Abstract
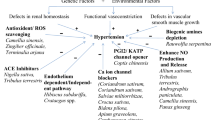
The effect of a herbomineral formulation (HMF) on early diabetic nephropathy was investigated. Diabetes was induced in Wistar rats by administering streptozotocin (55 mg/kg, intraperitoneally). The occurrence of early diabetic nephropathy in rats was revealed by high plasma glucose and depleted liver glycogen, decreased glucose uptake by peripheral tissue, impaired renal function, increased antioxidants and lipid peroxidation in kidney. These changes were accompanied by elevated malondialdehyde, glutathione and superoxide dismutase activity in kidney. Furthermore, increased total urine volume, urinary albumin excretion rate, urinary albumin to creatinine ratio, increased relative kidney weight, decreased glomerular filtration rate (GFR) and urinary creatinine were also observed in diabetic nephropathy rats. HMF treatment significantly lowered blood glucose, glycosylated hemoglobin, creatinine, blood urea nitrogen, triglycerides, total cholesterol, serum albumin level, total urine volume, urinary albumin excretion rate, urinary albumin to creatinine ratio and relative kidney weight, and increased urinary creatinine and GFR. Altered levels of antioxidants, viz. lipid peroxidation, glutathione and superoxide dismutase (SOD), in kidney of diabetic nephropathy rats were restored. Histopathological findings indicated dense mesangial matrix in the glomeruli of diabetic nephropathy rats, which may be due to over-activation of matrix metalloproteinases and was reduced following HMF treatment. Our experimental findings clearly demonstrate that HMF has an ability to prevent the progression of early diabetic nephropathy. Such protective effect of HMF might be due to the presence of flavonoids (catechin, quercetin, rutin) and triterpene saponins (oleanolic acid and gymnemic acid) which are known to possess potent antioxidant properties.




Similar content being viewed by others
References
Takako Y, Noriko Y, Eun JC, Tokako N, Shigeru O (2004) A study on the effect to diabetic nephropathy of Hachimi-jio-gan in rats. Nephron Exp Nephrol 97:e38–e48
Dronavalli S, Duka I, Bakris GL (2008) The pathogenesis of diabetic nephropathy. Nat Clin Pract Endocrinol Metab 4(8):444–452
Kikkawa R, Koya D, Haneda M (2003) Progression of diabetic nephropathy. Am J Kidney Dis 41(3–2):S19–S21
Wadood A, Wadood N, Shah SA (1989) Effects of Acacia arabica and Caralluma edulis on blood glucose levels of normal and alloxan diabetic rabbits. J Pak Med Assoc 39(8):208–212
Sagrawat H, Mann AS, Kharya MD (2006) Pharmacological potential of Eugenia jambolana: a review. Pharmacogn Mag 2(6):96–105
Gholap S, Kar A (2003) Effect of Inula racemosa root and Gymnema sylvestre leaf extract in the regulation of corticosteroid induced diabetes mellitus: involvement of thyroid hormones. Pharmazie 58:413–415
Robert A (2000) Zinc in relation to diabetes and oxidative disease. J Nutr 130(5):1509S–1511S
Sajeeth CI, Manna PK, Manavalan R, Jolly CI (2010) Quantitative estimation of gallic acid, rutin and quercetin in certain herbal plants by HPTLC method. Der Chemica Sinica 1(2):80–85
Nehete JY, Deshmukh VN, Shewale VV, Narkhede MR, Aurangabadkar VM (2009) Quantitation of oleanolic acid in Achyranthes aspera L. roots and leaves extracts by high-performance thin-layer chromatography. Int J Pharm Res Dev 2(7). ISSN 0974-9446
Trinder P (1969) Determination of blood glucose using an oxidase peroxidase system with a non-carcinogenic chromogen. Ann Clin Biochem 6:24–30
Anurag K, Anand S, Kanwaljit C (2009) Attenuation of reno-inflammatory cascade in experimental model of diabetic nephropathy by sesamol. J Agric Food Chem 57(14):6123–6128
Greg H, Terri J (2007) Methods in renal research: Rodent models of streptozotocin-induced diabetic nephropathy. Nephrol 12(3):261–266
Sassy-Prigent C, Heudes D, Jouquey S, Auberval D, Belair MF, Michel O et al (1995) Morphometric detection of incipient glomerular lesions in diabetic nephropathy in rats. Protective effects of ACE inhibition. Lab Invest 73:64–71
Godkar PB (1994) Clinical biochemistry: principles and practice, 3rd edn. Bhalani Publishing House, Bombay, pp 115–116
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissue by thiobarbituric acid reaction. Anal Biochem 95:351–355
Beutler E, Duron O, Kelly BM (1963) Improved method for determination of blood glutathione. J Lab Clin Med 61(5):882–888
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186(1):189–192
Yokozawa T, Nakagawa T, Wakaki K, Koizumi F (2001) Animal model of diabetic nephropathy. Exp Toxicol Pathol 53:359–363
