Abstract
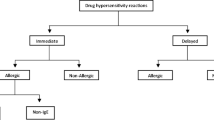
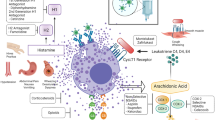
Although the reported incidence of hypersensitivity reactions (HSR) to antineoplastic agents is considered to be uncommon, it is difficult to evaluate their exact prevalence, mainly because their definition is vast and pathogenic mechanisms are vague. HSR include facial flushing, erythema, pruritus, fever, tachycardia, dyspnea, tongue swelling, rash/hives, headache, chills, weakness, vomiting, burning sensations, dizziness, and edema. Treatment and prevention consists of slowing the infusion rate, steroids, and type 1 and 2 histamine receptor antagonists. Desensitization could allow the small number of patients who experience severe HSR to receive effective therapy for their cancer. Reintroductions have only been reported as single case studies or small cohorts. Large-scale validation on desensitization strategies is still missing. With regard to oxaliplatin, knowledge of its rare but eminent toxicity is paramount, because this drug is widely used in treating colorectal cancer, the second-highest cause of cancer mortality in the United States.
Similar content being viewed by others
References and Recommended Reading
Shepherd GM: Hypersensitivity reactions to chemotherapeutic drugs. Clin Rev Allergy Immunol 2003, 24:253–262.
Robinson JB, Singh D, Bodurka-Bevers DC, et al.: Hypersensitivity reactions and the utility of oral and intravenous desensitization in patients with gynecologic malignancies. Gynecol Oncol 2001, 82:550–558.
Gruchalla RS: Drug allergy. J Allergy Clin Immunol 2003, 111(2 Suppl):S548–S559.
Rothenberg ML, Oza AM, Bigelow RH, et al.: Superiority of oxaliplatin and fluorouracil-leucovorin compared with either therapy alone in patients with progressive colorectal cancer after irinotecan and fluorouracil-leucovorin: interim results of a phase III trial. J Clin Oncol 2003, 21:2059–2069.
De Gramont A, Banzi M, Navarro M, et al.: Oxaliplatin/5-FU/LV in adjuvant colon cancer: results of the international randomized mosaic trial. Proc Am Soc Clin Oncol 2003, 22:253.
Maindrault-Goebel F, Andre T, Tournigand C, et al.: Allergic-type reactions to oxaliplatin: retrospective analysis of 42 patients. Eur J Cancer 2005, 41:2262–2267.
Thomas RR, Quinn MG, Schuler B, et al.: Hypersensitivity and idiosyncratic reactions to oxaliplatin. Cancer 2003, 97:2301–2307.
De Vries RS, Mattijssen EJ, van Sorge AA: Serious delayed hypersensitivity reaction to oxaliplatin. Ann Oncol 2006, 17:1723–1724.
Stahl M, Koster W, Wilke H: Reaction after oxaliplatin-prevention with corticosteroids. Ann Oncol 2005, 12:874.
Santini D, Tonini G, Salerno A, et al.: Idiosyncratic reactions after oxaliplatin infusion. Ann Oncol 2005, 12:132–133.
Newman Taylor AJ, Cullinan P, Lympany PA, et al.: Interaction of HLA phenotype and exposure intensity in sensitization to complex platinum salts. Am J Resp Crit Care Med 1991, 160:435–438.
Hetherington S, Hughes AR, Mosteller M, et al.: Genetic variations in HLA-B region and hypersensitivity reactions to abacavir. Lancet 2002, 359:1121–1122.
Bhargava P, Gammon D, McCormick MJ: Hypersensitivity and idiosyncratic reactions to oxaliplatin. Cancer 2004, 100:211–212.
Brandi G, Pantaleo MA, Galli C, et al.: Hypersensitivity reactions related to oxaliplatin (OHP). Br J Cancer 2003, 89:477–481.
