induced arthritis by in vivo transfection of nuclear factor kappaB decoy oligodeoxynucleotides as a gene therapy. Arthritis Rheum., 42, 2532–2542 (1999).
Torchilin, V., Tumor delivery of macromolecular drugs based on the EPR effect. Adv. Drug Deliv. Rev., 63, 131–135 (2011).
Turk, M. J., Breur, G. J., Widmer, W. R., Paulos, C. M., Xu, L. C., Grote, L. A., and Low, P. S., Folate-targeted imaging of activated macrophages in rats with adjuvant-induced arthritis. Arthritis Rheum., 46, 1947–1955 (2002).
Utada, A. S., Lorenceau, E., Link, D. R., Kaplan, P. D., Stone, H. A., and Weitz, D. A., Monodisperse double emulsions generated from a microcapillary device. Science, 308, 537–541 (2005).
Van Der Heijden, J. W., Oerlemans, R., Dijkmans, B. A., Qi, H., Van Der Laken, C. J., Lems, W. F., Jackman, A. L., Kraan, M. C., Tak, P. P., Ratnam, M., and Jansen, G., Folate receptor beta as a potential delivery route for novel folate antagonists to macrophages in the synovial tissue of rheumatoid arthritis patients. Arthritis Rheum., 60, 12–21 (2009).
Van Vollenhoven, R. F., Treatment of rheumatoid arthritis: state of the art 2009. Nat. Rev. Rheumatol., 5, 531–541 (2009).
Vanniasinghe, A. S., Bender, V., and Manolios, N., The potential of liposomal drug delivery for the treatment of inflammatory arthritis. Semin. Arthritis Rheum., 39, 182–196 (2009).
Wallis, W. J., Simkin, P. A., and Nelp, W. B., Protein traffic in human synovial effusions. Arthritis Rheum., 30, 57–63 (1987).
Wang, D., Miller, S. C., Liu, X. M., Anderson, B., Wang, X. S., and Goldring, S. R., Novel dexamethasone-HPMA copolymer conjugate and its potential application in treatment of rheumatoid arthritis. Arthritis Res. Ther., 9, R2 (2007).
Wolfe, F., Mitchell, D. M., Sibley, J. T., Fries, J. F., Bloch, D. A., Williams, C. A., Spitz, P. W., Haga, M., Kleinheksel, S. M., and Cathey, M. A., The mortality of rheumatoid arthritis. Arthritis Rheum., 37, 481–494 (1994).
Ye, J., Wang, Q., Zhou, X., and Zhang, N., Injectable actaritloaded solid lipid nanoparticles as passive targeting therapeutic agents for rheumatoid arthritis. Int. J. Pharm., 352, 273–279 (2008).
Yoo, J. W., Chambers, E., and Mitragotri, S., Factors that control the circulation time of nanoparticles in blood: challenges, solutions and future prospects. Curr. Pharm. Des., 16, 2298–2307 (2010a).
Yoo, J. W., Doshi, N., and Mitragotri, S., Endocytosis and Intracellular Distribution of PLGA Particles in Endothelial Cells: Effect of Particle Geometry. Macromol. Rapid Commun., 31, 142–148 (2010b).
Yoo, J. W. and Mitragotri, S., Polymer particles that switch shape in response to a stimulus. Proc. Natl. Acad. Sci. U. S. A., 107, 11205–11210 (2010).
Yoo, J. W., Doshi, N., and Mitragotri, S., Adaptive micro and nanoparticles: Temporal control over carrier properties to facilitate drug delivery. Adv. Drug Deliv. Rev., 63, 1247–1256 (2011a).
Yoo, J. W., Irvine, D. J., Discher, D. E., and Mitragotri, S., Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov., 10, 521–535 (2011b).
Zhigaltsev, I. V., Maurer, N., Edwards, K., Karlsson, G., and Cullis, P. R., Formation of drug-arylsulfonate complexes inside liposomes: a novel approach to improve drug retention. J. Control. Release, 110, 378–386 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitragotri, S., Yoo, JW. Designing micro- and nano-particles for treating rheumatoid arthritis. Arch. Pharm. Res. 34, 1887–1897 (2011). https://doi.org/10.1007/s12272-011-1109-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-1109-9