Pitchai B, Vishal C, Vijay K, Akash J, Jayarami R, Manjeet S (2008) Experimental models for nephropathy. J Renin Angiotensin Aldosterone Syst 9:189–195
Gojo A, Utsunomiya K, Taniguchi K (2007) The Rho-kinase inhibitor, fasudil, attenuates diabetic nephropathy in streptozotocin-induced diabetic rats. Eur J Pharmacol 568:242–247
Singh J, Budhiraja S, Lal H, Arora BR (2006) Renoprotection by telmisartan versus benazepril in streptozotocin induced diabetic nephropathy. Iran J Pharmacol Ther 5:135–139
Larkins RG, Dunlop ME (1992) The link between hyperglycemia and diabetic nephropathy. Diabetologia 35:499–504
Murali B, Dhananjay N, Umrani G, Ramesh G (2003) Effect of chronic treatment with losartan on streptozotocin-induced renal dysfunction. Mol Cell Biochem 249:85–90
Cooper ME (1998) Pathogenesis, prevention and treatment of diabetic nephropathy. Lancet 352:213–219
Yebe-nishimura C (1998) Aldose reductase in glucose toxicity: a potential target for the prevention of diabetic complications. Pharmacol Rev 50:21–33
Alhouli MJ, Thomson M, Al-Qattan K, Muslim Ali DJS (2010) Biochemical and histological changes in the kidney of streptozotocin-induced diabetic rats: effects of garlic and ginger compared to aspirin. FASEB J 24(Meeting abstract Supplement):659.1
Bjorn S, Stefan H, George J, Bo-Lennart J, Karin E, Britta I et al (2005) C-peptide prevents glomerular hypertrophy and mesangial matrix expansion in diabetic rats. Nephrol Dial Transplant 20:532–538
Derubertis FR, Craven PA (1994) Activation of protein kinase C in glomerular cells in diabetes: mechanisms and potential links to the pathogenesis of diabetic glomerulopathy. Diabetes 43:1–8
Saito T (1997) Abnormal lipid metabolism and renal disorders. Tohoku J Exp Med 181:321–333
Kramer-Guth A, Quaschning T, Greiber S, Wanner C (1996) Potential role of lipids in the progression of diabetic nephropathy. Clin Nephrol 46:262–265
Morgensen CE (1982) Long term antihypertensive treatment inhibiting progression of diabetic nephropathy. Br Med J 285:685–688
Bell DS (1991) Diabetic nephropathy: changing concepts of pathogenesis and treatment. Am J Med Sci 301:195–200
Williamson JR, Chang K, Frangos M, Hasan KS et al (1993) Hyperglycemic pseudohypoxia and diabetic complications. Diabetes 42(6):801–813
Ahmed FN, Naqvi FN, Shafiq F (2006) Lipid peroxidation and serum antioxidant enzymes in patients with type 2 diabetes mellitus. Ann N Y Acad Sci 1084:481–489
Evans J, Goldfine I, Maddux B, Grodsky G (2002) Oxidative stress and stress activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr Rev 23:599–622
Aksoy N, Vural H, Sabuncu T, Aksoy S (2003) Effects of melatonin on oxidative antioxidative status of tissues in streptozotocin induced diabetic rats. Cell Biochem Funct 21(2):121–125
Otsyula M, King MS, Ketcham TG, Sanders RA, Watkins JB (2003) Oxidative stress in rats after 60 days of hypergalactosemia or hyperglycemia. Int J Toxicol 22(6):423–427
Hazem H, Kataya H, Alaa Eldin A (2008) Red cabbage (Brassica oleracea) ameliorates diabetic nephropathy in rats. eCAM 5(3):281–287
Moore K, Roberts LJ (1998) Measurement of lipid peroxidation. Free Radic Res 28(6):659–671
Sundaram R, Mitra SK (2007) Antioxidant activity of ethyl acetate soluble fraction of Acacia arabica bark in rats. Ind J Pharmacol 39:33–38
Seyer-Hansen K (1976) Renal nephropathy in streptozotocin diabetic rats. Clin Sci Mol Med 51:551–555
Kang SS, Fears R, Noirot S, Mbanya JN, Yudkin J (1982) Changes in metabolism of rat kidney and liver caused by experimental diabetes by dietary sucrose. Diabetologia 22:285–288
Yotsumoto T, Naitoh T, Tanaka S (1997) Effects of specific antagonist of angiotensin II receptors and captopril on diabetic nephropathy in mice. Jpn J Pharmacol 75:59–64
Thrailkill KM, Clay Bunn R, Fowlkes JL (2009) Matrix metalloproteinases: their potential role in the pathogenesis of diabetic nephropathy. Endocrine 35(1):1–10
Acknowledgments
The authors wish to thank Prof. M.N. Navale, Founder President, Sinhgad Technical Education Society, Pune and Dr K.S. Jain, Principal, Sinhgad College of Pharmacy, Vadgaon, Pune, India for providing facilities to carryout this work.
Conflict of interest
All authors reported that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baig, M.A., Gawali, V.B., Patil, R.R. et al. Protective effect of herbomineral formulation (Dolabi) on early diabetic nephropathy in streptozotocin-induced diabetic rats. J Nat Med 66, 500–509 (2012). https://doi.org/10.1007/s11418-011-0614-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-011-0614-y