Meyer L, Zuberbier T, Worm M, et al.: Hypersensitivity reactions to oxaliplatin: cross-reactivity to carboplatin and the introduction of a desensitization schedule. J Clin Oncol 2002, 20:1146–1147.
Qureshi KM: Incidence of hypersensitivity reactions to oxaliplatin at Roswell Park Cancer Institute (RPCI) and protocol for desensitization. Proc Am Soc Clin Oncol 2003, 22:789.
Siu SW, Chan RT, Au GK: Hypersensitivity reactions to oxaliplatin: experience in a single institute. Ann Oncol 2005, 17:259–261.
Lee MY, Yang MH, Liu JH, et al.: Severe anaphylactic reactions in patients receiving oxaliplatin therapy: a rare but potentially fatal complication. Support Care Cancer 2007, 15:89–93.
Eloxatin (oxaliplatin injection) [package insert]. Bridgewater, NJ: Sanofi Aventis US; 2007. Available at: www.eloxatin.com.
Herrero T, Tornero P, Infante S, et al.: Diagnosis and management of hypersensitivity reactions caused by oxaliplatin. J Investig Allergol Clin Immunol 2006, 16:327–330.
Ulrich-Pur H, Penz M, Fiebiger WC, et al.: Oxaliplatin-induced fever and release of IL-6. Oncology 2000, 59:187–189.
Sampson HA, Muñoz-Furlong A, Campbell RL, et al.: Second symposium on the definition and management of anaphylaxis: summary report—Second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunol 2006, 117:391–397.
Lenz HJ: Management and preparedness for infusion and hypersensitivity reactions. Oncologist 2007, 12:601–609.
Christina VT: Hypersensitivity reactions to oxaliplatin in two Asian patients. Ann Pharmacother 2005, 39:1114–1118.
Zanotti KM, Markman M: Prevention and management of antineoplasticinduced hypersensitivity reactions. Drug Saf 2001, 24:767–779.
Gonzales ID, Saez RS, Rodilla EM, et al.: Hypersensitivity reactions to chemotherapy drugs. Alergol Immunol Clin 2000, 15:161–181.
Leguy-Seguin V, Jolimoy G, Coudert B, et al.: Diagnostic and predictive value of skin testing in platinum salt hypersensitivity. J Allergy Clin Immunol 2007, 119:726–730.
Zanotti K, Rybicki L, Kennedy A, et al.: Carboplatin skin testing: a skin-testing protocol for predicting hypersensitivity to carboplatin chemotherapy. J Clin Oncol 2001, 19:3126–3129.
Markman M, Zanotti K, Peterson G, et al.: Expanded experience with an intradermal skin test to predict for the presence or absence of carboplatin hypersensitivity. J Clin Oncol 2003, 21:4611–4614.
Garufi C, Cristaudo A, Vanni B, et al.: Skin testing and hypersensitivity reactions to oxaliplatin. Ann Oncol 2003, 14:497–498.
Mis L, Fernando NH, Hurwitz HI, Morse MA: Successful desensitization to oxaliplatin. Ann Pharmacother 2005, 39:966–968.
Edmondson DA, Gruling BJ, Urmanski AM, et al.: Oxaliplatin hypersensitivity: case report and successful repeat desensitization. Am J Ther 2007, 14:116–118.
Saif MW, Reardon J: Management of oxaliplatin-induced peripheral neuropathy. Ther Clin Risk Manage 2005, 1:249–258.
Saif MW: Oral calcium ameliorating oxaliplatin-induced peripheral neuropathy. J Applied Res 2004, 4:576–582.
Saif MW: Hypersensitivity reactions associated with oxaliplatin. Expert Opin Drug Saf 2006, 5:687–694.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Syrigou, E., Syrigos, K. & Saif, M.W. Hypersensitivity reactions to oxaliplatin and other antineoplastic agents. Curr Allergy Asthma Rep 8, 56–62 (2008). https://doi.org/10.1007/s11882-008-0011-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11882-008-0011-0