Abstract
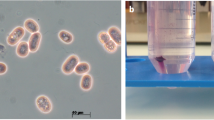
Using a pyrosequencing-based custom-made sequencer BIGIS-4, we sequenced a Gram-negative bacterium Glaciecola mesophila sp. nov. (Gmn) isolated from marine invertebrate specimens. We generated 152043 sequencing reads with a mean high-quality length of 406 bp, and assembled them using the BIGIS-4 post-processing module. No systematic low-quality data was detected beyond expected homopolymer-derived errors. The assembled Gmn genome is 5144318 bp in length and harbors 4303 annotated genes. A large number of metabolic genes correspond to various nutrients from surface marine invertebrates. Its abundant cold-tolerant and cellular signaling and related genes reveal a fundamental adaptation to low-temperature marine environment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Romanenko L A, Zhukova N V, Rohde M, et al. Glaciecola mesophila sp. nov., a novel marine agar-digesting bacterium. Int J Syst Evol Microbiol, 2003, 53: 647–651 12807181, 10.1099/ijs.0.02469-0, 1:CAS:528:DC%2BD3sXkvFOjur8%3D
Zhang D C, Yu Y, Chen B, et al. Glaciecola psychrophila sp. nov., a novel psychrophilic bacterium isolated from the Arctic. Int J Syst Evol Microbiol, 2006, 56: 2867–2869 17158989, 10.1099/ijs.0.64575-0, 1:CAS:528:DC%2BD2sXhtVWmtrg%3D
Van Trappen S, Tan T L, Yang J, et al. Glaciecola polaris sp. nov., a novel budding and prosthecate bacterium from the Arctic Ocean, and emended description of the genus Glaciecola. Int J Syst Evol Microbiol, 2004, 54: 1765–1771 15388742, 10.1099/ijs.0.63123-0
Baik K S, Park Y D, Seong C N, et al. Glaciecola nitratireducens sp. nov., isolated from seawater. Int J Syst Evol Microbiol, 2006, 56: 2185–2188 16957118, 10.1099/ijs.0.64330-0, 1:CAS:528:DC%2BD28XhtFektL%2FI
Chen L P, Xu H Y, Fu S Z, et al. Glaciecola lipolytica sp. nov., isolated from seawater near Tianjin city, China. Int J Syst Evol Microbiol, 2009, 59: 73–76 19126727, 10.1099/ijs.0.000489-0, 1:CAS:528:DC%2BD1MXisVCmtLk%3D
Matsuyama H, Hirabayashi T, Kasahara H, et al. Glaciecola chathamensis sp. nov., a novel marine polysaccharide-producing bacterium. Int J Syst Evol Microbiol, 2006, 56: 2883–2886 17158992, 10.1099/ijs.0.64413-0, 1:CAS:528:DC%2BD2sXhtVWmtrc%3D
Zhang Y J, Zhang X Y, Mi Z H, et al. Glaciecola arctica sp. nov., isolated from Arctic marine sediment. Int J Syst Evol Microbiol, 2010
Yong J J, Park S J, Kim H J, et al. Glaciecola agarilytica sp. nov., an agar-digesting marine bacterium from the East Sea, Korea. Int J Syst Evol Microbiol, 2007, 57: 951–953 17473239, 10.1099/ijs.0.64723-0, 1:CAS:528:DC%2BD2sXntVCgs74%3D
Prabagaran S R, Manorama R, Delille D, et al. Predominance of Roseobacter, Sulfitobacter, Glaciecola and Psychrobacter in seawater collected off Ushuaia, Argentina, Sub-Antarctica. FEMS Microbiol Ecol, 2007, 59: 342–355 17026513, 10.1111/j.1574-6941.2006.00213.x, 1:CAS:528:DC%2BD2sXhslGrs78%3D
Guo B, Chen X L, Sun C Y, et al. Gene cloning, expression and characterization of a new cold-active and salt-tolerant endo-beta-1,4-xylanase from marine Glaciecola mesophila KMM 241. Appl Microbiol Biotechnol, 2009, 84: 1107–1115 19506861, 10.1007/s00253-009-2056-y, 1:CAS:528:DC%2BD1MXht1Sqs7jM
Ronaghi M, Uhlen M, Nyren P. A sequencing method based on real-time pyrophosphate. Science, 1998, 281: 363–365 9705713, 10.1126/science.281.5375.363, 1:CAS:528:DyaK1cXkslagtL4%3D
Gordon D. Viewing and editing assembled sequences using Consed. Curr Protoc Bioinformatics, 2003, Chapter 11: Unit11.2
