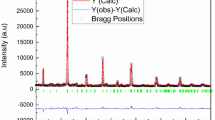
Abstract
Results from theoretical analysis of the crystal structure, electronic structure, and bonding properties of C46 and B6C40 carbon clathrates doped with selected alkali and alkaline earth metals cations (Li, Na, Mg, Ca) are presented. The ab initio calculations were performed by means of the WIEN2k package (full potential linearized augmented plane wave method (FP-LAPW) within density functional theory (DFT)) with PBESol and modified Becke–Johnson exchange–correlation potentials used in geometry optimization and electronic structure calculations, respectively. The bonding properties were analyzed by applying Bader’s quantum theory of atoms in molecules formalism to the topological properties of total electron density obtained from ab initio calculations. Analysis of the results obtained (i.a. equilibrium geometry, equation of state, cohesive energy, band structure, density of states—both total and projected on to particular atoms, and topological properties of bond critical points and net charges of topological atoms) is presented in detail.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G.S. Nolas, J.L. Cohn, G.A. Slack, and S.B. Schujman, Appl. Phys. Lett. 73, 178 (1998)
J.-T. Wang, C. Chen, D.-S. Wang, H. Mizuseki, and Y. Kawazoe, J. Appl. Phys. 107, 063507 (2010)
X. Blase, P. Gillet, A. San Miguel, and P. Mélinon, Phys. Rev. Lett. 92(21), 215505 (2004)
X. Blasé, Phys. Rev. B 67, 035211 (2003)
M. Bernasconi, S. Gaito, and G. Benedek, Phys. Rev. B 61, 12689 (2000)
F. Zipoli, M. Bernasconi, and G. Benedek, Phys. Rev. B 74, 205408 (2006)
N. Rey, A. Muñoz, P. Rodríguez-Hernández, and A. San Miguel J. Phys. Condens. Matter. 20, 215218 (2008) http://iopscience.iop.org/article/10.1088/0953-8984/20/21/21 5218/meta
D. Connétable, Phys. Rev. B 82, 075209 (2010)
R.F.W. Bader, Atoms in Molecules: A Quantum Theory (Clarendon Press, Oxford, 1990)
P. Blaha, K. Schwarz, G.K.H. Madsen, D. Kvasnicka, and J. Luitz, WIEN2K: An Augmented Plane Wave Plus Local Orbitals Program for Calculating Crystal Properties (Vienna Technological University, Vienna, 2001)
J.P. Perdew, A. Ruzsinszky, G.I. Csonka, O.A. Vydrov, G.E. Scuseria, L.A. Constantin, X. Zhou, and K. Burke, Phys. Rev. Lett. 100, 136406 (2008)
F. Tran and P. Blaha, Phys. Rev. Lett. 102, 226401 (2009)
R. Dovesi, R. Orlando, A. Erba, C.M. Zicovich-Wilson, B. Civalleri, S. Casassa, L. Maschio, M. Ferrabone, M.D.L. Pierre, P. D’Arco, Y. Noël, M. Causá, M. Rérat, and B. Kirtman, Int. J. Quantum Chem. 114, 1287 (2014)
R. Dovesi, V.R. Saunders, C. Roetti, R. Orlando, C.M. Zicovich-Wilson, F. Pascale, B. Civalleri, K. Doll, N.M. Harrison, I.J. Bush, P. D’Arco, M. Llunell, M. Causà, and Y. Noël, CRYSTAL14 User’s Manual (University of Torino, Torino, 2014)
M.F. Peintinger, D. Vilela, Oliveira, and T. Bredow, J. Comput. Chem. 34(6), 451 (2013)
A. Otero-de-la-Roza, M. Blanco, A.M. Pendás, and V. Luaña, Comput. Phys. Commun. 160(1), 157 (2009)
D.G. Cahill, S.K. Watson, and R.O. Pohl, Phys. Rev. B 46, 6131 (1992)
Acknowledgements
This research was supported by Polish National Science Center, Grant No. UMO-2013-09/B/ST8/02043 and in part by PL-Grid Infrastructure.
Open Access
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below are links to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
KoleŻyński, A., Szczypka, W. First-Principles Study of the Electronic Structure and Bonding Properties of X8C46 and X8B6C40 (X: Li, Na, Mg, Ca) Carbon Clathrates. J. Electron. Mater. 45, 1336–1345 (2016). https://doi.org/10.1007/s11664-015-4028-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-4028-6