Abstract
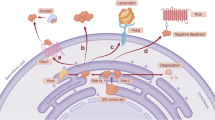
Signaling pathways transduce extracellular stimuli into cells through molecular cascades to regulate cellular functions. In stem cells, a small number of pathways, notably those of TGF-β/BMP, Hedgehog, Notch, and Wnt, are responsible for the regulation of pluripotency and differentiation. During embryonic development, these pathways govern cell fate specifications as well as the formation of tissues and organs. In adulthood, their normal functions are important for tissue homeostasis and regeneration, whereas aberrations result in diseases, such as cancer and degenerative disorders. In complex biological systems, stem cell signaling pathways work in concert as a network and exhibit crosstalk, such as the negative crosstalk between Wnt and Notch. Over the past decade, genetic and genomic studies have identified a number of potential drug targets that are involved in stem cell signaling pathways. Indeed, discovery of new targets and drugs for these pathways has become one of the most active areas in both the research community and pharmaceutical industry. Remarkable progress has been made and several promising drug candidates have entered into clinical trials. This review focuses on recent advances in the discovery of novel drugs which target the Notch and Wnt pathways.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andersson ER, Sandberg R, Lendahl U. Notch signaling: simplicity in design, versatility in function. Development, 2011, 138: 3593–3612
Koch U, Lehal R, Radtke F. Stem cells living with a notch. Development, 2013, 140: 689–704
Nusslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature, 1980, 287: 795–801
Rijsewijk F, Schuermann M, Wagenaar E, Parren P, Weigel D, Nusse R. The Drosophila homolog of the mouse mammary oncogene int-1 is identical to the segment polarity gene wingless. Cell, 1987, 50: 649–657
Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. Cell, 2012, 149: 1192–1205
Axelrod JD, Matsuno K, ArtavanisTsakonas S, Perrimon N. Interaction between wingless and Notch signaling pathways mediated by dishevelled. Science, 1996, 271: 1826–1832
Estrach S, Ambler CA, Lo Celso C, Hozumi K, Watt FM. Jagged 1 is a beta-catenin target gene required for ectopic hair follicle formation in adult epidermis. Development, 2006, 133: 4427–4438
Kwon C, Cheng P, King IN, et al. Notch post-translationally regulates beta-catenin protein in stem and progenitor cells. Nat Cell Biol, 2011, 13: 1244–1251
Proweller A, Tu LL, Lepore JJ, Cheng L, Lu MM, Seykora J, Millar SE, Pear WS, Parmacek MS. Impaired notch signaling promotes de novo squamous cell carcinoma formation. Cancer Res, 2006, 66: 7438–7444
Nicolas M, Wolfer A, Raj K, Kummer JA, Mill P, van Noort M, Hui CC, Clevers H, Dotto GP, Radtke F. Notch1 functions as a tumor suppressor in mouse skin. Nat Genet, 2003, 33: 416–421
Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD, Sklar J. TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell, 1991, 66: 649–661
Weng AP, Ferrando AA, Lee W, Morris JP 4th, Silverman LB, Sanchez-Irizarry C, Blacklow SC, Look AT, Aster JC. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science, 2004, 306: 269–271
Klinakis A, Lobry C, Abdel-Wahab O, Oh P, Haeno H, Buonamici S, van De Walle I, Cathelin S, Trimarchi T, Araldi E, Liu C, Ibrahim S, Beran M, Zavadil J, Efstratiadis A, Taghon T, Michor F, Levine RL, Aifantis I. A novel tumour-suppressor function for the Notch pathway in myeloid leukaemia. Nature, 2011, 473: 230–233
Ranganathan P, Weaver KL, Capobianco AJ. Notch signalling in solid tumours: a little bit of everything but not all the time. Nat Rev Cancer, 2011, 11: 338–351
Hao L, Rizzo P, Osipo C, Pannuti A, Wyatt D, Cheung LW, Sonenshein G, Osborne BA, Miele L. Notch-1 activates estrogen receptor-alpha-dependent transcription via IKKalpha in breast cancer cells. Oncogene, 2010, 29: 201–213
Wang NJ, Sanborn Z, Arnett KL, Bayston LJ, Liao W, Proby CM, Leigh IM, Collisson EA, Gordon PB, Jakkula L, Pennypacker S, Zou Y, Sharma M, North JP, Vemula SS, Mauro TM, Neuhaus IM, Leboit PE, Hur JS, Park K, Huh N, Kwok PY, Arron ST, Massion PP, Bale AE, Haussler D, Cleaver JE, Gray JW, Spellman PT, South AP, Aster JC, Blacklow SC, Cho RJ. Loss-of-function mutations in Notch receptors in cutaneous and lung squamous cell carcinoma. Proc Natl Acad Sci USA, 2011, 108: 17761–17766
Nusse R, Varmus HE. Many tumors induced by the mouse mammary-tumor virus contain a provirus integrated in the same region of the host genome. Cell, 1982, 31: 99–109
Papkoff J, Rubinfeld B, Schryver B, Polakis P. Wnt-1 regulates free pools of catenins and stabilizes APC-catenin complexes. Mol Cell Biol, 1996, 16: 2128–2134
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B, Kinzler KW. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science, 1997, 275: 1787–1790
Korinek V, Barker N, Morin PJ, van Wichen D, de Weger R, Kinzler KW, Vogelstein B, Clevers H. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science, 1997, 275: 1784–1787
Seshagiri S, Stawiski EW, Durinck S, Modrusan Z, Storm EE, Conboy CB, Chaudhuri S, Guan Y, Janakiraman V, Jaiswal BS, Guillory J, Ha C, Dijkgraaf GJ, Stinson J, Gnad F, Huntley MA, Degenhardt JD, Haverty PM, Bourgon R, Wang W, Koeppen H, Gentleman R, Starr TK, Zhang Z, Largaespada DA, Wu TD, de Sauvage FJ. Recurrent R-spondin fusions in colon cancer. Nature, 2012, 488: 660–664
Gurney A, Axelrod F, Bond CJ, Cain J, Chartier C, Donigan L, Fischer M, Chaudhari A, Ji M, Kapoun AM, Lam A, Lazetic S, Ma S, Mitra S, Park IK, Pickell K, Sato A, Satyal S, Stroud M, Tran H, Yen WC, Lewicki J, Hoey T. Wnt pathway inhibition via the targeting of Frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc Natl Acad Sci USA, 2012, 109: 11717–11722
Gupta PB, Onder TT, Jiang GZ, Tao K, Kuperwasser C, Weinberg RA, Lander ES. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell, 2009, 138: 645–659
Lu DS, Choi MY, Yu J, Castro JE, Kipps TJ, Carson DA. Salinomycin inhibits Wnt signaling and selectively induces apoptosis in chronic lymphocytic leukemia cells. Proc Natl Acad Sci USA, 2011, 108: 13253–13257
Liu J, Pan SF, Hsieh MH, et al. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc Natl Acad Sci USA, 2013, 110: 20224–20229
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
An, S.M., Ding, Q., Zhang, J. et al. Targeting stem cell signaling pathways for drug discovery: advances in the Notch and Wnt pathways. Sci. China Life Sci. 57, 575–580 (2014). https://doi.org/10.1007/s11427-014-4665-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4665-7