Abstract
Background
Recent evidence suggests that the peptidyl-prolyl isomerase Pin1 is an oncoprotein that acts as a novel therapeutic target in a variety of tumors. In this study, we investigated the clinical significance of Pin1 and its function in prostate cancer (PCa) tumor progression.
Methods
Immunohistochemical and quantitative RT-PCR analyses were performed to detect the expression of Pin1 in 86 PCa tissue samples. The functional role of Pin1 was evaluated by small interfering RNA-mediated depletion in PCa cells followed by analyses of cell proliferation and migration. Furthermore, the association between expression of Pin1 and levels of β-catenin and cyclin D1 was also evaluated.
Results
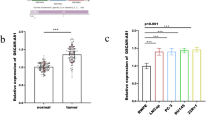
Our results showed that the high expression of Pin1 staining was 66 of 86 (76.74 %) PCa samples, and in 25 of 86 (29.07 %) BPH tissues, the difference was statistically significant (P < 0.001). Pin1 was significantly elevated in all PCa cell lines when compared to the normal RWPE-1 cells. We observed that proliferation and migration of LNCaP cells were inhibited by Pin1 knockdown. The levels of β-catenin and cyclin D1 in clinical PCa specimens were positively associated with Pin1 expression.
Conclusions
Our results suggest that Pin1 plays an important role in tumorigenesis of PCa, suggesting that targeting Pin1 pathway could represent a potential modality for treating PCa.





Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29.
Zhang L, Yang BX, Zhang HT, Wang JG, Wang HL. Zhao Xj. Prostate cancer: an emerging threat to the health of aging men in Asia. Asian J Androl. 2011;13:574–8.
Sommariva S, Tarricone R, Lazzeri M, Ricciardi W, Montorsi F. Prognostic value of the cell cycle progression score in patients with prostate cancer: a systematic review and meta-analysis. Eur Urol. 2014;. doi:10.1016/j.eururo.2014.11.038.
Nakamura K, Greenwood A, Binder L, Bigio EH, Denial S, Nicholson L, et al. Proline isomer-specific antibodies reveal the early pathogenic tau conformation in Alzheimer’s disease. Cell. 2012;149(1):232–44.
Tun-Kyi A, Finn G, Greenwood A, Nowak M, Lee TH, Asara JM, et al. Essential role for the prolyl isomerase Pin1 in Toll-like receptor signaling and type I interferon-mediated immunity. Nat Immunol. 2011;12(8):733–41.
Winkler KE, Swenson KI, Kornbluth S, Means AR. Requirement of the prolyl isomerase Pin1 for the replication checkpoint. Science (Wash. DC). 2000;287:1644–7.
Ayala G, Wang D, Wulf G, Frolov A, Li R, Sowadski J, et al. The prolyl isomerase Pin1 is a novel prognostic marker in human prostate cancer. Cancer Res. 2003;63:6244–51.
Lee TH, Chen CH, Suizu F, Huang P, Schiene-Fischer C, Daum S, et al. Death-associated protein kinase 1 phosphorylates Pin1 and inhibits its prolyl isomerase activity and cellular function. Mol Cell. 2011;42(2):147–59.
Bao L, Kimzey A, Sauter G, Sowadski JM, Lu KP, Wang DG. Prevalent overexpression of prolyl isomerase Pin1 in human cancers. Am J Pathol. 2004;164(5):1727–37.
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, et al. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol. 2014;65(1):124–37.
Saad F, Pantel K. The current role of circulating tumor cells in the diagnosis and management of bone metastases in advanced prostate cancer. Future Oncol. 2012;8(3):321–31.
Lu KP, Hanes SD, Hunter T. A human peptidyl-prolyl isomerase essential for regulation of mitosis. Nature. 1996;380:544–7.
Takahashi K, Akiyama H, Shimazaki K, Uchida C, Akiyama-Okunuki H, Tomita M, et al. Ablation of a peptidyl prolyl isomerase Pin1 from p53-null mice accelerated thymic hyperplasia by increasing the level of the intracellular form of Notch1. Oncogene. 2007;26(26):3835–45.
Liou YC, Zhou XZ, Lu KP. Prolyl isomerase Pin1 as a molecular switch to determine the fate of phosphoproteins. Trends Biochem Sci. 2011;36:501–14.
Fukuchi M, Fukai Y, Kimura H, Sohda M, Miyazaki T, Nakajima M, et al. Prolyl isomerase Pin1 expression predicts prognosis in patients with esophageal squamous cell carcinoma and correlates with cyclin D1 expression. Int J Oncol. 2006;29:329–34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
All specimens were collected in accordance with informed consents of patients, and all procedures complied with the protocol approved by the Ethical Committee of Affiliated Hospital of Weifang Medical University.
Rights and permissions
About this article
Cite this article
Zhu, Z., Zhang, H., Lang, F. et al. Pin1 promotes prostate cancer cell proliferation and migration through activation of Wnt/β-catenin signaling. Clin Transl Oncol 18, 792–797 (2016). https://doi.org/10.1007/s12094-015-1431-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1431-7