Delcher A L, Bratke K A, Powers E C, et al. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics, 2007, 23: 673–679 17237039, 10.1093/bioinformatics/btm009, 1:CAS:528:DC%2BD2sXkt1GhtL8%3D
Zdobnov E M, Apweiler R. InterProScan—an integration platform for the signature-recognition methods in InterPro. Bioinformatics (Oxford, England), 2001, 17: 847–848 10.1093/bioinformatics/17.9.847, 1:CAS:528:DC%2BD3MXotFehsro%3D
Schattner P, Brooks A N, Lowe T M. The tRNAscan-SE, snoscan and snoGPS web servers for the detection of tRNAs and snoRNAs. Nucleic Acids Res, 2005, 33: W686–W689 15980563, 10.1093/nar/gki366, 1:CAS:528:DC%2BD2MXlslyrt7c%3D
Siguier P, Perochon J, Lestrade L, et al. ISfinder: the reference centre for bacterial insertion sequences. Nucleic Acids Res, 2006, 34: D32–D36 16381877, 10.1093/nar/gkj014, 1:CAS:528:DC%2BD28XisFOhug%3D%3D
Kurtz S, Phillippy A, Delcher A L, et al. Versatile and open software for comparing large genomes. Genome Biol, 2004, 5: R12 14759262, 10.1186/gb-2004-5-2-r12
Bott O J, Bergmann J, Hoffmann I, et al. Analysis and specification of telemedical systems using modelling and simulation: the MOSAIK-M approach. Stud Health Technol Inform, 2005, 116: 503–508 16160307, 1:STN:280:DC%2BD2MvptVSlsg%3D%3D
Ivars-Martinez E, Martin-Cuadrado A B, D’Auria G, et al. Comparative genomics of two ecotypes of the marine planktonic copiotroph Alteromonas macleodii suggests alternative lifestyles associated with different kinds of particulate organic matter. ISME J, 2008, 2: 1194–1212 18670397, 10.1038/ismej.2008.74, 1:CAS:528:DC%2BD1MXhtVKitb0%3D
Collins T, Gerday C, Feller G. Xylanases, xylanase families and extremophilic xylanases. FEMS Microbiol Rev, 2005, 29: 3–23 15652973, 10.1016/j.femsre.2004.06.005, 1:CAS:528:DC%2BD2MXkvFeiug%3D%3D
Tosco A, Birolo L, Madonna S, et al. GroEL from the psychrophilic bacterium Pseudoalteromonas haloplanktis TAC 125: molecular characterization and gene cloning. Extremophiles, 2003, 7: 17–28 12579376, 1:CAS:528:DC%2BD3sXhtVKktbw%3D
Yamauchi S, Okuyama H, Morita E H, et al. Gene structure and transcriptional regulation specific to the groESL operon from the psychrophilic bacterium Colwellia maris. Arch Microbiol, 2003, 180: 272–278 12898133, 10.1007/s00203-003-0587-7, 1:CAS:528:DC%2BD3sXnslSntbY%3D
Xu K, Ma B G. Comparative analysis of predicted gene expression among deep-sea genomes. Gene, 2007, 397: 136–142 17544603, 10.1016/j.gene.2007.04.023, 1:CAS:528:DC%2BD2sXntFaqt7o%3D
Lopez-Lopez A, Bartual S G, Stal L, et al. Genetic analysis of house-keeping genes reveals a deep-sea ecotype of Alteromonas macleodii in the Mediterranean Sea. Environ Microbiol, 2005, 7: 649–659 15819847, 10.1111/j.1462-2920.2005.00733.x, 1:CAS:528:DC%2BD2MXksFCgsLg%3D
Gauthier G, Gauthier M, Christen R. Phylogenetic analysis of the genera Alteromonas, Shewanella, and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (emended) and Pseudoalteromonas gen. nov., and proposal of twelve new species combinations. Int J Syst Bacteriol, 1995, 45: 755–761 7547295, 10.1099/00207713-45-4-755, 1:CAS:528:DyaK28XivFGl
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
An erratum to this article can be found at http://dx.doi.org/10.1007/s11427-011-4228-0
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Yuan, L., Ren, L., Li, Y. et al. A complete genome assembly of Glaciecola mesophila sp. nov. sequenced by using BIGIS-4 sequencer system. Sci. China Life Sci. 54, 835–840 (2011). https://doi.org/10.1007/s11427-011-4211-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4211-9