Opinion statement
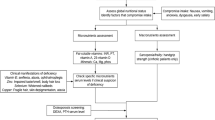
The treatment of children with end-stage liver disease involves the coordinated management of nutritional deficiencies, ascites, pruritus, encephalopathy, and portal hypertension. The implementation of management strategies depends upon a parent or guardian to administer the plan in the context of a child at different stages of developmental, physiologic, emotional, and physical maturity. Fat-soluble vitamins (A, D, E, and K) and micronutrient levels should be monitored routinely and supplemented if deficient. In some patients, supplemental nutrition to provide additional energy and protein is needed to ensure optimal growth and development. Ascites often respond to spironolactone and sodium restriction, but may require the addition of a loop diuretic or even abdominal paracentesis. Pruritus significantly impairs the quality of life of patients and is typically treated with ursodeoxycholic acid, rifampin, or an antihistamine. Partial biliary diversion, or liver transplant in some instances, is necessary for patients with self-mutilating pruritus that results from intrahepatic cholestasis. Hepatic encephalopathy is poorly defined in infants and small children. Elevated serum ammonia serves as a surrogate marker for encephalopathy, which is treated with dietary protein restriction and lactulose. The usefulness of medical prophylaxis for esophageal varices has been noted in adults, though such studies have not been performed in children. If variceal bleeding becomes problematic, treatment with endoscopic variceal banding or sclerotherapy is indicated. A surgical shunt to reduce portal pressure is needed in some cases. Orthotopic liver transplant ultimately may be necessary to overcome the unrelenting consequences of end-stage liver disease.
Similar content being viewed by others
References and Recommended Reading
Holt RI, Baker AJ, Miell JP: The pathogenesis of growth failure in pediatric liver disease. J Hepatol 1997, 27:413–423.
Moreno LA, Gottraud F, Holden S, et al.: Improvement of nutritional status in cholestatic children with supplemental nocturnal enteral nutrition. J Pediatr Gastroenterol Nutr 1991, 12:213–216.
Sokol RJ, Butler-Simon N, Conner C, et al.: Multicenter trial of d-alpha-tocopheryl polyethylene glycol 1000 succinate for treatment of vitamin E deficiency in children with chronic cholestasis. Gastroenterology 1993, 104:1727–1735.
Socha P, Koletzko B, Pawlowska J, et al.: Treatment of cholestatic children with water-soluble vitamin E (a-tocopheryl polyethylene glycol succinate): effects on serum vitamin E, lipid peroxides, and polyunsaturated fatty acids. J Pediatr Gastroenterol Nutr 1997, 24:189–193.
Argao EA, Heubi JE, Hollis W, Tsang RC: d-a-tocopheryl polyethylene glycol-1000 succinate enhances the absorption of vitamin D in chronic cholestatic liver disease of infancy and childhood. Pediatr Res 1992, 31:146–150.
Heubi JE, Hollis BW, Specker B, Tsang RC: Bone disease in chronic childhood cholestasis. I. Vitamin D absorption and metabolism. Hepatology 1989, 9:258–264.
Heubi JE, Hollis BW, Tsang RC: Bone disease in chronic childhood cholestasis II. Better absorption of 25-OH vitamin D than vitamin D in extrahepatic biliary atresia. Pediatr Res 1990, 27:26–31.
Bucuvala JC, Heubi JE, Specker BL, et al.: Calcium absorption in bone disease associated with liver cholestasis during childhood. Hepatology 1990, 12:1200–1205.
Bachrach S, Fisher J, Parks JS: An outbreak of vitamin D deficiency rickets in a susceptible population. Pediatrics 1979, 64:871–877.
Eugster EA, Sane KS, Brown DM: Minnesota rickets. Need for a policy change to support vitamin D supplementation. Minn Med 1996, 79:29–32.
Amedee-Manesme O, Furr HC, Alvarez F, et al.: Biochemical indicators of vitamin A depletion in children with cholestasis. Hepatology 1985, 6:1143–1148.
Smith FR, Goodman DS: Vitamin A transport in human vitamin A toxicity. N Engl J Med 1976, 294:805–808.
Houwen RHJ, Bouquet J, Bijleveld CMA: Bleeding as the first symptom of EHBA. Eur J Pediatr 1987, 146:426–426.
Aiza I, Perez GO, Schiff ER: Management of ascites in patients with chronic liver disease. Amer J Gastroenterol 1994, 89:1949–1956.
Runyon BA: Treatment of patients with cirrhosis and ascites. Semin Liver Dis 1997, 249–260.
Garcia-Tsao G: Current management of the complications of cirrhosis and portal hypertension: variceal hemorrhage, ascites, and spontaneous bacterial peritonitis. Gastroenterology 2001, 120:726–748.
Rossle M, Ochs A, Gulberg V, et al.: A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites. N Engl J Med 2000, 342:1701–1707.
Sort P, Navasa M, Arroyo V, et al.: Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med 1999, 341:403–309.
Jones EA, Bergasa NV: The pruritus of cholestasis: from bile acids to opiate agonists. Hepatology 1990, 11:884–887.
Cynamon HA, Andres JM, Iafrate RP: Rifampin relieves pruritus in children with cholestatic liver disease. Gastroenterology 1990, 98:1013–1016.
Carson KL, Tran TT, Cotton P, et al.: Pilot study of the use of naltrexone to treat the severe pruritus of cholestatic liver disease. Am J Gastroenterol 1996, 91:1022–1023.
Bergasa NV, Jones EA: The pruritus of cholestasis: potential pathogenic and therapeutic implications of opioids. Gastroenterology 1995, 108:1582–1588.
Wolfhagen FHJ, Sternieri E, Hop WCJ, et al.: Oral naltrexone treatment for cholestatic pruritus: a double blind, placebo-controlled study. Gastroenterology 1997, 113:1264–1269.
Jones EA, Bergasa NV: The pruritus of cholestasis and the opioid system. JAMA 1992, 268:3359–3362.
Whitington PF, Whitington GL: Partial external diversion of bile for the treatment of intractable pruritus associated with intrahepatic cholestasis. Gastroenterology 1988, 95:130–136.
Record CO: Neurochemistry of hepatic encephalopathy. Gut 1991, 32:1261–1263.
Jalan R, Hayes PC: Hepatic encephalopathy and ascites. Lancet 1997, 350:1309–1315.
Riordan SM, Williams R: Treatment of hepatic encephalopathy. N Engl J Med 1997, 337:473–479.
Ullrich D, Birchner J: Lactulose in pediatrics. In Hepatic Encephalopathy: Management with Lactulose and Related Carbohydrates. Edited by Conn HO, Bircher J. East Lansing, MI: Medi Ed Press; 1988:315–331.
Siafakas C, Fox VL, Nurko S: Use of octreotide for the treatment of severe gastrointestinal bleeding in children. J Pediatr Gastroenterol Nutr 1998, 26:356–359.
Laine L, Cook D: Endoscopic ligation compared with sclerotherapy for treatment of esophageal variceal bleeding. Ann Intern Med 1995, 123:280–287.
Fox VL, Carr-Locke DL, Connors PJ, Leichtner AM: Endoscopic ligation of esophageal varices in children. J Pediatr Gastroenterol Nutr 1995: 20:202–208.
Proujansky R, Orenstein SR, Kocoshis S: Patient and procedure variables associated with complications following variceal sclerotherapy in children. J Pediatr Gastroenterol Nutr 1991, 12:33–38.
Hill ID, Bowie MD: Endoscopic sclerotherapy for control of bleeding varices in children. Am J Gastroenterol 1991, 86:472–476.
Tam F, Chow H, Prindiville T, et al.: Bacterial peritonitis following esophageal injection sclerotherapy for variceal hemorrhage. Gastrointest Endosc 1990, 36:131–133.
Baydur A, Korula J: Cardiorespiratory effects of endoscopic esophageal variceal sclerotherapy. Am J Med 1990, 89:477–482.
Stanley AJ, Redhead DN, Hayes PC: Review article: update on the role of transjugular intrahepatic portosystemic stent-shunt (TIPSS) in the management of complications of portal hypertension. Aliment Pharmacol Ther 1997, 11:261–272.
Shun A, Delaney DP, Martin HCO, et al.: Portosystemic shunting for paediatric portal hypertension. J Pediatr Surg 1997, 32:489–493.
Reyes J, Vazariegos GV, Bueno J, et al.: The role of portosystemic shunting in children in the transplant era. J Pediatr Surg 1999, 34:117–123.
De Ville de Goyet J, Alberti D, Calpuyt P, et al.: Direct bypassing of extrahepatic portal venous obstruction in children: a new technique for combined hepatic portal revascularization and treatment of extrahepatic portal hypertension. J Pediatr Surg 1998, 33:597–601.
2000 Drug Topics Red Book. Montvale, NJ: Medical Economics Co; 2000.
Micromedex Healthcare Series, vol 108 [online database]. Greenwood Village, CO: Micromedex, Inc. URL: www.micromedex.com. Accessed June 2001.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Squires, R.H. End-stage liver disease in children. Curr Treat Options Gastro 4, 409–421 (2001). https://doi.org/10.1007/s11938-001-0006-6
Issue Date:
DOI: https://doi.org/10.1007/s11938-001-0006-6